Abstract
Biomaterial-associated infections are the major cause of implant failure and can also develop many years after implantation. Recent advances in nanotechnology and the development of new nano-materials have led to the design of anti-biofilm coatings on implant surfaces. In this study, the inhibition of biofilm formation on silver-doped hydroxyapatite (Ag-HAp) nanoparticles-coated glass slides is reported. Ag-HAp was synthesized using low-temperature-modified sol–gel method. The release of silver ions from the Ag-HAp reduced the adhesion and prevented formation of biofilms of Escherichia coli, Staphylococcus epidermidis and Pseudomonas aeruginosa when studied over a 10-day period. These coatings released an initial high amount of silver ions followed by a slow and gradual release facilitating sustained anti-biofilm activity. This initial high release of silver is beneficial for reducing bacterial adhesion which is the first step in the development of a biofilm. The biocompatibility of silver-doped hydroxyapatite coatings has also been confirmed. As success or failure of an implant depends on the balance between host tissue integration and bacterial colonization, the competitive growth between the mammalian and bacterial cells by inoculating E. coli into actively growing MG63 osteosarcoma cells on Ag-HAp coatings has been investigated. This could mimic the peri-operative contamination model.
Graphical Abstract
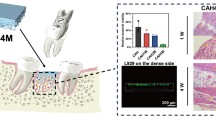
Ag-HAp-coated slides exhibit anti-biofilm activity over a period of 10 days, while supporting the growth of osteosarcoma cells (MG63). Co-culturing of MG63 cells with bacteria showed healthy growth of MG63 cells and no growth of bacteria. HAp control did not support growth of MG63 cells in the presence of bacteria.









Similar content being viewed by others
References
Trampuz A, Widmer AF (2006) Infections associated with orthopedic implants. Curr Opin Infect Dis 19(4):349–356
Maathuis PG, Neut D, Busscher HJ, van der Mei HC, van Horn JR (2005) Perioperative contamination in primary total hip arthroplasty. Clin Orthop Relat Res 433:136–139
Costerton J, Montanaro L, Arciola C (2005) Biofilm in implant infections: its production and regulation. Int J Artif Organs 28(11):1062–1068
Danese PN (2002) Antibiofilm approaches: prevention of catheter colonization. Chem Biol 9(8):873–880
Mah T-FC, O’Toole GA (2001) Mechanisms of biofilm resistance to antimicrobial agents. Trends Microbiol 9(1):34–39
Vasilev K, Cook J, Griesser HJ (2009) Antibacterial surfaces for biomedical devices. Expert Rev Med Devises 6(5):553–567
Abdelghany SM, Quinn DJ, Ingram RJ, Gilmore BF, Donnelly RF, Taggart CC, Scott CJ (2012) Gentamicin-loaded nanoparticles show improved antimicrobial effects towards Pseudomonas aeruginosa infection. Int J Nanomed 7:4053
Wan Y, Raman S, He F, Huang Y (2007) Surface modification of medical metals by ion implantation of silver and copper. Vacuum 81(9):1114–1118
Ersek RA, Denton DR (1984) Silver-impregnated porcine xenografts for treatment of meshed autografts. Ann Plast Surg 13(6):482–487
MacKeen PC, Person S, Warner SC, Snipes W, Stevens S (1987) Silver-coated nylon fiber as an antibacterial agent. Antimicrob Agents Chemother 31(1):93–99
Liedberg H, Lundeberg T (1990) Silver alloy coated catheters reduce catheter-associated bacteriuria. Br J Urol 65(4):379–381
Bosetti M, Masse A, Tobin E, Cannas M (2002) Silver coated materials for external fixation devices: in vitro biocompatibility and genotoxicity. Biomaterials 23(3):887–892
Furno F, Morley KS, Wong B, Sharp BL, Arnold PL, Howdle SM, Bayston R, Brown PD, Winship PD, Reid HJ (2004) Silver nanoparticles and polymeric medical devices: a new approach to prevention of infection? J Antimicrob Chemother 54(6):1019–1024
Stobie N, Duffy B, McCormack DE, Colreavy J, Hidalgo M, McHale P, Hinder SJ (2008) Prevention of Staphylococcus epidermidis biofilm formation using a low-temperature processed silver-doped phenyltriethoxysilane sol–gel coating. Biomaterials 29(8):963–969
Babapour A, Yang B, Bahang S, Cao W (2011) Low-temperature sol–gel-derived nanosilver-embedded silane coating as biofilm inhibitor. Nanotechnology 22(15):155602
Naik K, Kowshik M (2014) Anti-biofilm efficacy of low temperature processed AgCl–TiO 2 nanocomposite coating. Mater Sci Eng C 34:62–68
Stanić V, Janaćković D, Dimitrijević S, Tanasković SB, Mitrić M, Pavlović MS, Krstić A, Jovanović D, Raičević S (2011) Synthesis of antimicrobial monophase silver-doped hydroxyapatite nanopowders for bone tissue engineering. Appl Surf Sci 257(9):4510–4518
Rameshbabu N, Sampath Kumar T, Prabhakar T, Sastry V, Murty K, Prasad Rao K (2007) Antibacterial nanosized silver substituted hydroxyapatite: synthesis and characterization. J Biomed Mater Res Part A 80(3):581–591
Jadalannagari S, Deshmukh K, Ramanan SR, Kowshik M (2014) Antimicrobial activity of hemocompatible silver doped hydroxyapatite nanoparticles synthesized by modified sol–gel technique. Appl Nanosci 4(2):133–141
Chen Y, Zheng X, Xie Y, Ding C, Ruan H, Fan C (2008) Anti-bacterial and cytotoxic properties of plasma sprayed silver-containing HA coatings. J Mater Sci Mater Med 19(12):3603–3609
Jeon H-J, Yi S-C, Oh S-G (2003) Preparation and antibacterial effects of Ag–SiO 2 thin films by sol–gel method. Biomaterials 24(27):4921–4928
Babapour A, Akhavan O, Azimirad R, Moshfegh A (2006) Physical characteristics of heat-treated nano-silvers dispersed in sol–gel silica matrix. Nanotechnology 17(3):763
Jadalannagari S, More S, Kowshik M, Ramanan SR (2011) Low temperature synthesis of hydroxyapatite nano-rods by a modified sol–gel technique. Mater Sci Eng C 31(7):1534–1538
Ciobanu CS, Iconaru SL, Chifiriuc MC, Costescu A, Le Coustumer P, Predoi D (2013) Synthesis and antimicrobial activity of silver-doped hydroxyapatite nanoparticles. BioMed Res Int 2013:916218. doi:10.1155/2013/916218
Tsukamoto M, Ando Y, Noda I, Akiyama T, Eto S, Yonekura Y, Kawano S, Sonohata M, Miyamoto H, Mawatari M (2013) The hydroxyapatite coating containing silver inhibits the biofilm formation in a flow condition for two weeks. Bone Jt J 95(Supp. 15):352
Klausen M, Heydorn A, Ragas P, Lambertsen L, Aaes-Jørgensen A, Molin S, Tolker-Nielsen T (2003) Biofilm formation by pseudomonas aeruginosa wild type, flagella and type IV pili mutants. Mol Microbiol 48(6):1511–1524
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65(1):55–63
Kim IS, Kumta PN (2004) Sol–gel synthesis and characterization of nanostructured hydroxyapatite powder. Mater Sci Eng B 111(2):232–236
Brett DW (2006) A discussion of silver as an antimicrobial agent: alleviating the confusion. Ostomy/Wound Manag 52(1):34–41
Kumar R, Münstedt H (2005) Polyamide/silver antimicrobials: effect of crystallinity on the silver ion release. Polym Int 54(8):1180–1186
Chaw K, Manimaran M, Tay FE (2005) Role of silver ions in destabilization of intermolecular adhesion forces measured by atomic force microscopy in Staphylococcus epidermidis biofilms. Antimicrob Agents Chemother 49(12):4853–4859
Kalishwaralal K, BarathManiKanth S, Pandian SRK, Deepak V, Gurunathan S (2010) Silver nanoparticles impede the biofilm formation by Pseudomonas aeruginosa and Staphylococcus epidermidis. Colloids Surf B 79(2):340–344
Krall D-IT (1999) A new technology of microdispersed silver in polyurethane induces antimicrobial activity in central venous catheters. Infection 27(1):S16–S23
Donlan RM, Costerton JW (2002) Biofilms: survival mechanisms of clinically relevant microorganisms. Clin Microbiol Rev 15(2):167–193
Gollwitzer H, Ibrahim K, Meyer H, Mittelmeier W, Busch R, Stemberger A (2003) Antibacterial poly (D, L-lactic acid) coating of medical implants using a biodegradable drug delivery technology. J Antimicrob Chemother 51(3):585–591
Pavithra D, Doble M (2008) Biofilm formation, bacterial adhesion and host response on polymeric implants—issues and prevention. Biomed Mater 3(3):034003
Stahl J-P, Dupon M, Dutronc H, Perpoint T (2010) Recommendations for bone and joint prosthetic device infections in clinical practice (prosthesis, implants, osteosynthesis). Med et mal Infect 40(4):185–211
Dickinson GM, Bisno AL (1989) Infections associated with indwelling devices: concepts of pathogenesis; infections associated with intravascular devices. Antimicrob Agents Chemother 33(5):597
Acknowledgments
The authors are grateful to Mr. Aarif, NIO, Goa, for their assistance with SEM and EDAX analysis and Dr. K. M. Paknikar, ARI, Pune, for help with AAS studies for silver release experiments.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Deshmukh, K.R., Ramanan, S.R. & Kowshik, M. Low-temperature-processed biocompatible Ag-HAp nanoparticles with anti-biofilm efficacy for tissue engineering applications. J Sol-Gel Sci Technol 80, 738–747 (2016). https://doi.org/10.1007/s10971-016-4149-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10971-016-4149-2