Abstract
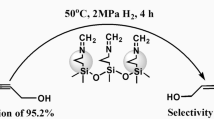
The dual-modified SBA-15 materials (A–S–Si) with Al ions inserting in the framework and −SO3H groups grafting on the pore wall were prepared via a co-condensation method and characterized by a series of techniques. The characterization results implied that the A–S–Si samples have typically mesoporous structure and different acid properties. The catalytic performance of A–S–Si catalysts in the additive reaction of acetic acid with isobutene was studied. The results showed that the conversion of acetic acid strongly depended on the Brönsted acidity, while the selectivity of tert-butyl acetate (TBAC) was linear correlated with the ratio of Brönsted and Lewis acid amounts. The conversion of acetic acid could reach 72 % with the selectivity of TBAC as high as 97 % over A5–S10–Si with balanced Brönsted and Lewis acid under the optimal conditions. An additional advantage was that the dual-modified catalyst can be recycled and regenerate with good stability.










Similar content being viewed by others
References
K. Nagasawa, Synth. Commun. 13, 2033 (1990)
G.W. Anderson, F.M. Callaham, J. Am. Chem. Soc. 82, 3359 (1960)
D. Luo, R. Corey, R. Propper, J. Collins, A. Komorniczak, M. Davis, N. Berger, S. Lum, Environ. Sci. Policy 14, 585 (2011)
W. Faber, D. Kirkpatrick, P. Coder, A. Li, S. Borghoff, M. Banton, Regul. Toxicol. Pharmacol. 68, 332 (2014)
F.K. Behbahani, M. Farahani, H.A. Oskooie, J. Korean Chem. Soc. 55, 633 (2011)
Y. Du, C. Li, X. Sun, X. Liang, C. Qi, Asia-Pac. J. Chem. Eng. 6, 933 (2011)
F. Rajabi, Tetrahedron Lett. 50, 395 (2009)
Z. Duan, Y. Gu, Y. Deng, J. Mol. Catal. A: Chem. 246, 70 (2006)
K. Nagasawa, K. Ohhashi, A. Yamashita, K. Ito, Chem. Lett. 23, 209 (1994)
E.M. Kaiser, R.A. Woodruff, J. Org. Chem. 4, 1198 (1970)
S. Takimoto, Bull. Chem. Soc. Jpn 8, 2335 (1976)
A. Armstrong, Tetrahedron Lett. 20, 2483 (1988)
B.T. Dorris, J. Sowa, A. Nieuwland, J. Am. Chem. Soc. 56, 2689 (1934)
H.A. Bruson, T.W. Riener, J. Am. Chem. Soc. 67, 723 (1945)
I.J. Dijs, H.L.F. van Ochten, C.A. van Walree, J.W. Geus, J. Mol. Catal. A: Chem. 188, 209 (2002)
D.R. Cova, C.W. Roos, Ind. Eng. Chem. Prod. Res. Dev. 7, 301 (1968)
L.O. Raether, A.J. Lauck, H.R. Gamrath, Ind. Eng. Chem. Prod. Res. Dev. 2, 133 (1963)
A. Heidekum, M.A. Harmer, W.F. Hoelderich, J. Catal. 181, 217 (1999)
J.D. Kemp, E. Cerrito, US Patent 3678099 (1972)
M.P. Kapoor, S. Inagaki, Bull. Chem. Soc. Jpn 79, 1463 (2006)
S. Wang, Microporous Mesoporous Mater. 117, 1 (2009)
J. Lee, J. Kim, T. Hyeon, Adv. Mater. 18, 2073 (2006)
F. Zhou, J. Tang, X. Chen, Z. Fei, M. Cui, X. Qiao, Acta Pet. Sin. 30, 629 (2014)
C.A. Grande, A.E. Rodrigues, Ind. Eng. Chem. Res. 43, 8057 (2004)
D. Vargas-Hernández, M.A. Pérez-Cruz, R. Hernández-Huesca, Adsorption 21, 153 (2015)
A. Galarneau, A. Barodawalla, T.J. Pinnavaia, Nature 374, 529 (1995)
A. Galarneau, D. Desplantier-Giscard, F. di Renzo, F. Fajula, Catal. Today 68, 191 (2001)
F. Zhou, J. Tang, Z. Fei, X. Chen, M. Cui, X. Qiao, J. Porous Mater. 21, 149 (2014)
P. Bhange, D.S. Bhange, S. Pradhan, V. Ramaswamy, Appl. Catal. A Gen. 400, 176 (2011)
Y. Zheng, J. Li, N. Zhao, W. Wei, Y. Sun, Fine Chem. 23, 972 (2006)
S. Lin, L. Shi, M.M.L. Ribeiro Carrott, P.J.M. Carrott, J. Rocha, M.R. Li, X.D. Zou, Microporous Mesoporous Mater. 142, 526 (2011)
R. van Grieken, G. Calleja, G.D. Stucky, J.A. Melero, R.A. Garcia, J. Iglesias, Langmuir 19, 3966 (2003)
X. Yang, W. Dai, H. Chen, J. Xu, Y. Cao, H. Li, K. Fan, Appl. Catal. A Gen. 283, 1 (2005)
M.L. Martínez, M.B. Gómez Costa, G.A. Monti, O.A. Anunziata, Microporous Mesoporous Mater. 144, 183 (2011)
Y. Wang, Q. Zhang, T. Shishido, K. Takehira, J. Catal. 209, 186 (2002)
A.R. Silva, K. Wilson, J.H. Clark, C. Freire, Microporous Mesoporous Mater. 91, 128 (2006)
N. He, J. Cao, S. Bao, Q. Xu, Mater. Lett. 31, 133 (1997)
S.K. Badamali, A. Sakthivel, Catal. Lett. 65, 153 (2000)
B. Xue, Y. Li, L. Deng, Catal. Commun. 10, 1609 (2009)
A. Ungureanu, B. Dragoi, V. Hulea, T. Cacciaguerra, D. Meloni, V. Solinas, Microporous Mesoporous Mater. 163, 51 (2012)
C.A. Emeis, J. Catal. 141, 347 (1993)
Acknowledgments
Natural Science Foundation of China (21276126, 21306089), Jiangsu Province Higher Education Natural Science Foundation of Jiangsu Province (13KJB530006).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Chen, X., Feng, J., Fei, Z. et al. Synthesis of tert-butyl acetate via eco-friendly additive reaction over mesoprous silica catalysts with balanced Brönsted and Lewis acid sites. J Porous Mater 23, 255–262 (2016). https://doi.org/10.1007/s10934-015-0077-8
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10934-015-0077-8