Abstract
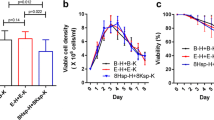
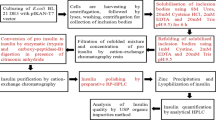
The first report of complete nucleotide sequences for α- and β-globin chains from the Siamese hemoglobin (Crocodylus siamensis) is given in this study. The cDNAs encoding α- and β-globins were cloned by RT-PCR using the degenerate primers and by the rapid amplification of cDNA ends method. The full-length α-globin cDNA contains an open reading frame of 423 nucleotides encoding 141 amino acid residues, whereas the β-globin cDNA contains an open reading frame of 438 nucleotides encoding 146 amino acid residues. The authenticity of both α- and β-globin cDNA clones were also confirmed by the heterologous expression in Escherichia coli (E. coli). This is the first time that the recombinant C. siamensis globins were produced in prokaryotic system. Additionally, the heme group was inserted into the recombinant proteins and purified heme-bound proteins were performed by affinity chromatography using Co2+-charged Talon resins. The heme-bound proteins appeared to have a maximum absorbance at 415 nm, indicated that the recombinant proteins bound to oxygen and formed active oxyhemoglobin (HbO2). The results indicated that recombinant C. siamensis globins were successfully expressed in prokaryotic system and possessed an activity as ligand binding protein.






Similar content being viewed by others
Abbreviations
- GSP:
-
Gene specific primers
- Hb:
-
Hemoglobin
- IPTG:
-
Isopropyl-β-D-thiogalactopyranoside
- RACE-PCR:
-
Rapid amplification of cDNA ends
References
Alam SL, Dutton DP, Satterlee JD (1994) Biochem 33:10337–10344
Altschul S, Madden T, Schaffer A, Zhang J, Zhang Z, Miller W, Lipman D (1997) Nucleic Acids Res 25:3389–3402
Bonini-Domingos CR, Silva MB, Romero RM, Zamaro PJA, Ondei LS, Zago CES, Moreira SB, Salgado CG (2007) Genet Mol Res 6:415–421
Bordin S, Meza AN, Saad STO, Ogo SH, Costa FF (1997) Biochem Mol Biol Int 42:255–260
Brunori M, Cutruzzola F, Vallone B (1995) Curr Biol 5:462–465
Bunn HF, Forget BG (1986) Animal Hemoglobin. In: John D (ed) Hemoglobin: molecular, genetic and clinical aspects in reptile. W. B. Saunders Company, Philadelphia, pp 151–152
Dafre AL, Brandao TAS, Reischl E (2007) J Zoology 85:404–412(409)
Deng L, Pan X, Wang Y, Wang L, Zhou XE, Li M, Feng Y, Wu Q, Wang B, Huang N (2009) Hum Reprod 24:211–218
Falk JE (1964) Dynamic coordination chemistry of metalloporphyrins. In: Peter H (ed) Porphyrins and Metalloporphyrins. Elsevier, Amsterdam, p 240
Hoffman B, Key B, Ofer B, Kiryat T (2002) In (United States) US 6,340,667
Jandaruang J, Siritapetawee J, Thumanu K, Songsiriritthigul C, Krittanai C, Daduang S, Dhiravisit A, Thammasirirak S (2012) Protein J 31:43–50
Kleinschmidt T, Sgouros J (1987) Biol Chem Hoppe Seyler 368:579–615
Komiyama N, Nagai K (1999) In (United States) US 5,942,488
Komiyama NH, Miyazaki G, Tame J, Nagai K (1995) Nature 373:244–246
Liepke C, Baxmann S, Heine C, Breithaupt N, Standker L, Forssmann WG (2003) J Chromatogr B 791:345–356
Melo MB, Bordin S, Duarte ASS, Ogo SH, Torsoni MA, Saad STO, Costa FF (2003) Comp Biochem Physiol Part B 134:389–395
Merchant M, Pallansch M, Paulman R, Wells J, Nalca A, Ptak R (2005) Antiviral Res 66:35–38
Merchant M, Roche C, Elsey R, Prudhomme J (2003) Comp Biochem Physiol Part B 136:505–513
Merchant M, Thibodeaux D, Loubser K, Elsey R (2004) J Parasitol 90:1480–1483
Mook CC (1921) Bull Am Mus Nat Hist XLIV:67–100
Nedjar-Arroume N, Dubois-Delval V, Adje EY, Traisnel J, Krier F, Mary P, Kouach M, Briand G, Guillochon D (2008) Peptides 29:969–977
Oinuma KI, Hashimoto Y, Konishi K, Goda M, Noguchi T, Higashibata H, Kobayashi K (2003) J Biol Chem 278:29600–29608
Pata S, Yaraksa N, Daduang S, Temsiripong Y, Svasti J, Araki T, Thammasirirak S (2011) Dev Comp Immunol 35:545–553
Preecharram S, Daduang S, Bunyatratchata W, Araki T, Thammasirirak S (2008) Afr Biotechnol 7:3121–3128
Preecharram S, Jearranaiprepame P, Daduang S, Temsiripong Y, Somdee T, Fukamizo T, Svasti J, Araki T, Thammasirirak S (2010) Anim Sci J 81:393–401
Rajesh R, Pattabhi V (2003–2004) ICA New Lett
Shaharbanay M, Gollop N, Ravin S, Golomb E, Demarco L, Ferriera PC, Boson WL, Friedman E (1999) J Antimicrob Chemother 44:416–418
Shishikura F (2002) Zool Sci 19:197–206
Siegel LM, Murphy MJ, Kamin H (1973) J Biol Chem 248:251–264
Siroski PA, Pina CI, Larriera A, Merchant ME, Conza JD (2009) Zool Stud 48:238–242
Srihongthong S, Pakdeesuwan A, Daduang S, Araki T, Dhiravisit A, Thammasirirak S (2012) Protein J 31:466–476
Sun Q, Luo Y, Shen H, Li X, Yao L (2012) Food Sci Technol 47:148–154
Tamura K, Dudley J, Nei M, Kumar S (2007) Mol Biol Evol 24:1596–1599
Acknowledgments
This research was supported by the Royal Golden Jubilee (RGJ) Ph.D Program of Thailand Research Fund and National Research University Project of Thailand, Office of the Higher Education Commission through financial support. We would also like to thank the Protein and Proteomics Research Group at the Department of Biochemistry, Faculty of Science, Khon Kaen University, Mahidol University, Thailand and Department of Bioscience, School of Agriculture, Tokai University, Japan for their supports. We wish to acknowledge the support of the Khon Kaen University Publication Clinic, Research and Technology Transfer Affairs, Khon Kaen University, for their assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anwised, P., Kabbua, T., Temsiripong, T. et al. Molecular Cloning and Expression of α-Globin and β-Globin Genes from Crocodile (Crocodylus siamensis). Protein J 32, 172–182 (2013). https://doi.org/10.1007/s10930-013-9474-5
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-013-9474-5