Abstract
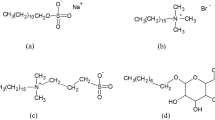
The inherently present seventeen disulfide bonds of the circulatory protein, human serum albumin (HSA) provide the necessary structural stability. Various spectroscopic approaches were used to investigate the effect of reduction of these disulfide bonds and its binding with the anionic surfactant, sodium dodecyl sulfate (SDS). Based on several spectroscopic analyses, our investigations highlight the following interesting aspects: (1) HSA on reduction loses not only its tertiary structure but also a significant amount of secondary structure as well. However, the reduced state of the protein is not like the molten-globule, (2) this structural loss of the protein due to reduction is more prominent than that caused by higher SDS concentrations alone and can certainly be attributed to the role of disulfide bonds, (3) lower surfactant concentrations provide marginal structural rigidity to the native state of the protein, whereas, higher concentrations of SDS induces secondary structure to the reduced state of HSA, (4) the binding of SDS with both the native and reduced states of HSA, occurred in three distinct stages which was followed by a saturation stage. However, the nature of such binding is different for both the states as investigated by using the Stern–Volmer equations and estimating the thermodynamic parameters. Besides, in contrast to the native state, the reduced state of HSA shows that the lone tryptophan residue gets more buried. However, there occurs a sudden decrement in the lifetime of the tryptophan and the hydrodynamic diameter increases by twofold.





Similar content being viewed by others
Abbreviations
- HSA:
-
Human serum albumin
- SDS:
-
Sodium dodecyl sulfate
- BSA:
-
Bovine serum albumin
- DTT:
-
Dithiothretol
- Trp:
-
Tryptophan
- Cys:
-
Cysteine
- DLS:
-
Dynamic light scattering
- CD:
-
Circular dichroism
- DTNB:
-
5,5′-Dithiobis(2-nitrobenzoic acid)
- UV–Vis:
-
Ultra violet–visible
- dH:
-
Hydrodynamic diameter
References
Shaw BF, Schneider GF, Arthanari H, Narovlyansky M, Moustakas DT, Durazo A, Wagner G, Whitesides GM (2011) Complexes of native ubiquitin and dodecyl sulfate illustrate the nature of hydrophobic and electrostatic interactions in the binding of proteins and surfactants. J Am Chem Soc 133:17681–17695
Mukherjee S, Sen P, Halder A, Sen S, Dutta P, Bhattacharyya K (2003) Solvation dynamics in a protein–surfactant aggregate. TNS in HSA–SDS. Chem Phys Lett 379:471–478
Mandal U, Ghosh S, Mitra G, Adhikary A, Dey S, Bhattacharyya K (2008) A femtosecond study of the interaction of human serum albumin with a surfactant (SDS). Chem Asian J 3:1430–1434
Sen P, Dutta P, Halder A, Mukherjee S, Sen S, Bhattacharyya K (2003) Solvation dynamics in a protein–surfactant complex. Chem Phys Lett 377:229–235
Sahu K, Mondal SK, Roy D, Karmakar R, Bhattacharyya K (2005) Study of interaction of a cationic protein with a cationic surfactant using solvation dynamics: lysozyme: CTAB. Chem Phys Lett 413:484–489
Sahu K, Roy D, Mondal SK, Karmakar R, Bhattacharyya K (2005) Study of protein–surfactant interaction using excited state proton transfer. Chem Phys Lett 404:341–345
Anand U, Jash C, Mukherjee S (2010) Spectroscopic probing of the microenvironment in a protein–surfactant assembly. J Phys Chem B 114:15839–15845
Anand U, Jash C, Boddepalli RK, Shrivastava A, Mukherjee S (2011) Exploring the mechanism of fluorescence quenching in proteins induced by tetracycline. J Phys Chem B 115:2320–6312
Takeda K, Sasa K, Kawamoto K, Wada A, Aoki K (1988) Secondary structure changes of disulfide bridge-cleaved bovine serum albumin in solutions of urea, guanidine hydrochloride, and sodium dodecyl sulfate. J Colloid Int Sci 124:284–289
Takeda K, Moriyama Y (2007) Comment on the misunderstanding of the BSA–SDS complex model: concern about publications of an impractical model. J Phys Chem B 111:1244
Patel S, Datta A (2007) Steady state and time-resolved fluorescence investigation of the specific binding of two chlorin derivatives with human serum albumin. J Phys Chem B 111:10557–10562
Krishnakumar SS, Panda D (2002) Spatial relationship between the prodan site, Trp-214, and Cys-34 residues in human serum albumin and loss of structure through incremental unfolding. Biochemistry 41:7443–7452
Santra MK, Banerjee A, Rahaman O, Panda D (2005) Unfolding pathways of human serum albumin: evidence for sequential unfolding and folding of its three domains. Int J Biol Macromol 37:200–204
Hazra P, Chakrabarty D, Chakraborty A, Sarkar N (2004) Probing protein–surfactant interaction by steady state and time-resolved fluorescence spectroscopy. Biochem Bhiophys Res Commun 314:543–549
Chakraborty A, Seth D, Setua P, Sarkar N (2006) Photoinduced electron transfer in a protein–surfactant complex: probing the interaction of SDS with BSA. J Phys Chem B 110:16607–16617
Duggan EL, Luck FM (1948) The combination of organic anions with serum albumin. J Biol Chem 172:205–220
Anand U, Jash C, Mukherjee S (2011) Protein unfolding and subsequent refolding: a spectroscopic investigation. Phys Chem Chem Phys 13:20418–20426
Wedemeyer WJ, Welker E, Narayan M, Scheraga HA (2000) Disulfide bonds and protein folding. Biochemistry 39:4207–4216
Creighton TE (1986) Disulfide bonds as probes of protein folding pathways. Methods Enzymol 131:83–106
Gekko K, Kimoto A, Kamiyama T (2003) Effects of disulfide bonds on compactness of protein molecules revealed by volume, compressibility, and expansibility changes during reduction. Biochemistry 42:13746–13753
Bramanti E, Lomonte C, Onor M, Zamboni R, Raspi G, D’Ulivo A (2004) Study of the disulfide reduction of denatured proteins by liquid chromatography coupled with on-line cold-vapor-generation atomic-fluorescence spectrometry (LC-CVGAFS). Anal Bioanal Chem 380:310–318
Lee JY, Hirose M (1992) Partially folded state of the disulfide-reduced form of human serum albumin as an intermediate for reversible denaturation. J Biol Chem 267:14753–14758
Anfinsen CB, Haber E, Sela M, White FH Jr (1961) The kinetics of formation of native ribonuclease during oxidation of the reduced polypeptide chain. Proc Natl Acad Sci USA 47:1309–1314
Creighton TE (1980) Role of the environment in the refolding of reduced pancreatic trypsin inhibitor. J Mol Biol 144:521–550
Kumar N, Kella D, Kinsella JE (1985) A method for the controlled cleavage of disulfide bonds in proteins in the absence of denaturants. J Biochem Biophys Methods 11:251–263
Raspi G, Lo Moro A, Spinetti M, Tesi G (1997) Radiochromatographic method for –SH determination in proteins by [203Hg] p-hydroxymercuribenzoate. Anal Commun 34:307–309
Means GE, Feeney RE (1971) Chemical modification of proteins. Holden Day, San Francisco
Fontana A, Toniolo C (1974) Detection and determination of thiols. In: Patai S (ed) The chemistry of the thiol group. Wiley, New York, p 272
Jocelyn P (1987) Chemical reduction of disulfides. Methods Enzymol 143:246–256
Singh R, Whitesides GM (1991) A reagent for reduction of disulfide bonds in proteins that reduces disulfide bonds faster than does dithiothreitol. J Org Chem 56:2332–2337
Anfinsen CB (1973) Principles that govern the folding of protein chains. Science 181:223–230
Tanford C, Kawahara K, Lapanje S (1967) Proteins as random coils. I. Intrinsic viscosities and sedimentation coefficients in concentrated guanidine hydrochloride. J Am Chem Soc 89:729–736
Kella NK, Kang YJ, Kinsella JE (1988) Effect of oxidative sulfitolysis of disulfide bonds of bovine serum albumin on its structural properties: a physicochemical study. J Protein Chem 7:535–548
al-Obeidi AM, Light A (1988) Size-exclusion high performance liquid chromatography of native trypsinogen, the denatured protein, and partially refolded molecules. Further evidence that non-native disulfide bonds are dominant in refolding the completely reduced protein. J Biol Chem 263:8642–8645
Volles MJ, Xu X, Scheraga HA (1999) Distribution of disulfide bonds in the two-disulfide intermediates in the regeneration of bovine pancreatic ribonuclease A: further insights into the folding process. Biochemistry 38:7284–7293
Zhou JM, Fan YX, Kihara H, Kimura K, Amemiya Y (1998) The compactness of ribonuclease A and reduced ribonuclease A. FEBS Lett 430:275–277
Chen L, Wildegger G, Kiefhaber T, Hodgson KO, Doniach S (1998) Kinetics of lysozyme refolding: structural characterization of a non-specifically collapsed state using time-resolved X-ray scattering. J Mol Biol 276:225–237
Ueki T, Hiragi Y, Kataoka M, Inoko Y, Amemiya Y, Izumi Y, Tagawa H, Muroga Y (1985) Aggregation of bovine serum albumin upon cleavage of its disulfide bonds, studied by the time-resolved small-angle X-ray scattering technique with synchrotron radiation. Biophys Chem 23:115–124
Peters T (1996) All about albumin: biochemistry, genetics, and medical applications. Academic, San Diego
Peters T (1985) Serum albumin. Adv Protein Chem 37:161–245
He XM, Carter DC (1992) Atomic structure and chemistry of human serum albumin. Nature 358:209–215
Brown JR (1977) In: Rosenoer VM, Oratz M, Rothschild MA (eds) Albumin structure, functions and uses. Pergamon Press, Oxford
Sugio S, Kashima A, Mochizuki S, Noda M, Kobayashi K (1999) Crystal structure of human serum albumin at 2.5 Å resolution. Protein Eng 12:439–446
Narazaki R, Maruyama T, Otagiri M (1997) Probing the cysteine 34 residue in human serum albumin using fluorescence techniques. Biochim Biophys Acta 1338:275–281
Diaz X, Abuin E, Lissi E (2003) Quenching of BSA intrinsic fluorescence by alkylpyridinium cations: its relationship to surfactant-protein association. J Photochem Photobiol A Chem 155:157–162
Santra MK, Banerjee A, Krishnakumar SS, Rahaman O, Panda D (2004) Multiple-probe analysis of folding and unfolding pathways of human serum albumin. Evidence for a framework mechanism of folding. Eur J Biochem 271:1789–1797
Brahma A, Mandal C, Bhattacharyya D (2005) Characterization of a dimeric unfolded state of bovine serum albumin under mildly acidic conditions. Biochim Biophys Acta 1751:159–169
Sridevi K, Udgaonkar JB (2002) Unfolding rates of barstar determined in native and low denaturant conditions indicate the presence of intermediates. Biochemistry 41:1568–1578
Chardot TP, Wedding RT (1992) Role of cysteine in activation and allosteric regulation of maize phosphoenolpyruvate carboxylase. Plant Physiol 98:780–783
Hong Y, Feng C, Yu Y, Liu J, Lam JWY, Luo KQ, Tang BZ (2010) Quantitation, visualization, and monitoring of conformational transitions of human serum albumin by a tetraphenylethene derivative with aggregation-induced emission characteristics. Anal Chem 82:7035–7043
Kumar Y, Tayyab S, Muzammil S (2004) Molten-globule like partially folded states of human serum albumin induced by fluoro and alkyl alcohols at low pH. Archives Biochem Biophys 426:3–10
Muzammil S, Kumar Y, Tayyab S (1999) Molten globule-like state of human serum albumin at low pH. Eur J Biochem 266:26–32
Smeller L, Meersman F, Heremans K (2008) Stable misfolded states of human serum albumin revealed by high-pressure infrared spectroscopic studies. Eur Biophys J 37:1127–1132
Varshney A, Ahmad B, Khan RH (2008) Comparative studies of unfolding and binding of ligands to human serum albumin in the presence of fatty acid: spectroscopic approach. Int. J Biol Macromol 42:483–490
Turro NJ, Lei XG, Ananthapadmanabhan KP, Aronson M (1995) Spectroscopic probe analysis of protein–surfactant Interactions: the BSA/SDS System. Langmuir 11:2525–2533
Lakowicz JR (2006) Principles of fluorescence spectroscopy, 3rd edn. Springer, Berlin
Pertich JW, Chang MC, McDonald DB, Fleming GR (1983) On the origin of nonexponential fluorescence decay in tryptophan and its derivatives. J Am Chem Soc 105:3824–3832
Yang ST, Marchio JL, Yen JW (1994) A dynamic light scattering study of beta-galactosidase: environmental effects on protein conformation and enzyme activity. Biotechnol Prog 10:525–531
Dev S, Surolia A (2006) Dynamic light scattering study of peanut agglutinin: size, shape and urea denaturation. J Biosci 31:551–556
Mitra RK, Sinha SS, Pal SK (2007) Hydration in protein folding: thermal unfolding/refolding of human serum albumin. Langmuir 23:10224–10229
Dubin PL, Principi JM (1998) Failure of universal calibration for size-exclusion chromatography of rodlike macromolecules vs. random coils and globular proteins. Macromolecules 22:1891–1896
Dubin PL, Edwards SL, Mehta MS, Tomalia D (1993) Quantitation of non-ideal behavior in protein size-exclusion chromatography. J Chromatogr 635:51–60
Das BK, Bhattacharyya T, Roy S (1995) Characterization of a urea induced molten globule intermediate state of glutaminyl-tRNA synthetase from escherichia coli. Biochemistry 34:5242–5247
Semisotnov GV, Rodionova NA, Razgulyaev OI, Uversky VN, Cripas AF, Cilmanshin RI (1991) Study of the “molten globule” intermediate state in protein folding by a hydrophobic fluorescent probe. Biopolymer 31:119–128
Eftink MR, Ghiron CA (1976) Exposure of tryptophanyl residues in proteins. Quantitative determination by fluorescence quenching studies. Biochemistry 15:672–680
Eftink MR, Ghiron CA (1976) Fluorescence quenching of indole and model micelle systems. J Phys Chem 80:486–493
Eftink MR, Ghiron CA (1977) Exposure of tryptophanyl residues and protein dynamics. Biochemistry 16:5546–5551
Lehrer SS (1971) Solute perturbation of protein fluorescence. Quenching of the tryptophyl fluorescence of model compounds and of lysozyme by iodide ion. Biochemistry 10:3254–3263
Chadborn N, Bryant J, Bain AJ, O’Shea P (1999) Ligand-dependent conformational equilibria of serum albumin revealed by tryptophan fluorescence quenching. Biophys J 76:2198–2207
Acknowledgments
We thank Professor Vinod Kumar Singh, Director IISER Bhopal for his constant encouragement and support. SM sincerely thanks Professor Sandeep Verma, IIT Kanpur for many stimulating discussions and suggestions. We sincerely thank Professor A. Dasgupta, University of Calcutta for allowing us to use the DLS instrumental facility. We are grateful to Professor Soumen Basak, Chemical Science Division, SINP, Kolkata for his guidance during the CD measurements. We also thank Dr. Nabendu S. Chatterjee, Biochemistry Division, National Institute of Cholera and Enteric Diseases, Kolkata, for FPLC (Fast Protein Liquid Chromatography) instrumental facility. UA thanks CSIR, SG thanks UGC for providing fellowship and SR thanks University of Calcutta for providing financial support. SM thanks the DST-Fast track scheme (No.: SR/FT/CS-19/2011) SERB for financial support.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Uttam Anand and Sutapa Ray have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Anand, U., Ray, S., Ghosh, S. et al. Structural Aspects of a Protein–Surfactant Assembly: Native and Reduced States of Human Serum Albumin. Protein J 34, 147–157 (2015). https://doi.org/10.1007/s10930-015-9606-1
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-015-9606-1