Abstract
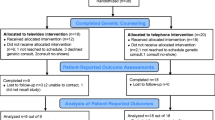
Telegenetics—genetic counseling via live videoconferencing—can improve access to cancer genetic counseling (CGC) in underserved areas, but studies on cancer telegenetics have not applied randomized methodology or assessed cost. We report cost, patient satisfaction and CGC attendance from a randomized trial comparing telegenetics with in-person CGC among individuals referred to CGC in four rural oncology clinics. Participants (n = 162) were randomized to receive CGC at their local oncology clinic in-person or via telegenetics. Cost analyses included telegenetics system; mileage; and personnel costs for genetic counselor, IT specialist, and clinic personnel. CGC attendance was tracked via study database. Patient satisfaction was assessed 1 week post-CGC via telephone survey using validated scales. Total costs were $106 per telegenetics patient and $244 per in-person patient. Patient satisfaction did not differ by group on either satisfaction scale. In-person patients were significantly more likely to attend CGC than telegenetics patients (89 vs. 79 %, p = 0.03), with bivariate analyses showing an association between lesser computer comfort and lower attendance rate (Chi-square = 5.49, p = 0.02). Our randomized trial of telegenetics vs. in-person counseling found that telegenetics cost less than in-person counseling, with high satisfaction among those who attended. This study provides support for future randomized trials comparing multiple service delivery models on longer-term psychosocial and behavioral outcomes.

Similar content being viewed by others
References
American College of Surgeons (2012). Cancer program standards 2012: Ensuring patient-centered care. Retrieved from http://www.facs.org.
American Society of Clinical Oncology. (2003). American Society of Clinical Oncology policy statement update: genetic testing for cancer susceptibility. Journal of Clinical Oncology, 21(12), 2397–2406.
Berry, D. A., Iversen, E. S., Jr., Gudbjartsson, D. F., Hiller, E. H., Garber, J. E., Peshkin, B. N., Lerman, C., Watson, P., Lynch, H. T., Hilsenbeck, S. G., Rubinstein, W. S., Hughes, K. S., & Parmigiani, G. (2002). BRCAPRO validation, sensitivity of genetic testing of BRCA1/BRCA2, and prevalence of other breast cancer susceptibility genes. Journal of Clinical Oncology, 20(11), 2701–2712.
Bish, A., Sutton, S., Jacobs, C., Levene, S., Ramirez, A., & Hodgson, S. (2002). Changes in psychological distress after cancer genetic counseling: a comparison of affected and unaffected women. British Journal of Cancer, 86, 43–50.
Braithwaite, D., Emery, J., Walter, F., Prevost, A. T., & Sutton, S. (2006). Psychological impact of genetic counseling for familial cancer: a systematic review and meta-analysis. Familial Cancer, 5(1), 61–75.
Buchanan, A. H., Skinner, C. E., Calingaert, B., Schildkraut, J. M., King, R. H., & Marcom, P. K. (2009). Cancer genetic counseling in rural North Carolina oncology clinics: program establishment and patient characteristics. Community Oncology, 6, 70–77.
Bunz, U. (2004). The Computer-Email-Web (CEW) fluency scale-development and validation. International Journal of Human Computer Interaction, 17(4), 479–506.
Chen, S., Wang, W., Lee, S., Nafa, K., Lee, J., Romans, K., Watson, P., Gruber, S. B., Euhus, D., Kinzler, K. W., Jass, J., Gallinger, S., Lindor, N. M., Casey, G., Ellis, N., Giardiello, F. M., Offit, K., Parmigiani, G., & Colon Cancer Family, R. (2006). Prediction of germline mutations and cancer risk in the Lynch syndrome. JAMA, 296(12), 1479–1487.
Coelho, J. J., Arnold, A., Nayler, J., Tischkowitz, M., & MacKay, J. (2005). An assessment of the efficacy of cancer genetic counselling using real-time videoconferencing technology (telemedicine) compared to face-to-face consultations. European Journal of Cancer, 41(15), 2257–2261.
d’Agincourt-Canning, L., McGillivray, B., Panabaker, K., Scott, J., Pearn, A., Ridge, Y., & Portigal-Todd, C. (2008). Evaluation of genetic counseling for hereditary cancer by videoconference in British Columbia. British Columbia Medical Journal, 50(10), 554–559.
Daly, M., Axilbund, J., Bryant, E., Buys, S., Esserman, L., Farrell, C., et al. (2008). NCCN practice guidelines in oncology: Genetic/familial high-risk assessment: breast and ovarian. Version 1.2008. Retrieved Feb 11, 2011, from http://www.nccn.org/professionals/physician_gls/pdf/genetics_screening.pdf. Accessed 11 Feb 2011.
Datta, S. K., Buchanan, A. H., Hollowell, G. P., Bereford, H. F., Marcom, P. K., & Adams, M. B. (2011). Telemedicine vs in-person cancer genetic counseling: measuring satisfaction and conducting economic analysis. Journal of Comparative Effectiveness Research, 1, 43–50.
Demarco, T., Peshkin, B., Mars, B., & Tercyak, K. (2004). Patient satisfaction with cancer genetic counseling: a pyschometric analysis of the genetic counseling satisfaction scale. Journal of Genetic Counseling, 13(4), 293–304.
Drummond, M. F., O'Brien, B. J., Stoddart, G. L., & Torrance, G. W. (1999). Chapter 4: Cost analysis. In Methods for the economic evaluation of health care programmes (2nd ed.). Oxford: Oxford University Press.
Erblich, J., Brown, K., Kim, Y., Valdimarsdottir, H. B., Livingston, B. E., & Bovbjerg, D. H. (2005). Development and validation of a Breast Cancer Genetic Counseling Knowledge Questionnaire. Patient Education and Counseling, 56(2), 182–191.
Gray, J., Brain, K., Iredale, R., Alderman, J., France, E., & Hughes, H. (2000). A pilot study of telegenetics. Journal of Telemedicine and Telecare, 6(4), 245–247.
Hadley, D. W., Jenkins, J. F., Dimond, E., de Carvalho, M., Kirsch, I., & Palmer, C. G. (2004). Colon cancer screening practices after genetic counseling and testing for hereditary nonpolyposis colorectal cancer. Journal of Clinical Oncology, 22(1), 39–44.
Hammers, E. (2009). Innovations in service delivery in the age of genomics: Workshop summary. Washington, D. C.: National Academy of Sciences.
Hampel, H., Sweet, K., Westman, J. A., Offit, K., & Eng, C. (2004). Referral for cancer genetics consultation: a review and compilation of risk assessment criteria. Journal of Medical Genetics, 41(2), 81–91.
Hilgart, J. S., Hayward, J. A., Coles, B., & Iredale, R. (2012). Telegenetics: a systematic review of telemedicine in genetics services. Genetics in Medicine, 14(9), 765–776.
Hughes, C., Gomez-Caminero, A., Benkendorf, J., Kerner, J., Isaacs, C., Barter, J., & Lerman, C. (1997). Ethnic differences in knowledge and attitudes about BRCA1 testing in women at increased risk. Patient Education and Counseling, 32(1–2), 51–62.
Hughes, C., Lerman, C., Schwartz, M., Peshkin, B., Wenzel, L., Narod, S., Corio, C., Tercyak, K. P., Hanna, D., Main, D., & Isaacs, C. (2002). All in the family: Evaluation of the processand content of sisters’ communication about BRCA1 and BRCA2 genetic test results. American Journal of Medical Genetics, 107, 143–150.
Jarvis-Selinger, S., Chan, E., Payne, R., Plohman, K., & Ho, K. (2008). Clinical telehealth across the disciplines: lessons learned. Telemedicine Journal and e-Health, 14(7), 720–725.
Lea, D. H., Johnson, J. L., Ellingwood, S., Allan, W., Patel, A., & Smith, R. (2005). Telegenetics in Maine: Successful clinical and educational service delivery model developed from a 3-year pilot project. Genetics in Medicine, 7(1), 21–27.
Levine, T., & Donitsa-Schmidt, S. (1998). Computer use, confidence, attitudes, and knowledge: a causal analysis. Computers in Human Behavior, 14(1), 125–146.
Olopade, O. I., Fackenthal, J. D., Dunston, G., Tainsky, M. A., Collins, F., & Whitfield-Broome, C. (2003). Breast cancer genetics in African Americans. Cancer, 97(1 Suppl), 236–245.
Schwartz, M. D., Lerman, C., Brogan, B., Peshkin, B. N., Halbert, C. H., DeMarco, T., et al. (2004). Impact of BRCA1/BRCA2 counseling and testing on newly diagnosed breast cancer patients. Journal of Clinical Oncology, 22(10), 1823–1829.
Schwartz, M. D., Valdimarsdottir, H. B., Peshkin, B. N., Mandelblatt, J., Nusbaum, R., Huang, A. T., Chang, Y., Graves, K., Isaacs, C., Wood, M., McKinnon, W., Garber, J., McCormick, S., Kinney, A. Y., Luta, G., Kelleher, S., Leventhal, K.-G., Vegella, P., Tong, A., & King, L. (2014). Randomized noninferiority trial of telephone versus in-person genetic counseling for hereditary breast and ovarian cancer. Journal of Clinical Oncology, 32(7), 618–626.
Stacey, D., DeGrasse, C., & Johnston, L. (2002). Addressing the support needs of women at high risk for breast cancer: evidence-based care by advanced practice nurses. Oncology Nursing Forum, 29(6), E77–E84.
Trepanier, A., Ahrens, M., McKinnon, W., Peters, J., Stopfer, J., Grumet, S. C., Manley, S., Culver, J. O., Acton, R., Larsen-Haidle, J., Correia, L. A., Bennett, R., Pettersen, B., Ferlita, T. D., Costalas, J. W., Hunt, K., Donlon, S., Skrzynia, C., Farrell, C., Callif-Daley, F., Vockley, C. W., & National Society of Genetic, C. (2004). Genetic cancer risk assessment and counseling: recommendations of the National Society of Genetic Counselors. Journal of Genetic Counseling, 13(2), 83–114.
U. S. Preventive Services Task Force. (2005). Genetic risk assessment and BRCA mutation testing for breast and ovarian cancer susceptibility: recommendation statement. Annals of Internal Medicine, 143(5), 355–361.
U.S. Census Bureau (2010). North Carolina QuickFacts. 2010 Census of Population and Housing, Demographic Profile,. Retrieved May 10, 2013, from http://www.quickfacts.census.gov/qfd/states/37000.html.
U.S. Department of Health & Human Services (2006). Coverage and reimbursement of genetic tests and services: Report of the Secretary’s Advisory Committee on Genetics, Health, and Society. http://oba.od.nih.gov/oba/sacghs/reports/CR_report.pdf.
Ware, J. E., Jr., & Hays, R. D. (1988). Methods for measuring patient satisfaction with specific medical encounters. Medical Care, 26(4), 393–402.
Watson, M., Kash, K. M., Homewood, J., Ebbs, S., Murday, V., & Eeles, R. (2005). Does genetic counseling have any impact on management of breast cancer risk? Genetic Testing, 9(2), 167–174.
Weitzel, J. N., McCaffrey, S. M., Nedelcu, R., MacDonald, D. J., Blazer, K. R., & Cullinane, C. A. (2003). Effect of genetic cancer risk assessment on surgical decisions at breast cancer diagnosis. Archives of Surgery, 138(12), 1323–1328. discussion 1329.
Wham, D., Vu, T., Chan-Smutko, G., Kobelka, C., Urbauer, D., & Heald, B. (2010). Assessment of clinical practices among cancer genetic counselors. Familial Cancer, 9(3), 459–468.
Zilliacus, E., Meiser, B., Lobb, E., Dudding, T. E., Barlow-Stewart, K., & Tucker, K. (2010a). The virtual consultation: practitioners’ experiences of genetic counseling by videoconferencing in Australia. Telemedicine Journal and e-Health, 16(3), 350–357.
Zilliacus, E. M., Meiser, B., Lobb, E. A., Kirk, J., Warwick, L., & Tucker, K. (2010b). Women’s experience of telehealth cancer genetic counseling. Journal of Genetic Counseling, 19(5), 463–472.
Zilliacus, E. M., Meiser, B., Lobb, E. A., Kelly, P. J., Barlow-Stewart, K., Kirk, J. A., Spigelman, A. D., Warwick, L. J., & Tucker, K. M. (2011). Are videoconferenced consultations as effective as face-to-face consultations for hereditary breast and ovarian cancer genetic counseling? Genetics in Medicine, 13(11), 933–941.
Acknowledgments
The authors wish to thank the Duke Cancer Network and participating clinics for their support of this study, and of genetic counseling in underserved communities.
Funding
The study described here was supported by grant DISP0707781 from Susan G. Komen for the Cure, which had no role in the design of the study described here or in the development or approval of this manuscript. Portions of the salaries of Mr. Buchanan, Drs. Datta, Hollowell, and Adams, and Mr. Beresford, Mr. Freeland, Mr. Rogers, and Mr. Boling were funded by this grant. Drs. Skinner and Marcom did not receive salary or other support from this grant.
This manuscript was made possible by Grant Number 1 UL1 RR024128-01 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research. Its contents are solely the responsibility of the authors and do not necessarily represent the official view of NCRR or NIH.
Conflict of Interest
Mr. Buchanan’s work has been funded by the NIH. Dr. Datta’s work has been funded by the Department of Defense and VA Health Services Research & Development. Dr. Skinner’s work has been funded by the NIH and the Cancer Prevention Research Institute of Texas. Dr. Hollowell’s work has been funded by the Howard Hughes Medical Institute. Mr. Rogers’ work has been funded by the Alpha-1 Foundation. Dr. Marcom’s work has been funded by the NIH and Department of Defense. Dr. Adams has served as a consultant to Sanofi, Drexel University, CustomID and MED-IQ. The authors declare that these relationships are not directly or indirectly related to the research described in this manuscript.
Human Studies and Informed Consent
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study.
Animal Studies
This article does not contain any studies with animals performed by any of the authors.
Author information
Authors and Affiliations
Corresponding author
Additional information
ClinicalTrials.gov Identifier: NCT00609505
Rights and permissions
About this article
Cite this article
Buchanan, A.H., Datta, S.K., Skinner, C.S. et al. Randomized Trial of Telegenetics vs. In-Person Cancer Genetic Counseling: Cost, Patient Satisfaction and Attendance. J Genet Counsel 24, 961–970 (2015). https://doi.org/10.1007/s10897-015-9836-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10897-015-9836-6