Abstract
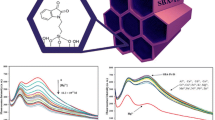
A novel organic–inorganic hybrid nanomaterial (SBA-15-CA) was prepared by covalent immobilization of chromotropic acid onto the surface of mesoporous silica material SBA-15. Different techniques such as XRD, TEM, FT-IR, N2 adsorption–desorption and TGA analyses were employed to characterize the grafting process. The data showed that the organic moiety (0.41 mmol g−1) was successfully grafted to the SBA-15 and the primary hexagonally ordered mesoporous structure of SBA-15 was preserved after the grafting procedure. SBA-15-CA has been realized as a highly sensitive and selective fluorescent probe towards Fe3+ and I− ions in aqueous media. SBA-15-CA exhibited a remarkable fluorescent quenching in the presence of Fe3+ ion over other competitive cations including Na+, Mg2+, Al3+, K+, Ca2+, Cr3+, Mn2+, Fe2+, Co2+, Ni2+, Cu2+, Zn2+, Cd2+, Hg2+, and Pb2+ as well as I− ion among a series of anions including F−, Cl−, Br−, CO3 2−, HCO3 −, CN−, NO3 −, NO2 −, SCN−, SO4 2−, H2PO4 −, HPO4 2−, and CH3COO−. A good linear response was observed between the concentration of the quenchers (Fe3+ and I− ions) and fluorescence intensity of SBA-15-CA with detection limits of 1.5 × 10−7 M for Fe3+ and 0.2 × 10−7 M for I−. Moreover, the effects of various pH values on the sensing ability of SBA-15-CA were investigated. Finally, the proposed method was successfully utilized for the determination of Fe3+ and I− ions in river water, well water and tap water samples.











Similar content being viewed by others
References
A. Chmyrov, T. Sandén, J. Widengren, J. Phys. Chem. B 114, 11282 (2010)
N. Kumar, V. Bhalla, M. Kumar, Coord. Chem. Rev. 257, 2335 (2013)
S.K. Kim, J.L. Sessler, Chem. Soc. Rev. 39, 3784 (2010)
R. Martínez-Máñez, F. Sancenón, Chem. Rev. 103, 4419 (2003)
H. Zhu, J. Fan, B. Wang, X. Peng, Chem. Soc. Rev. 44, 4337 (2015)
M. Wenzel, J.R. Hiscock, P.A. Gale, Chem. Soc. Rev. 41, 480 (2012)
R.A. Bissell, A.P. de Silva, H.N. Gunaratne, P.M. Lynch, G.E. Maguire, C.P. McCoy, S.K. Sandanayake, Photoinduced Electron Transfer V (Springer, Berlin, 1993), p. 223
E. Beutler, V. Felitti, T. Gelbart, N. Ho, Drug Metab. Dispos. 29, 495 (2001)
W.A. Jefferies, D.L. Dickstein, M. Ujiie, J. Alzheimer’s Dis. 3, 339 (2001)
F.L. Martin, S.J. Williamson, K.E. Paleologou, R. Hewitt, O. El-Agnaf, D. Allsop, J. Neurochem. 87, 620 (2003)
G. Aumont, J.-C. Tressol, Analyst 111, 841 (1986)
D. Nacapricha, P. Sangkarn, C. Karuwan, T. Mantim, W. Waiyawat, P. Wilairat, T. Cardwell, I. McKelvie, N. Ratanawimarnwong, Talanta 72, 626 (2007)
F. Azizi, M. Hedayati, M. Rahmani, R. Sheikholeslam, S. Allahverdian, N. Salarkia, J. Endocrinol. Invest. 28, 23 (2005)
G.-P. Tao, Q.-Y. Chen, X. Yang, K.-Y. Wang, Dyes Pigm. 95, 338 (2012)
A. Goel, S. Umar, P. Nag, A. Sharma, L. Kumar, Z. Hossain, J.R. Gayen, A. Nazir, Chem. Commun. 51, 5001 (2015)
M. Wang, G. Meng, Q. Huang, Q. Xu, G. Liu, Anal. Methods 4, 2653 (2012)
J. Liu, Y.-Q. Xie, Q. Lin, B.-B. Shi, P. Zhang, Y.-M. Zhang, T.-B. Wei, Sens. Actuators B 186, 657 (2013)
Z.B. Shang, Y. Wang, W.J. Jin, Talanta 78, 364 (2009)
Z. Li, H. Yu, T. Bian, Y. Zhao, C. Zhou, L. Shang, Y. Liu, L.-Z. Wu, C.-H. Tung, T. Zhang, J. Mater. Chem. C 3, 1922 (2015)
M. Zhang, B.-C. Ye, Chem. Commun. 48, 3647 (2012)
C. Yi, B. Song, W. Tian, X. Cui, Q. Qi, W. Jiang, Z. Qi, Y. Sun, Tetrahedron Lett. 55, 5119 (2014)
D.T. Quang, J.S. Kim, Chem. Rev. 110, 6280 (2010)
K.B. Kim, H. Kim, E.J. Song, S. Kim, I. Noh, C. Kim, Dalton Trans. 42, 16569 (2013)
T.-B. Wei, P. Zhang, B.-B. Shi, P. Chen, Q. Lin, J. Liu, Y.-M. Zhang, Dyes Pigm. 97, 297 (2013)
G.-y. Gao, W.-j. Qu, B.-b. Shi, P. Zhang, Q. Lin, H. Yao, W.-l. Yang, Y.-m. Zhang, T.-b. Wei, Spectrochim. Acta Part A 121, 514 (2014)
Z. Aydin, Y. Wei, M. Guo, Inorg. Chem. Commun. 20, 93 (2012)
Y.R. Bhorge, H.-T. Tsai, K.-F. Huang, A.J. Pape, S.N. Janaki, Y.-P. Yen, Spectrochim. Acta Part A 130, 7 (2014)
N. Singh, D.O. Jang, Org. Lett. 9, 1991 (2007)
M. Vetrichelvan, R. Nagarajan, S. Valiyaveettil, Macromolecules 39, 8303 (2006)
H.J. Jung, N. Singh, D.Y. Lee, O.D. Jang, Tetrahedron Lett. 51, 3962 (2010)
T. Asefa, C.T. Duncan, K.K. Sharma, Analyst 134, 1980 (2009)
E. Rampazzo, E. Brasola, S. Marcuz, F. Mancin, P. Tecilla, U. Tonellato, J. Mater. Chem. 15, 2687 (2005)
S.A. El-Safty, M.A. Shenashen, Sens. Actuators B 183, 58 (2013)
R. Gong, H. Mu, Y. Sun, X. Fang, P. Xue, E. Fu, J. Mater. Chem. B 1, 2038 (2013)
H. Lou, Y. Zhang, Q. Xiang, J. Xu, H. Li, P. Xu, X. Li, Sens. Actuators B 166, 246 (2012)
M. Karimi, A. Badiei, N. Lashgari, J. Afshani, G.M. Ziarani, J. Lumin. 168, 1 (2015)
M. Ghaedi, K. Mortazavi, M. Montazerozohori, A. Shokrollahi, M. Soylak, Mater. Sci. Eng. C 33, 2338 (2013)
P. Bermejo-Barrera, A. Moreda-Pineiro, M. Aboal-Somoza, J. Moreda-Pineiro, A. Bermejo-Barrera, J. Anal. At. Spectrom. 9, 483 (1994)
B. Michalke, H. Witte, J. Trace Elem. Med. Biol. 29, 63 (2015)
N. Krebs, C. Langkammer, W. Goessler, S. Ropele, F. Fazekas, K. Yen, E. Scheurer, J. Trace Elem. Med. Biol. 28, 1 (2014)
M. Lu, R.G. Compton, Electroanalysis 25, 1123 (2013)
L. Yang, L. Zou, G. Li, B. Ye, Talanta 147, 634 (2016)
M. Karimi, A. Badiei, G.M. Ziarani, J. Fluoresc. 25, 1297 (2015)
J. Afshani, A. Badiei, M. Karimi, N. Lashgari, G.M. Ziarani, J. Fluoresc. 26, 263 (2016)
J. Afshani, A. Badiei, N. Lashgari, G.M. Ziarani, RSC Adv. 6, 5957 (2016)
M. Karimi, A. Badiei, G.M. Ziarani, Inorg. Chim. Acta 450, 346 (2016)
M. Karimi, A. Badiei, G.M. Ziarani, RSC Adv. 5, 36530 (2015)
N. Lashgari, A. Badiei, G.M. Ziarani, J. Phys. Chem. Solids 103, 238 (2017)
H.I. Lee, J.H. Kim, G.D. Stucky, Y. Shi, C. Pak, J.M. Kim, J. Mater. Chem. 20, 8483 (2010)
J. Mondal, A. Modak, M. Nandi, H. Uyama, A. Bhaumik, RSC Adv. 2, 11306 (2012)
P. Zarabadi-Poor, A. Badiei, A.A. Yousefi, J. Barroso-Flores, J. Phys. Chem. C 117, 9281 (2013)
J.R. Lakowicz, Principles of Fluorescence Spectroscopy (Springer Science & Business Media, New York, 2013)
D.Y. Lee, N. Singh, O.D. Jang, Tetrahedron Lett. 51, 1103 (2010)
W. Lin, L. Long, L. Yuan, Z. Cao, J. Feng, Anal. Chim. Acta 634, 262 (2009)
K. Velmurugan, J. Prabhu, L. Tang, T. Chidambaram, M. Noel, S. Radhakrishnan, R. Nandhakumar, Anal. Methods 6, 2883 (2014)
Z. Yang, M. She, B. Yin, J. Cui, Y. Zhang, W. Sun, J. Li, Z. Shi, J. Org. Chem. 77, 1143 (2012)
L. Zhao, X. Xin, P. Ding, A. Song, Z. Xie, J. Shen, G. Xu, Anal. Chim. Acta 926, 99 (2016)
N. Lashgari, A. Badiei, G. Mohammadi Ziarani, J. Fluoresc. 26, 1885 (2016)
M. Yadavi, A. Badiei, J. Fluoresc. 24, 523 (2014)
J.-Q. Wang, L. Huang, M. Xue, Y. Wang, L. Gao, J.H. Zhu, Z. Zou, J. Phys. Chem. C 112, 5014 (2008)
Y. Xiang, A. Tong, Org. Lett. 8, 1549 (2006)
R. Nudelman, O. Ardon, Y. Hadar, Y. Chen, J. Libman, A. Shanzer, J. Med. Chem. 41, 1671 (1998)
K. Bowman-James, A. Bianchi, E. García-Espana, Anion Coordination Chemistry (Wiley, Weinheim, 2012)
B.L. Schottel, H.T. Chifotides, K.R. Dunbar, Chem. Soc. Rev. 37, 68 (2008)
Acknowledgements
The authors thank the research council of University of Tehran for financial support.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karimi, M., Badiei, A., Lashgari, N. et al. A chromotropic acid modified SBA-15 as a highly sensitive fluorescent probe for determination of Fe3+ and I− ions in water. J Porous Mater 25, 137–146 (2018). https://doi.org/10.1007/s10934-017-0427-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-017-0427-9