Abstract
Purpose
Gastrointestinal disease occurs frequently in antibody deficiencies. This study aims to explore the relation between gastrointestinal infections and mucosal homeostasis in patients with antibody deficiencies.
Methods
We performed an observational study including 54 pediatric antibody deficient patients (48 % CVID, 41 % CVID-like, 11 % XLA) and 66 healthy controls. Clinical symptom scores and stool samples were collected prospectively. Stool samples were evaluated for bacteria, parasites, viruses, secretory IgA- and for calprotectin levels. Results were compared between patients and controls.
Results
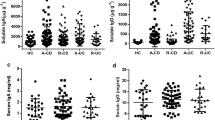
24 % of antibody deficient patients versus 9 % of healthy controls tested positive for gastrointestinal viruses (p = 0.028). Fecal calprotectin levels were significantly higher in virus positive patients compared to virus negative patients (p = 0.002). However, in controls, fecal calprotectin levels were similar between virus positive and virus negative controls. Moreover, gastrointestinal virus positive patients had low serum IgA levels in 13/14 cases (94 %) versus 40/62 (62 %) patients in the virus negative patient group (p = 0.04). The virus positive patient group also displayed significantly lower secretory IgA levels in stool (median 13 ug/ml) than patients without gastrointestinal viruses detected or healthy controls (median 155 ug/ml) (p = 0.046).
Conclusion
We here report an increased prevalence of gastrointestinal viruses and gastrointestinal complaints in antibody deficient patients. Patients that tested positive for gastrointestinal viruses showed diminished serum- and secretory IgA levels, and only in patients, virus positivity was associated with signs of mucosal inflammation. These findings suggest that particularly patients with low IgA are at risk for longstanding replication of gastrointestinal viruses, which may eventually result in CVID-related enteropathy.


Similar content being viewed by others
Abbreviations
- CVID:
-
Common variable immunodeficiency
- GI:
-
Gastrointestinal
- IBD:
-
Inflammatory bowel disease
- IgA:
-
Immunoglobulin A
- PID:
-
Primary immunodeficiencies
- XLA:
-
X-linked agammaglobulinemia
- RT-PCR:
-
Real time polymerase chain reaction
- ELISA:
-
Enzyme-linked immunosorbent assay
References
Salzer U, Warnatz K, Peter HH. Common variable immunodeficiency - an update. Arthritis Res Ther. 2012;14(5):223. doi:10.1186/ar4032.
Conley ME, Notarangelo LD, Etzioni A. Diagnostic criteria for primary immunodeficiencies. Representing PAGID (Pan-American Group for Immunodeficiency) and ESID (European Society for Immunodeficiencies). Clin Immunol. 1999;93(3):190–7.
Park MA, Li JT, Hagan JB, Maddox DE, Abraham RS. Common variable immunodeficiency: a new look at an old disease. Lancet. 2008;372(9637):489–502. doi:10.1016/S0140-6736(08)61199-X.
Agarwal S, Mayer L. Pathogenesis and treatment of gastrointestinal disease in antibody deficiency syndromes. J Allergy Clin Immunol. 2009;124(4):658–64. doi:10.1016/j.jaci.2009.06.018.
Agarwal S, Mayer L. Diagnosis and treatment of gastrointestinal disorders in patients with primary immunodeficiency. Clin Gastroenterol Hepatol. 2013;11(9):1050–63. doi:10.1016/j.cgh.2013.02.024.
Resnick ES, Moshier EL, Godbold JH, Cunningham-Rundles C. Morbidity and mortality in common variable immune deficiency over 4 decades. Blood. 2012;119(7):1650–7. doi:10.1182/blood-2011-09-377945.
Chapel H, Lucas M, Lee M, Bjorkander J, Webster D, Grimbacher B, et al. Common variable immunodeficiency disorders: division into distinct clinical phenotypes. Blood. 2008;112(2):277–86. doi:10.1182/blood-2007-11-124545.
Malamut G, Verkarre V, Suarez F, Viallard JF, Lascaux AS, Cosnes J, et al. The enteropathy associated with common variable immunodeficiency: the delineated frontiers with celiac disease. Am J Gastroenterol. 2010;105(10):2262–75. doi:10.1038/ajg.2010.214.
Teahon K, Webster AD, Price AB, Weston J, Bjarnason I. Studies on the enteropathy associated with primary hypogammaglobulinaemia. Gut. 1994;35(9):1244–9.
Daniels JA, Lederman HM, Maitra A, Montgomery EA. Gastrointestinal tract pathology in patients with common variable immunodeficiency (CVID): a clinicopathologic study and review. Am J Surg Pathol. 2007;31(12):1800–12. doi:10.1097/PAS.0b013e3180cab60c.
Agarwal S, Smereka P, Harpaz N, Cunningham-Rundles C, Mayer L. Characterization of immunologic defects in patients with common variable immunodeficiency (CVID) with intestinal disease. Inflamm Bowel Dis. 2011;17(1):251–9. doi:10.1002/ibd.21376.
Packwood K, Drewe E, Staples E, Webster D, Witte T, Litzman J, et al. NOD2 polymorphisms in clinical phenotypes of common variable immunodeficiency disorders. Clin Exp Immunol. 2010;161(3):536–41. doi:10.1111/j.1365-2249.2010.04216.x.
Akram S, Murray JA, Pardi DS, Alexander GL, Schaffner JA, Russo PA, et al. Adult autoimmune enteropathy: Mayo Clinic Rochester experience. Clin Gastroenterol Hepatol. 2007;5(11):1282–90. doi:10.1016/j.cgh.2007.05.013. quiz 45.
Catassi C, Mirakian R, Natalini G, Sbarbati A, Cinti S, Coppa GV, et al. Unresponsive enteropathy associated with circulating enterocyte autoantibodies in a boy with common variable hypogammaglobulinemia and type I diabetes. J Pediatr Gastroenterol Nutr. 1988;7(4):608–13.
Cunningham-Rundles C, Bodian C. Common variable immunodeficiency: clinical and immunological features of 248 patients. Clin Immunol. 1999;92(1):34–48. doi:10.1006/clim.1999.4725.
Quinti I, Soresina A, Spadaro G, Martino S, Donnanno S, Agostini C, et al. Long-term follow-up and outcome of a large cohort of patients with common variable immunodeficiency. J Clin Immunol. 2007;27(3):308–16. doi:10.1007/s10875-007-9075-1.
Oksenhendler E, Gerard L, Fieschi C, Malphettes M, Mouillot G, Jaussaud R, et al. Infections in 252 patients with common variable immunodeficiency. Clin Infect Dis. 2008;46(10):1547–54. doi:10.1086/587669.
Resnick ES, Cunningham-Rundles C. The many faces of the clinical picture of common variable immune deficiency. Curr Opin Allergy Clin Immunol. 2012;12(6):595–601. doi:10.1097/ACI.0b013e32835914b9.
Mantis NJ, Forbes SJ. Secretory IgA: arresting microbial pathogens at epithelial borders. Immunol Investig. 2010;39(4–5):383–406. doi:10.3109/08820131003622635.
Cerutti A, Cols M, Gentile M, Cassis L, Barra CM, He B, et al. Regulation of mucosal IgA responses: lessons from primary immunodeficiencies. Ann N Y Acad Sci. 2011;1238:132–44. doi:10.1111/j.1749-6632.2011.06266.x.
Mantis NJ, Rol N, Corthesy B. Secretory IgA’s complex roles in immunity and mucosal homeostasis in the gut. Mucosal Immunol. 2011;4(6):603–11. doi:10.1038/mi.2011.41.
Espanol T, Catala M, Hernandez M, Caragol I, Bertran JM. Development of a common variable immunodeficiency in IgA-deficient patients. Clin Immunol Immunopathol. 1996;80(3 Pt 1):333–5.
van de Ven AA, Douma JW, Rademaker C, van Loon AM, Wensing AM, Boelens JJ, et al. Pleconaril-resistant chronic parechovirus-associated enteropathy in agammaglobulinaemia. Antivir Ther. 2011;16(4):611–4. doi:10.3851/IMP1792.
Joshi S, Lewis SJ, Creanor S, Ayling RM. Age-related faecal calprotectin, lactoferrin and tumour M2-PK concentrations in healthy volunteers. Ann Clin Biochem. 2010;47(Pt 3):259–63. doi:10.1258/acb.2009.009061.
Roseth AG, Schmidt PN, Fagerhol MK. Correlation between faecal excretion of indium-111-labelled granulocytes and calprotectin, a granulocyte marker protein, in patients with inflammatory bowel disease. Scand J Gastroenterol. 1999;34(1):50–4.
Lundberg JO, Hellstrom PM, Fagerhol MK, Weitzberg E, Roseth AG. Technology insight: calprotectin, lactoferrin and nitric oxide as novel markers of inflammatory bowel disease. Nat Clin Pract Gastroenterol Hepatol. 2005;2(2):96–102. doi:10.1038/ncpgasthep0094.
Cerutti A, Rescigno M. The biology of intestinal immunoglobulin a responses. Immunity. 2008;28(6):740–50. doi:10.1016/j.immuni.2008.05.001.
Cerutti A, Chen K, Chorny A. Immunoglobulin responses at the mucosal interface. Annu Rev Immunol. 2011;29:273–93. doi:10.1146/annurev-immunol-031210-101317.
Pabst O. New concepts in the generation and functions of IgA. Nat Rev Immunol. 2012;12(12):821–32. doi:10.1038/nri3322.
Shulzhenko N, Morgun A, Hsiao W, Battle M, Yao M, Gavrilova O, et al. Crosstalk between B lymphocytes, microbiota and the intestinal epithelium governs immunity versus metabolism in the gut. Nat Med. 2011;17(12):1585–93. doi:10.1038/nm.2505.
Dion C, Montagne P, Bene MC, Faure G. Measurement of faecal immunoglobulin a levels in young children. J Clin Lab Anal. 2004;18(3):195–9. doi:10.1002/jcla.20022.
Aghamohammadi A, Mohammadi J, Parvaneh N, Rezaei N, Moin M, Espanol T, et al. Progression of selective IgA deficiency to common variable immunodeficiency. Int Arch Allergy Immunol. 2008;147(2):87–92. doi:10.1159/000135694.
Acknowledgements
The authors would like to thank Jessica van der Aa and Géanne Krabben for assistance with participant inclusions and Karin van Veghel and Lysette Ebskamp of U-DAIR with determination of sIgA levels.
Funding
This study was supported by non-restricted educational grants from Baxter The Netherlands and GlaxoSmithKline The Netherlands.
Author information
Authors and Affiliations
Corresponding author
Additional information
A.A.J.M. van de Ven W.J.M. Janssen, A.M.J. Wensing and J.M. van Montfrans have equal contributions.
Rights and permissions
About this article
Cite this article
van de Ven, A.A.J.M., Janssen, W.J.M., Schulz, L.S. et al. Increased Prevalence of Gastrointestinal Viruses and Diminished Secretory Immunoglobulin a Levels in Antibody Deficiencies. J Clin Immunol 34, 962–970 (2014). https://doi.org/10.1007/s10875-014-0087-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-014-0087-3