Abstract
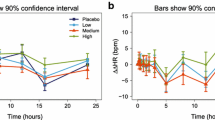
In spite of the evidence regarding high variability in the response to evoked pain, little attention has been paid to its impact on the screening of drugs for inflammatory and neuropathic pain. In this study, we explore the feasibility of introducing optimality concepts to experimental protocols, enabling estimation of parameter and model uncertainty. Pharmacokinetic (PK) and pharmacodynamic data from different experiments in rats were pooled and modelled using nonlinear mixed effects modelling. Pain data on gabapentin and placebo-treated animals were generated in the complete Freund’s adjuvant model of neuropathic pain. A logistic regression model was applied to optimise sampling times and dose levels to be used in an experimental protocol. Drug potency (EC50) and interindividual variability (IIV) were considered the parameters of interest. Different experimental designs were tested and validated by SSE (stochastic simulation and estimation) taking into account relevant exposure ranges. The pharmacokinetics of gabapentin was described by a two-compartment PK model with first order absorption (CL = 0.159 l h−1, V2 = 0.118 l, V3 = 0.253 l, Ka = 0.26 h−1, Q = 1.22 l h−1). Drug potency (EC50) for the anti-allodynic effects was estimated to be 1400 ng ml−1. Protocol optimisation improved bias and precision of the EC50 by 6 and 11.9. %, respectively, whilst IIV estimates showed improvement of 31.89 and 14.91 %, respectively. Our results show that variability in behavioural models of evoked pain response leads to uncertainty in drug potency estimates, with potential impact on the ranking of compounds during screening. As illustrated for gabapentin, ED-optimality concepts enable analysis of discrete data taking into account experimental constraints.





Similar content being viewed by others
References
Agresti A (1999) Modelling ordered categorical data: recent advances and future challenges. Stat Med 18:2191–2207
Backonja M, Woolf CJ (2010) Future directions in neuropathic pain therapy: closing the translational loop. Oncologist 15(Suppl. 2):24–29
Blackburn-Munro G (2004) Pain-like behaviours in animals—how human are they? Trends Pharmacol Sci 25:299–305
Bouillon-Pichault M, Jullien V, Bazzoli C, Pons G, Tod M (2011) Pharmacokinetic design optimization in children and estimation of maturation parameters: example of cytochrome P450 3A4. J Pharmacokinet Pharmacodyn 38:25–40
Danhof M, Lange EC, Ploeger BA, Voskuyl RA, Della Pasqua O (2008) Mechanism-based pharmacokinetic–pharmacodynamic (PK–PD) modeling in translational drug research. Trends Pharmacol Sci 29:186–191
D’Argenio DZ (1981) Optimal sampling times for pharmacokinetic experiments. J Pharmacokinet Biopharm 9:739–756
Dodds MG, Hooker AC, Vicini P (2005) Robust population pharmacokinetic experiment design. J Pharmacokinet Pharmacodyn 32:33–64
Gilron I, Coderre TJ (2007) Emerging drugs in neuropathic pain. Expert Opin Emerg Drugs 12:113–126
Hooker AC, Foracchia M, Dodds MG, Vicini P (2003) An evaluation of population D-optimal designs via pharmacokinetic simulations. Ann Biomed Eng 31:98–111
Huntjens DR, Danhof M, Della Pasqua O (2005) Pharmacokinetic-pharmacodynamic correlations and biomarkers in the development of COX-2 inhibitors. Rheumatology 44:846–859
Huntjens DR, Spalding DJ, Danhof M, Della Pasqua O (2009) Differences in the sensitivity of behavioural measures of pain to the selectivity of cyclo-oxygenase inhibitors. Eur J Pain 13:448–457
Joshi SK, Honore P, Hernandez G, Schmidt R, Gomtsyan A et al (2009) Additive antinociceptive effects of the selective Nav1.8 blocker A-803467 and selective TRPV1 antagonists in rat inflammatory and neuropathic pain models. J Pain 10:306–315
Le Bars D, Gozariu M, Cadden SW (2001) Animal models of nociception. Pharmacol Rev 53:597–652
Nestorov I, Graham G, Duffull S, Aarons L, Fuseau E et al (2001) Modeling and simulation for clinical trial design involving a categorical response: a phase II case study with naratriptan. Pharm Res 18:1210–1219
Nyberg J, Karlsson MO, Hooker AC (2009) Simultaneous optimal experimental design on dose and sample times. J Pharmacokinet Pharmacodyn 36:125–145
Ogungbenro K, Aarons L (2007) Design of population pharmacokinetic experiments using prior information. Xenobiotica 37:1311–1330
Ogungbenro K, Aarons L (2010) Optimal design of pharmacokinetic studies. Basic Clin Pharmacol Toxicol 106:250–255
Pang MH, Kim Y, Jung KW, Cho S, Lee DH (2012) A series of case studies: practical methodology for identifying antinociceptive multi-target drugs. Drug Discov Today 17:425–434
R Development Core Team (2010) R: a language and environment for statistical computing, ed. R Foundation for statistical computing, R Foundation for statistical computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org
Sheiner LB, Beal SL (1981) Some suggestions for measuring predictive performance. J Pharmacokinet Biopharm 9:503–512
Stewart BH, Kugler AR, Thompson PR, Bockbrader HN (1993) A saturable transport mechanism in the intestinal absorption of gabapentin is the underlying cause of the lack of proportionality between increasing dose and drug levels in plasma. Pharm Res 10:276–281
Taneja A, Di Iorio VL, Danhof M, Della Pasqua O (2012) Translation of drug effects from experimental models of neuropathic pain and analgesia to humans. Drug Discov Today 17:837–849
Tod M, Rocchisani JM (1997) Comparison of ED, EID, and API criteria for the robust optimization of sampling times in pharmacokinetics. J Pharmacokinet Biopharm 25:515–537
Walker K, Fox AJ, Urban LA (1999) Animal models for pain research. Mol Med Today 5:319–321
Schoemaker RC, van Gerven JM, Cohen AF (1998) Estimating potency for the Emax model without attaiing maximal effects. J Pharmacokinet Biopharm 26:581–593
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was conducted on behalf of the members of the Pain Project of the TI Pharma mechanism-based PKPD modelling platform. The members are Benson N, Marshall S (Modelling & Simulation, Pfizer, Sandwich, UK); Machin I (Pain Research Unit, Sandwich, UK); DeAlwis D (Global PK/PD/TS Europe, Eli Lilly, Erl Wood, UK).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Taneja, A., Nyberg, J., de Lange, E.C.M. et al. Application of ED-optimality to screening experiments for analgesic compounds in an experimental model of neuropathic pain. J Pharmacokinet Pharmacodyn 39, 673–681 (2012). https://doi.org/10.1007/s10928-012-9278-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10928-012-9278-9