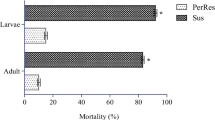
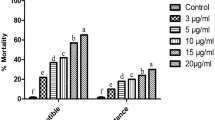
Abstract
Mosquito coils are insecticides commonly used for protection against mosquitoes due to their toxic effects on mosquito populations. These effects on mosquitoes could induce the expression of metabolic enzymes in exposed populations as a counteractive measure. Cytochrome P450 family 4 (CYP4) are metabolic enzymes associated with a wide range of biological activities including insecticide resistance. In this study, the efficacies of three commercial mosquito coils with different pyrethroid active ingredients were assessed and their potential to induce the expression of CYP4 genes in Aedes albopictus analyzed by real-time quantitative PCR. Coils containing 0.3 % d-allethrin and 0.005 % metofluthrin exacted profound toxic effects on Ae. albopictus, inducing high mortalities (≥90 %) compared to the 0.2 % d-allethrin reference coil. CYP4H42 and CYP4H43 expressions were significantly higher in 0.3 % d-allethrin treated mosquitoes compared to the other treated populations. Short-term (KT50) exposure to mosquito coils induced significantly higher expression of both genes in 0.005 % metofluthrin exposed mosquitoes. These results suggest the evaluated products provided better protection than the reference coil; however, they also induced the expression of metabolic genes which could impact negatively on personal protection against mosquito.


Similar content being viewed by others
Abbreviations
- CYP:
-
Cytochrome P450
- CYP4:
-
Cytochrome P450 family 4
- KT:
-
Knockdown time
- KT50 :
-
50 % Knockdown time
- KT90 :
-
90 % Knockdown time
References
World Health Organization (2014) Dengue and severe dengue, fact sheet no. 117. Updated March 2014. http://www.who.int/mediacentre/factsheets/fs117/en/. Accessed 3 Apr 2014
World Health Organization (2009) Dengue: guidelines for diagnosis, treatment, prevention and control—new edition. http://www.who.int/tdr/publications/documents/dengue-diagnosis.pdf. Accessed 1 Feb 2014
Beatty ME, Letson GW, Margolis HS (2009) Estimating the global burden of dengue. Am J Trop Med Hyg 81(Suppl. 1):231
Bhatt S, Gething PW, Brady OJ, Messina JP, Farlow AW, Moyes CL, Drake JM, Brownstein JS, Hoen AG, Sankoh O, Myers MF, George DB, Jaenisch T, Wint GRW, Simmons CP, Scott TW, Farrar JJ, Hay SI (2013) The global distribution and burden of dengue. Nature 496:504–507
Sabchareon A, Wallace D, Sirivichayakul C, Limkittikul K, Chanthavanich P, Suvannadabba S, Jiwariyavej V, Dulyachai W, Pengsaa K, Wartel TA, Moureau A, Saville M, Bouckenooghe A, Viviani S, Tornieporth NG, Lang J (2012) Protective efficacy of the recombinant, live-attenuated, CYD tetravalent dengue vaccine in Thai schoolchildren: a randomised, controlled phase 2b trial. Lancet 380:1559–1567
Halstead SB (2012) Dengue vaccine development: a 75% solution? Lancet 380:1535–1536
World Health Organization (2012) Global strategy for dengue prevention and control 2012–2020. http://reliefweb.int/sites/reliefweb.int/files/resources/9789241504034_eng.pdf. Accessed 1 Feb 2014
World Health Organization (2009) Guidelines for efficacy testing of household insecticide products. Mosquito coils, vaporizer mats, liquid vaporizers, ambient emanators and aerosols. http://whqlibdoc.who.int/hq/2009/WHO_HTM_NTD_WHOPES_2009.3_eng.pdf. Accessed 7 Sep 2013
Davies TGE, Field LM, Usherwood PNR, Williamson MS (2007) DDT, pyrethrins, pyrethroids and insect sodium channels. IUBMB Life 59:151–162
Khambay BPS, Jewess PJ (2010) In: Gilbert LI, Gill SS (eds) Insect control: biological and synthetic agents. Elsevier, Oxford
Ranson H, Burhani J, Lumjuan N, Black WC IV (2010) Insecticide resistance in dengue vectors. TropIKA.net 1. http://journal.tropika.net/scielo.php?script=sci_arttext&pid=S2078-86062010000100003&lng=en. Accessed 6 Apr 2014
Temu EA, Maxwell C, Munyekenye G, Howard AFV, Munga S, Avicor SW, Poupardin R, Jones JJ, Allan R, Kleinschmidt I, Ranson H (2012) Pyrethroid resistance in Anopheles gambiae, in Bomi County, Liberia, compromises malaria vector control. PLoS One 7:e44986
Vontas J, Kioulos E, Pavlidi N, Morou E, della Torre A, Ranson H (2012) Insecticide resistance in the major dengue vectors Aedes albopictus and Aedes aegypti. Pestic Biochem Physiol 104:126–131
Scott JG, Wen Z (2001) Cytochromes P450 of insects: the tip of the iceberg. Pest Manag Sci 57:958–967
Li X, Schuler MA, Berenbaum MR (2007) Molecular mechanisms of metabolic resistance to synthetic and natural xenobiotics. Annu Rev Entomol 52:231–253
Feyereisen R (2005) In: Gilbert LI, Iatrou K, Gill SS (eds) Comprehensive molecular insect science, vol 4. Elsevier, Oxford
Feyereisen R (1999) Insect P450 enzymes. Annu Rev Entomol 44:507–533
Karatolos N, Williamson MS, Denholm I, Gorman K, Ffrench-Constant RH, Bass C (2012) Over-expression of a cytochrome P450 is associated with resistance to pyriproxyfen in the greenhouse whitefly Trialeurodes vaporariorum. PLoS One 7:e31077
Zhu F, Gujar H, Gordon JR, Haynes KF, Potter MF, Palli SR (2013) Bed bugs evolved unique adaptive strategy to resist pyrethroid insecticides. Sci Rep 3:1456
David JP, Boyer S, Mesneau A, Ball A, Ranson H, Dauphin-Villemant C (2006) Involvement of cytochrome P450 monooxygenases in the response of mosquito larvae to dietary plant xenobiotics. Insect Biochem Mol Biol 36:410–420
Schuler MA (2011) P450s in plant–insect interactions. Biochim Biophys Acta 1814:36–45
Riaz MA, Poupardin R, Reynaud S, Strode C, Ranson H, David JP (2009) Impact of glyphosate and benzo[a]pyrene on the tolerance of mosquito larvae to chemical insecticides. Role of detoxification genes in response to xenobiotics. Aquat Toxicol 93:61–69
Karunker I, Benting J, Lueke B, Ponge T, Nauen R, Roditakis E, Vontas J, Gorman K, Denholm I, Morin S (2008) Over-expression of cytochrome P450 CYP6CM1 is associated with high resistance to imidacloprid in the B and Q biotypes of Bemisia tabaci (Hemiptera: Aleyrodidae). Insect Biochem Mol Biol 38:634–644
Petersen RA, Zangerl AR, Berenbaum MR, Schuler MA (2001) Expression of CYP6B1 and CYP6B3 cytochrome P450 monooxygenases and furanocoumarin metabolism in different tissues of Papilio polyxenes (Lepidoptera: Papilionidae). Insect Biochem Mol Biol 31:679–690
Wen Z, Pan L, Berenbaum MR, Schuler MA (2003) Metabolism of linear and angular furanocoumarins by Papilio polyxenes CYP6B1 co-expressed with NADPH cytochrome P450 reductase. Insect Biochem Mol Biol 33:937–947
Giraudo M, Unnithan GC, Le Goff G, Feyereisen R (2010) Regulation of cytochrome P450 expression in Drosophila: genomic insights. Pestic Biochem Physiol 97:115–122
Willoughby L, Chung H, Lumb C, Robin C, Batterham P, Daborn PJ (2006) A comparison of Drosophila melanogaster detoxification gene induction responses for six insecticides, caffeine and phenobarbital. Insect Biochem Mol Biol 36:934–942
Kalajdzic P, Markaki M, Oehler S, Savakis C (2013) Imidacloprid does not induce Cyp genes involved in insecticide resistance of a mutant Drosophila melanogaster line. Food Chem Toxicol 60:355–359
Global Invasive Species Database (2014) Aedes albopictus. http://www.issg.org/database/species/ecology.asp?si=109&fr=1&sts=&lang=EN. Accessed 14 Mar 2014
Mulla MS, Thavara U, Tawatsin A, Kong-Ngamsuk W, Chompoosri J (2001) Mosquito burden and impact on the poor: measures and costs for personal protection in some communities in Thailand. J Am Mosq Control Assoc 17:153–159
Lawrance CE, Croft AM (2004) Do mosquito coils prevent malaria? A systematic review of trials. J Travel Med 11:92–96
Xue RD, Qualls WA, Phillips JD, Zhao TY (2012) Insecticidal activity of five commercial mosquito coils against Anopheles albimanus, Aedes albopictus, and Culex quinquefasciatus. J Am Mosq Control Assoc 28:131–133
Avicor SW, Owusu EO, Wajidi MFF (2013) D-allethrin based mosquito coils for mosquito control: knockdown and mortality effects on the malaria vector Anopheles gambiae sensu lato. Int J Agric Biol 15:1035–1038
Deguchi Y, Yamada T, Hirose Y, Nagahori H, Kushida M, Sumida K, Sukata T, Tomigahara Y, Nishioka K, Uwagawa S, Kawamura S, Okuno Y (2009) Mode of action analysis for the synthetic pyrethroid metofluthrin-induced rat liver tumors: evidence for hepatic CYP2B induction and hepatocyte proliferation. Toxicol Sci 108:69–80
Vences-Mejía A, Gómez-Garduño J, Caballero-Ortega H, Dorado-González V, Nosti-Palacios R, Labra-Ruíz N, Espinosa-Aguirre JJ (2012) Effect of mosquito mats (pyrethroid-based) vapor inhalation on rat brain cytochrome P450s. Toxicol Mech Methods 22:41–46
Andersen CL, Jensen JL, Ørntoft TF (2004) Normalization of real-time quantitative reverse transcription-PCR data: a model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res 64:5245–5250
Pfaffl MW (2001) A new mathematical model for relative quantification in real-time RT-PCR. Nucleic Acids Res 29:e45
Abbott WS (1987) A method of computing the effectiveness of an insecticide. 1925. J Am Mosq Control Assoc 3:302–303
Lozano-Fuentes S, Saavedra-Rodriguez K, Black WC IV, Eisen L (2012) QCal: a software application for the calculation of dose-response curves in insecticide resistance bioassays. J Am Mosq Control Assoc 28:59–61
Payne RW, Murray DA, Harding SA, Baird DB, Soutar DM (2006) An introduction to GenStat for Windows, 9th edn. VSN International, Hemel Hempstead
Debboun M, Strickman D (2013) Insect repellents and associated personal protection for a reduction in human disease. Med Vet Entomol 27:1–9
Becker N, Petrić D, Zgomba M, Boase C, Madon M, Dahl C, Kaiser A (2010) Mosquitoes and their control. Springer, Heidelberg
Surendran SN, Kajatheepan A (2007) Perception and personal protective measures toward mosquito bites by communities in Jaffna District, northern Sri Lanka. J Am Mosq Control Assoc 23:182–186
Lukwa N, Chiwade T (2008) Lack of insecticidal effect of mosquito coils containing either metofluthrin or esbiothrin on Anopheles gambiae sensu lato mosquitoes. Trop Biomed 25:191–195
Ujihara K, Mori T, Iwasaki T, Sugano M, Shono Y, Matsuo N (2004) Metofluthrin: a potent new synthetic pyrethroid with high vapor activity against mosquitoes. Biosci Biotechnol Biochem 68:170–174
Sugano M, Ishiwatari T (2012) The biological activity of a novel pyrethroid: metofluthrin. Top Curr Chem 314:203–220
Poupardin R, Riaz MA, Vontas J, David JP, Reynaud S (2010) Transcription profiling of eleven cytochrome P450s potentially involved in xenobiotic metabolism in the mosquito Aedes aegypti. Insect Mol Biol 19:185–193
Kalajdzic P, Oehler S, Reczko M, Pavlidi N, Vontas J, Hatzigeorgiou AG, Savakis C (2012) Use of mutagenesis, genetic mapping and next generation transcriptomics to investigate insecticide resistance mechanisms. PLoS One 7:e40296
Kim CH, Muturi EJ (2012) Relationship between leaf litter identity, expression of cytochrome P450 genes and life history traits of Aedes aegypti and Aedes albopictus. Acta Trop 122:94–100
Liu N, Li T, Reid WR, Yang T, Zhang L (2011) Multiple cytochrome P450 genes: their constitutive overexpression and permethrin induction in insecticide resistant mosquitoes, Culex quinquefasciatus. PLoS One 6:e23403
Acknowledgments
The authors are grateful for the supports of The World Academy of Sciences (TWAS) and the Universiti Sains Malaysia (USM) for a TWAS-USM postgraduate fellowship, USM Research University grant [Grant #1001/PJJAUH/815095] and the Malaysian Ministry of Education [ERGS Grant #203/PPJAUH/6730097]. The logistical support of staff of the Vector Control Research Unit, Universiti Sains Malaysia is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Avicor, S.W., Wajidi, M.F.F., El-garj, F.M.A. et al. Insecticidal Activity and Expression of Cytochrome P450 Family 4 Genes in Aedes albopictus After Exposure to Pyrethroid Mosquito Coils. Protein J 33, 457–464 (2014). https://doi.org/10.1007/s10930-014-9580-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-014-9580-z