Abstract
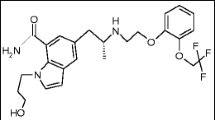
A highly sensitive and simple spectrofluorimetric method was developed for the determination of Amlodipine besylate (AML) in its pharmaceutical formulations and spiked human plasma. The proposed method is based on the investigation of the fluorescence spectral behaviour of AML in Tween-80 micellar system. In aqueous solution, the fluorescence intensity of AML was greatly enhanced (160 %) in the presence of Tween-80. The fluorescence intensity was measured at 427 nm after excitation at 385 nm. The fluorescence–concentration plot was rectilinear over the concentration range 0.1–4.0 μg/ml, with lower detection limit of 0.03 μg/ml. The suggested method was successfully applied for the analysis of AML in its commercial tablets alone or in combination with either Atorvastatin or Valsartan. The application of the proposed method was extended to the assay of AML in spiked human plasma and stability studies of AML after exposure to different forced degradation conditions, such as acidic, alkaline, photo- and oxidative conditions, according to ICH guidelines. The results were statistically compared to those obtained by comparison methods and were found to be in good agreement.






Similar content being viewed by others
References
Sweetman SC (2009) Martindale: the complete drug reference. Pharmaceutical Press, London
Taylor SH (1989) Am Heart J 118: 1123–1126.
Szymusiak-Mutnick B (1994) Comprehensive Pharmacy Review. In: Shargel L (ed) 2nd edition (Middle East edition), Mass Publishing Co. Giza, Egypt
The United States Pharmacopoeia 34 and NF 29 (2008) American Pharmaceutical Association, Washington, DC
The European Pharmacopoeia (2008) 6th edition. Council of Europe, Strasbourg, Vol. I and II
British Pharmacopoeia (2013) Her Majesty’s Stationary Office, London, UK
Ali A, Pasha K, Raju SA, Ahmed A (2010) Int J Pharmacy Pharm Sci 2:128–129
Askal HF, Abdelmageed OH, Sayed SM (2010) Abo El Hamed M. J Bull Pharm Sci Assiut Uni 33:201–215
Mali SL, Dhabale PN, Gonjari ID, Deshmukh V, Chanekar PD (2010) Int J Pharmacy Pharm Sci 2:71–74
Ramesh D, Ramakrishna S (2010) Int. J Pharmacy Pharm Sci 2:215–219
Darwish HW, Hassan SA, Salem MY, El-Zeiny BA (2011) Spectrochim Acta A Mol Biomol Spectrosc 83:140–148
Derayea SM, Askal HF, Abdel-Megeed OH, El Hamd MA (2012) Journal of Applied Pharmaceutical Science 2:84–89
Mahmoud AM, Abdel-Wadood HM, Mohamed NA (2012) Journal of Pharmaceutical Analysis 2:334–341
Darwish HW, Hassan SA, Salem MY, El-Zeany BA (2013) Spectrochim Acta A Mol Biomol Spectrosc 104:70–76
El Hamd MA, Derayea SM, Abdelmageed OH, Askal HF (2013) The. Am J Anal Chem 4:148–156
Farouk M, Elaziz OA, Tawakkol SM, Hemdan A, Shehata MA (2014) Spectrochim Acta A Mol Biomol Spectrosc 123:473–481
Abdel-Wadood HM, Mohamed NA, Mahmoud AM (2008) Spectrochim Acta A Mol Biomol Spectrosc 70:564–570
Shaalan RA, Belal TS (2010) Drug Test Anal 2:489–493
Moussa BA, El-Zaher AA, Mahrouse MA, Ahmed MS (2013) Anal Chem Insights 8:107–115
Patil KR, Rane VP, Sangshetti JN, Yeole RD, Shinde DB (2010) J Chromatogr Sci 48:601–606
El-Gizawy SM, Abdelmageed OH, Omar MA, Deryea SM, Abdel-Megied AM (2012) Am J Anal Chem 3:422–430
Kumar KK, Rao CK, Madhusudan G, Mukkanti K (2012) Am J Anal Chem 3:50–58
Patel DB, Mehta FA, Bhatt KK (2012) Sci Pharm 80:581–590
Tengli AR, Gurupadayya BM, Soni N (2013) Int J Chem Anal Sci 4:33–38
Argekar AP, Powar SG (2000) J Pharm Biomed Anal 21:1137–1142
Meyyanathan SN, Suresh B (2005) J Chromatogr Sci 43:73–75
Rajput Pankaj K, Navdeep K, Amanjot K, Vinod G, Ganti SSPS (2012) International Journal of Universal Pharmacy and Life Sciences 2:154–163
Scharpf F, Riedel KD, Laufen H, Leitold M (1994) J Chromatogr B Biomed Appl 655:225–233
Monkman SC, Ellis JS, Cholerton S, Thomason JM, Seymour RA, Idle JR (1996) J Chromatogr B Biomed Appl 678:360–364
Quaglia MG, Barbato F, Fanali S, Santucci E, Donati E, Carafa M, Marianecci C (2005) J Pharm Biomed Anal 37:73–79
Alnajjar AO (2011) J AOAC Int 94:498–502
Gazy AA (2004) Talanta 62:575–582
Kazemipour MM, Ansari M, Mohammadi A, Beitollahi H, Ahmadi R (2009) J Anal Chem 64:65–70
Matalka K, El-Thaher T, Saleem M, Arafat T, Jehanli A, Badwan A (2001) J Clin Lab Anal 15:47–53
Hinze WL, Singh HN, Baba Y, Harvey NG (1984) TrAC Trends Anal Chem 3:193–199
McLntire GL, Dorsey JG (1990) Crit Rev Anal Chem 21:257–278
Wang CC, Masi AN, Fernandez L (2008) Talanta 75:135–140
Pesez M, Bartos J (1974) Colorimetric and fluorimetric analysis of organic compounds and drugs. Marcel Dekker Inc., New York
Walash MI, Belal F, El-Enany N, Eid M, El-Shaheny RN (2011) Luminescence 26:670–679
Leung R, Shah DO (1986) J Colloid Interface Sci 113:484–499
Skoog DA, Holler FJ, Crouch SR (2007) Principles of instrumental analysis, 6th edn. Belmont, NV, Thomson
Skoog DA, West DM, Holler FJ, Crouch SR (2004) Fundamentals of analytical chemistry, 8th edn. Philadelphia, PA, Saunders College Publishing
ICH Harmonized tripartite guideline, validation of analytical procedures: text and methodology, Q2(R1), 2005: http://www.ich.org/LOB/media/MEDIA417.pdf (accessed 15 February 2008).
Miller J, Miller J (2005) Statistics and chemometrics for analytical chemistry,5th edn. Harlow, UK, Pearson Education
Abdine H, Belal F, Gadkariem EA, Zoman N (2001) Journal of Liquid Chromatography & Related Technologies 24:3213–3225
Walash MI, Belal F, El-Enany N, Abdelal A (2007) Journal of Liquid Chromatography & Related Technologies 30:1015–1034
Mascoli V, Kuruganti U, Bapuji AT, Wang R, Damle B (2013) J Bioequiv Availab 5:76–79
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mohamed, AM.I., Omar, M.A., Hammad, M.A. et al. Development and Validation of highly Sensitive Stability Indicating Spectrofluorimetric Method for Determination of Amlodipine in Pharmaceutical Preparations and Human Plasma. J Fluoresc 26, 2141–2149 (2016). https://doi.org/10.1007/s10895-016-1910-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10895-016-1910-4