Abstract
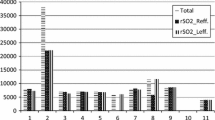
In this study we investigated the responsiveness of near-infrared spectroscopy (NIRS) recordings measuring regional cerebral tissue oxygenation (rSO2) during hypoxia in apneic divers. The goal was to mimic dynamic hypoxia as present during cardiopulmonary resuscitation, laryngospasm, airway obstruction, or the “cannot ventilate cannot intubate” situation. Ten experienced apneic divers performed maximal breath hold maneuvers under dry conditions. SpO2 was measured by Masimo™ pulse oximetry on the forefinger of the left hand. NIRS was measured by NONIN Medical’s EQUANOX™ on the forehead or above the musculus quadriceps femoris. Following apnea median cerebral rSO2 and SpO2 values decreased significantly from 71 to 54 and from 100 to 65 %, respectively. As soon as cerebral rSO2 and SpO2 values decreased monotonically the correlation between normalized cerebral rSO2 and SpO2 values was highly significant (Pearson correlation coefficient = 0.893). Prior to correlation analyses, the values were normalized by dividing them by the individual means of stable pre-apneic measurements. Cerebral rSO2 measured re-saturation after termination of apnea significantly earlier (10 s, SD = 3.6 s) compared to SpO2 monitoring (21 s, SD = 4.4 s) [t(9) = 7.703, p < 0.001, r2 = 0.868]. Our data demonstrate that NIRS monitoring reliably measures dynamic changes in cerebral tissue oxygen saturation, and identifies successful re-saturation faster than SpO2. Measuring cerebral rSO2 may prove beneficial in case of respiratory emergencies and during pulseless situations where SpO2 monitoring is impossible.




Similar content being viewed by others
References
Jöbsis FF. Noninvasive, infrared monitoring of cerebral and myocardial oxygen sufficiency and circulatory parameters. Science. 1977;198:1264–7.
Scheeren TW, Schober F, Schwarte LA. Monitoring tissue oxygenation by near infrared spectroscopy (NIRS): background and current applications. J Clin Monit Comput. 2012;26:279–87.
La Monaca M, David A, Gaeta R, Lentini S. Near infrared spectroscopy for cerebral monitoring during cardiovascular surgery. Clin Ter. 2010;161:549–53.
Vernieri F, Silvestrini M, Tibuzzi F, Pasqualetti P, Altamura C, Passarelli F, Matteis M, Rossini PM. Hemoglobin oxygen saturation as a marker of cerebral hemodynamics in carotid artery occlusion: an integrated transcranial doppler and near-infrared spectroscopy study. J Neurol. 2006;253:1459–65.
Moritz S, Kasprzak P, Arlt M, Taeger K, Metz C. Accuracy of cerebral monitoring in detecting cerebral ischemia during carotid endarterectomy: a comparison of transcranial Doppler sonography, near-infrared spectroscopy, stump pressure, and somatosensory evoked potentials. Anesthesiology. 2007;107:563–9.
Hansen JH, Schlangen J, Armbrust S, Jung O, Scheewe J, Kramer HH. Monitoring of regional tissue oxygenation with near-infrared spectroscopy during the early postoperative course after superior cavopulmonary anastomosis. Eur J Cardiothorac Surg. 2013;43:e37–43.
Beilman GJ, Groehler KE, Lazaron V, Ortner JP. Near-infrared spectroscopy measurement of regional tissue oxyhemoglobin saturation during hemorrhagic shock. Shock. 1999;12:196–200.
Rhee P, Langdale L, Mock C, Gentilello LM. Near-infrared spectroscopy: continuous measurement of cytochrome oxidation during hemorrhagic shock. Crit Care Med. 1997;25:166–70.
Zweifel C, Castellani G, Czosnyka M, Carrera E, Brady KM, Kirkpatrick PJ, Pickard JD, Smielewski P. Continuous assessment of cerebral autoregulation with near-infrared spectroscopy in adults after subarachnoid hemorrhage. Stroke J Cereb Circ. 2010;41:1963–8.
Ito N, Nanto S, Nagao K, Hatanaka T, Nishiyama K, Kai T. Regional cerebral oxygen saturation on hospital arrival is a potential novel predictor of neurological outcomes at hospital discharge in patients with out-of-hospital cardiac arrest. Resuscitation. 2012;83:46–50.
Cohn SM, Nathens AB, Moore FA, Rhee P, Puyana JC, Moore EE, Beilman GJ. Tissue oxygen saturation predicts the development of organ dysfunction during traumatic shock resuscitation. J Trauma. 2007;62:44–55; discussion 54–55.
Moore FA, Nelson T, McKinley BA, Moore EE, Nathens AB, Rhee P, Puyana JC, Beilman GJ, Cohn SM. Massive transfusion in trauma patients: tissue hemoglobin oxygen saturation predicts poor outcome. J Trauma. 2008;64:1010–23.
Heringlake M, Garbers C, Käbler JH, Anderson I, Heinze H, Schön J, Berger KU, Dibbelt L, Sievers HH, Hanke T. Preoperative cerebral oxygen saturation and clinical outcomes in cardiac surgery. Anesthesiology. 2012;114:58–69.
Eichhorn JH. Pulse oximetry as a standard of practice in anesthesia. Anesthesiology. 1993;78:423–6.
Shah N, Trivedi NK, Clack SL, Shah M, Shah PP, Barker S. Impact of hypoxemia on the performance of cerebral oximeter in volunteer subjects. J Neurosurg Anesthesiol. 2000;12:201–9.
Ricci M, Lombardi P, Schultz S, Galindo A, Coscarella E, Vasquez A, Rosenkranz E. Near-infrared spectroscopy to monitor cerebral oxygen saturation in single-ventricle physiology. J Thorac Cardiovasc Surg. 2006;131:395–402.
Kusaka T, Isobe K, Nagano K, Okubo K, Yasuda S, Kondo M, Itoh S, Hirao K, Onishi S. Quantification of cerebral oxygenation by full-spectrum near-infrared spectroscopy using a two-point method. Comp Biochem Physiol A Mol Integr Physiol. 2002;132:121–32.
Ainslie PN, Ogoh S. Regulation of cerebral blood flow in mammals during chronic hypoxia: a matter of balance. Exp Physiol. 2010;95:251–62.
Watzman HM, Kurth CD, Montenegro LM, Rome J, Steven JM, Nicolson SC. Arterial and venous contributions to near-infrared cerebral oximetry. Anesthesiology. 2000;93:947–53.
Reynolds JC, Salcido D, Koller AC, Sundermann ML, Frisch A, Suffoletto BP, Menegazzi JJ. Tissue oximetry by near-infrared spectroscopy in a porcine model of out-of-hospital cardiac arrest and resuscitation. Resuscitation. 2013;84:843–7.
Schön J, Heringlake M, Berger K, Volker Groesdonk H, Sedemund-Adib B, Paarmann H. Relationship between mixed venous oxygen saturation and regional cerebral oxygenation in awake, spontaneously breathing cardiac surgery patients. Minerva Anestesiol. 2011;77:952–8.
Moreno GE, Pilán ML, Manara C, Magliola R, Vassallo JC, Balestrini M, Lenz AM, Krynski M, Althabe M, Landry L. Regional venous oxygen saturation versus mixed venous saturation after paediatric cardiac surgery. Acta Anaesthesiol Scand. 2013;57:373–9.
Zander R, Rehfisch P. Noninvasive measurement of cerebral hemoglobin-oxygen saturation. Anästhesiol Intensivmed Notfallmedizin Schmerzther (AINS). 1997;32(2 Suppl):220–3.
Alboni P, Alboni M, Gianfranchi L. Diving bradycardia: a mechanism of defence against hypoxic damage. J Cardiovasc Med. 2011;12:422–7.
Watkin SL, Spencer SA, Dimmock PW, Wickramasinghe YA, Rolfe P. A comparison of pulse oximetry and near infrared spectroscopy (NIRS) in the detection of hypoxaemia occurring with pauses in nasal airflow in neonates. J Clin Monit Comput. 1999;15:441–7.
Pizza F, Biallas M, Wolf M, Werth E, Bassetti CL. Nocturnal cerebral hemodynamics in snorers and in patients with obstructive sleep apnea: a near-infrared spectroscopy study. Sleep. 2010;33:205–10.
Mohsenin V. Obstructive sleep apnea and hypertension: a critical review. Curr Hypertens Rep. 2014;16:482.
Nishiyasu T, Tsukamoto R, Kawai K, Hayashi K, Koga S, Ichinose M. Relationships between the extent of apnea-induced bradycardia and the vascular response in the arm and leg during dynamic two-legged knee extension exercise. Am J Physiol. 2012;302:H864–71.
Perini R, Tironi A, Gheza A, Butti F, Moia C, Ferretti G. Heart rate and blood pressure time courses during prolonged dry apnoea in breath-hold divers. Eur J Appl Physiol. 2008;104:1–7.
Leuenberger UA, Hardy JC, Herr MD, Gray KS, Sinoway LI. Hypoxia augments apnea-induced peripheral vasoconstriction in humans. J Appl Physiol. 1985;90:1516–22.
Macefield VG, Gandevia SC, Henderson LA. Neural sites involved in the sustained increase in muscle sympathetic nerve activity induced by inspiratory capacity apnea: a fMRI study. J Appl Physiol. 2006;100:266–73.
Heusser K, Dzamonja G, Tank J, Palada I, Valic Z, Bakovic D, Obad A, Ivancev V, Breskovic T, Diedrich A, Joyner MJ, Luft FC, Jordan J, Dujic Z. Cardiovascular regulation during apnea in elite divers. Hypertension. 2009;53:719–24.
Kolb JC, Ainslie PN, Ide K, Poulin MJ. Protocol to measure acute cerebrovascular and ventilatory responses to isocapnic hypoxia in humans. Respir Physiol Neurobiol. 2004;141:191–9.
Wilson MH, Newman S, Imray CH. The cerebral effects of ascent to high altitudes. Lancet Neurol. 2009;8:175–91.
Ainslie PN, Barach A, Murrell C, Hamlin M, Hellemans J, Ogoh S. Alterations in cerebral autoregulation and cerebral blood flow velocity during acute hypoxia: rest and exercise. Am J Physiol Heart Circ Physiol. 2007;292:H976–83.
Rupp T, Leti T, Jubeau M, Millet GY, Bricout VA, Levy P, Wuyam B, Perrey S, Verges S. Tissue deoxygenation kinetics induced by prolonged hypoxic exposure in healthy humans at rest. J Biomed Opt. 2013;18:095002.
Ahn A, Nasir A, Malik H, D’Orazi F, Parnia S. A pilot study examining the role of regional cerebral oxygen saturation monitoring as a marker of return of spontaneous circulation in shockable (VF/VT) and non-shockable (PEA/asystole) causes of cardiac arrest. Resuscitation. 2013;84:1713–6.
Schewe JC, Thudium MO, Kappler J, Steinhagen F, Eichhorn L, Erdfelder F, Heister U, Ellerkmann R. Monitoring of cerebral oxygen saturation during resuscitation in out-of-hospital cardiac arrest: a feasibility study in a physician staffed emergency medical system. Scand J Trauma Resusc Emerg Med. 2014;5:58.
Meex I, De Deyne C, Dens J, Scheyltjens S, Lathouwers K, Boer W, Vundelinckx G, Heylen R, Jans F. Feasibility of absolute cerebral tissue oxygen saturation during cardiopulmonary resuscitation. Crit Care. 2013;17:R36.
Ahn A, Yang J, Inigo-Santiago L, Parnia S. A feasibility study of cerebral oximetry monitoring during the post-resuscitation period in comatose patients following cardiac arrest. Resuscitation. 2014;85:522–6. doi:10.1016/j.resuscitation.2013.
Acknowledgments
Special thanks to all volunteers of the study and to Anja Reckendorf for critical reading of the manuscript. L. Eichhorn was supported by a scholarship of Else-Kröner-Fresenius Stiftung.
Conflict of interest
The authors have no conflicts of interest. The authors have no financial relationship with NONIN Medical’s EQUANOX™. All machines were already available in our University hospital.
Ethical standard
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Author information
Authors and Affiliations
Corresponding author
Additional information
Lars Eichhorn and Felix Erdfelder have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Eichhorn, L., Erdfelder, F., Kessler, F. et al. Evaluation of near-infrared spectroscopy under apnea-dependent hypoxia in humans. J Clin Monit Comput 29, 749–757 (2015). https://doi.org/10.1007/s10877-015-9662-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10877-015-9662-2