Abstract
We compared pheromone production and response for populations of western pine beetle, Dendroctonus brevicomis LeConte, from sites in northern Arizona and northern California. Volatiles were collected from individuals of both sexes that had mined as a pair in a Pinus ponderosa log for 1 d, and they were subsequently analyzed by gas chromatography coupled to mass-spectrometry. Principal component analysis of quantities of Dendroctonus pheromone components indicated strong site-associated clustering of blend composition for females but not males. Much of the clustering in females evidently was due to differences in the production of endo- and exo-brevicomin, which occurred in average ratios of 0.1:1 and 19:1 for populations in the California and Arizona sites, respectively. In the California site, exo- was better than endo-brevicomin in enhancing trap catches of both sexes to lures containing the host-tree odor α-pinene and the male-produced aggregation pheromone component frontalin. In an identical test in the Arizona site, endo- was a better adjuvant than exo-brevicomin for male attraction, whereas females did not show a significant preference. At neither location were the isomers antagonistic to one another in activity. Thus, one aggregation pheromone has apparently diverged between these populations, concurrent with published evidence that D. brevicomis on either side of the Great Basin are genetically distinct and are possibly different species. Furthermore, production of and response to the isomers of brevicomin by flying Dendroctonus frontalis Zimmermann in the Arizona site were similar to those of sympatric D. brevicomis. This interspecific signal overlap is likely sustainable since joint species mass-attacks may assist both species in overcoming host defenses, thereby increasing host availability.






Similar content being viewed by others
References
Bedard WD, Tilden PE, Wood DL, Silverstein RM, Brownlee RG, Rodin JO (1969) Western pine beetle: Field response to its sex pheromone and a synergistic host terpene, myrcene. Science 164:1284–1285
Bedard WD, Wood DL, Tilden PE, Lindahl KQ Jr., Silverstein RM, Rodin JO (1980a) Field responses of the western pine beetle and one of its predators to host- and beetle-produced compounds. J Chem Ecol 6:625–641
Bedard WD, Tilden PE, Lindahl KQ Jr., Wood DL, Rauch PA (1980b) Effects of verbenone and trans-verbenol on the response of Dendroctonus brevicomis to natural and synthetic attractant in the field. J Chem Ecol 6:997–1013
Bedard WD, Lindahl KQ Jr., Tilden PE, Wood DL (1985) Behavior of the western pine beetle during host colonization. J Chem Ecol 11:1249–1261
Berryman AA (1972) Resistance of conifers to invasion by bark beetle-fungus associations. Bioscience 22:598–602
Bertram SL, Paine TD (1994) Influence of aggregation inhibitors (verbenone and ipsdienol) on landing and attack behavior of Dendroctonus brevicomis (Coleoptera: Scolytidae). J Chem Ecol 20:1617–1629
Borden JH (1982) Aggregation pheromones. In: Mitton JB, Sturgeon KB (eds) Bark beetles in north American conifers: a system for the study of evolutionary biology. University of Texas Press, Autstin, pp. 74–139
Bracewell RR, Six DL (2014) Broadscale specificity in a bark beetle–fungal symbiosis: a spatio-temporal analysis of the mycangial fungi of the western pine beetle. Microb Ecol 28:859–870
Browne LE, Wood DL, Bedard WD, Silverstein RM, West JR (1979) Quantitative estimates of the Western pine beetle attractive pheromone components, exo-brevicomin, frontalin, and myrcene in nature. J Chem Ecol 5:397–414
Byers JA (1982) Male-specific conversion of the host plant compound, myrcene, to the pheromone, (+)-ipsdienol, in the bark beetle, Dendroctonus brevicomis. J Chem Ecol 8:363–371
Byers JA (1988) Novel diffusion-dilution method for release of semiochemicals: testing pheromone component ratios on western pine beetle. J Chem Ecol 14:199–212
Byers JA (1989) Behavioral mechanisms involved in reducing competition in bark beetles. Holarct Ecol 12:466–476
Byers JA (1995) Host tree chemistry affecting colonization in bark beetles. In: RT C, Bell WJ (eds) Chemical ecology of insects 2. Chapman and Hall, New York, pp. 154–213
Byers JA, Wood DL, Craig J, Hendry LB (1984) Attractive and inhibitory pheromones produced in the bark beetle, Dendroctonus brevicomis, during host colonization: regulation of inter- and intraspecific competition. J Chem Ecol 10:861–877
Davis TS, Hofstetter RW (2009) Effects of gallery density and species ratio on the fitness and fecundity of two sympatric bark beetles (Coleoptera: Curculionidae). Environ Entomol 38:639–650
Furniss MM, Carolin VM (1977) Western forest insects. USDA For Serv Misc Publ No 1339:654
Hofstetter RW, Chen Z, Gaylord ML, McMillin JD, Wagner MR (2008) Synergistic effects of alpha-pinene and exo-brevicomin on pine bark beetles and associated insects in Arizona. J Appl Entomol 132:387–397
Hofstetter RW, Gaylord ML, Martinson S, Wagner MR (2012) Attraction to monoterpenes and beetle-produced compounds by syntopic Ips and Dendroctonus bark beetles and their predators. Agric For Entomol 14:207–215
Hughes PR (1973) Dendroctonus: production of pheromones and related compounds in response to host monoterpenes. Z Angew Entomol 73:294–312
Kelley ST, Farrell BD (1998) Is specialization a dead end? The phylogeny of host use in Dendroctonus bark beetles (Scolytidae). Evolution 52:1731–1743
Kelley ST, Mitton JB, Paine TD (1999) Strong differentiation in mitochondrial DNA of Dendroctonus brevicomis (Coleoptera: Scolytidae) on different subspecies of ponderosa pine. Ann Entomol Soc Am 92:193–197
Lanier GN, Hendrichs JP, Flores JE (1988) Biosystematics of the Dendroctonus frontalis (Coleoptera: Scolytidae) complex. Ann Entomol Soc Am 81:403–418
Libbey LM, Morgan ME, Putnam TB, Rudinsky JA (1974) Pheromones released during inter- and intra-sex response of the scolytid beetle Dendroctonus brevicomis. J Insect Physiol 20:1667–1671
Lindgren BS (1983) A multiple funnel trap for scolytid bark beetles (Coleoptera). Can Entomol 115:229–302
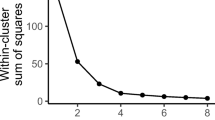
McGarigal K, Cushman S, Stafford S (2000) Multivariate statistics for wildlife and ecology research. Springer, New York
Moreno B, Macías J, Sullivan BT, Clarke SR (2008) Field response of Dendroctonus frontalis (Coleoptera: Scolytinae) to synthetic semiochemicals in Chiapas, Mexico. J Econ Entomol 101:1821–1825
Niño-Domínguez A, Sullivan BT, López-Urbina JH, Macías-Sámano JE (2015) Pheromone-mediated mate location and discrimination by two syntopic sibling species of Dendroctonus bark beetles in Chiapas, Mexico. J Chem Ecol 41:746–756
Osgood EAJ, Clark EW (1963) Methods of sexing and sex ratios of the southern pine beetle, Dendroctonus frontalis Zimm. Can Entomol 95:1106–1109
Pitman GB, Vité JP, Kinzer GW, Fentiman AF Jr. (1969) Specificity of population-aggregating pheromones in Dendroctonus. J Insect Physiol 15:363–366
Powell J, Tams J, Bentz B, Logan J (1998) Theoretical analysis of “switching” in a localized model for mountain pine beetle mass attack. J Theor Biol 194:49–63
Pureswaran DS, Sullivan BT (2012) Semiochemical emission from individual galleries of the southern pine beetle, (Coleoptera: Curculionidae: Scolytinae), attacking standing trees. J Econ Entomol 105:140–148
Pureswaran DS, Sullivan BT, Ayres MP (2006) Fitness consequences of pheromone production and host selection strategies in a tree-killing bark beetle (Coleoptera: Curculionidae: Scolytinae). Oecologia 148:720–728
Pureswaran DS, Hofstetter RW, Sullivan BT (2008a) Attraction of the southern pine beetle, Dendroctonus frontalis, to pheromone components of the western pine beetle, Dendroctonus brevicomis (Coleoptera: Curculionidae: Scolytinae), in an allopatric zone. Environ Entomol 37:70–78
Pureswaran DS, Sullivan BT, Ayres MP (2008b) High individual variation in pheromone production by tree-killing bark beetles (Coleoptera: Curculionidae: Scolytinae). Naturwissenschaften 95:33–44
Pureswaran DS, Hofstetter RW, Sullivan BT, Potter KA (2016) The role of multimodal signals in species recognition between tree-killing bark beetles in a narrow sympatric zone. Environ Entomol doi:10.1093/ee/nvw022
Raffa KF, Berryman AA (1987) Interacting selective pressures in conifer-bark beetle systems: a basis for reciprocal adaptations? Am Nat 129:234–262
Raffa KF, Phillips TW, Salom SM (1993) Strategies and mechanisms of host colonization by bark beetles. In: Schowalter TD, Filip GM (eds) Beetle-pathogen interactions in conifer forests. Academic Press, New York, pp. 103–128
Renwick JAA, Vité JP (1970) Systems of chemical communication in Dendroctonus. Contrib Boyce Thompson Inst 24:283–292
Renwick JAA, Pitman GB, Vité JP (1976) 2-phenylethanol isolated from bark beetles. Naturwissenschaften 63:198
Sanchez-Martinez G, Wagner MR (2002) Bark beetle community structure under four ponderosa pine forest stand conditions in northern Arizona. For Ecol Manag 170:145–160
Seybold SJ, Huber DPW, Lee JC, Graves AD, Bohlmann J (2006) Pine monoterpenes and pine bark beetles: a marriage of convenience for defense and chemical communication. Phytochem Rev 5:143–178
Silverstein RM, Brownlee RG, Bellas TE, Wood DL, Browne LE (1968) Brevicomin: principal sex attractant in the frass of the female western pine beetle. Science 159:889–891
Six DL, Bracewell RR (2015) Dendroctonus. In: Vega FE, Hofstetter RW (eds) Bark beetles: biology and ecology of native and invasive species. Academic Press, San Diego, pp. 305–350
Skillen, EL, Berisford, CW, Camaan, MA, Reardon, RC (1997) Semiochemicals of forest and shade tree insects in North America and management applications. USDA Forest Service Forest Health Technology Enterprise Team Publication FHTET-96-15
Smith RH (1977) Monoterpenes of ponderosa pine xylem resin in western United States. USDA, For Serv Tech Bull No 1532 p 48
Sullivan BT (2005) Electrophysiological and behavioral responses of Dendroctonus frontalis (Coleoptera: Curculionidae) to volatiles isolated from conspecifics. J Econ Entomol 98:2067–2078
Sullivan BT (2011) Southern pine beetle behavior and semiochemistry. In: RN C, KD K (eds) Southern pine beetle II. vol general technical report SRS-140. USDA Forest Service Southern Research Station Gen. Tech. Rep. SRS-140, Asheville, pp. 25–50
Sullivan BT, Mori K (2009) Spatial displacement of release point can enhance activity of an attractant pheromone synergist of a bark beetle. J Chem Ecol 35:1222–1233
Sullivan BT, Shepherd WP, Pureswaran DS, Tashiro T, Mori K (2007) Evidence that (+)-endo-brevicomin is a male-produced component of the southern pine beetle aggregation pheromone. J Chem Ecol 33:1510–1527
Symonds MRE, Elgar MA (2004) The mode of pheromone evolution: evidence from bark beetles. Proc R Soc Lond B Biol Sci 271:839–846
Vité JP, Pitman GB (1969a) Aggregation behaviour of Dendroctonus brevicomis in response to synthetic pheromnes. J Insect Physiol 15:1617–1622
Vité JP, Pitman GB (1969b) Insect and host odors in aggregation of western pine beetle. Can Entomol 101:113–117
Vité JP, Billings RF, Ware CW, Mori K (1985) Southern pine beetle: enhancement or inhibition of aggregation response mediated by enantiomers of endo-brevicomin. Naturwissenschaften 72:99–100
Wood DL (1982a) The role of pheromones, kairomones and allomones in the host selection and colonization of bark beetles. Annu Rev Entomol 27:411–446
Wood SL (1982b) The bark and ambrosia beetles of North and Central America (Coleoptera: Scolytidae), a taxonomic monograph. Great Basin Nat Mem 6:1–1356
Wood DL, Bedard WD (1977) The role of pheromones in the population dynamics of the western pine beetle. Proc XV Int Congr Entomol 15:643–652
Wood DL, Browne LE, Ewing B, Lindahl K, Bedard WD, Tilden PE, Mori K, Pitman GB, Hughes PR (1976) Western pine beetle - specificity among enantiomers of male and female components of an attractant pheromone. Science 192:896–898
Yan W, Hunt L (2002) Biplot analysis of diallel data. Crop Sci 42:21–30
Acknowledgments
We thank Joanne Barrett, Eli Jensen, Seth Davis, and Chris Foelker for trap collection, identification, and data entry. We thank Daniel Cluck for assistance with collection of infested material in California. Rob Johns and Kathy Bleiker provided comments on an early draft. Research was funded by Rocky Mountain Research Station JV aggreement 08-JV-11221633-250 with R.W.H and an operating grant to D.S.P. from the Canadian Forest Service.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pureswaran, D.S., Hofstetter, R.W., Sullivan, B.T. et al. Western Pine Beetle Populations in Arizona and California Differ in the Composition of Their Aggregation Pheromones. J Chem Ecol 42, 404–413 (2016). https://doi.org/10.1007/s10886-016-0696-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10886-016-0696-9