Abstract
Purpose
Autosomal recessive hyper-IgE syndrome is a rare combined immunodeficiency characterized by susceptibility to viral infections, atopic eczema, high serum IgE and defective T cell activation. The genetic etiologies are diverse. Null mutations in DOCK8 and TYK2 are responsible for many cases. This study aims to provide a detailed clinical and immunological characterization of the disease and explore the underlying genetic defects among a large series of patients followed by a single center. The available data might improve our understanding of the disease pathogenesis and prognosis.
Methods
Clinical data of twenty-five patients diagnosed with AR-HIES were collected. Seventeen patients screened for STAT3, TYK2 and DOCK8 mutations.
Results
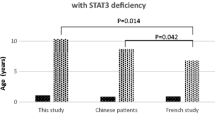
Sinopulmonary infections, dermatitis, hepatic disorders, cutaneous and systemic bacterial, fungal and viral infections were the most common clinical features. The rate of hepatic disorders and systemic infections were high. Twelve patients died with a median age of 10 years. CMV infection was the only statistically significant predicting factor for poor prognosis (early death). Three novel DOCK8 mutations and two large deletions were found in thirteen patients. No mutations found in STAT3 or TYK2 genes.
Conclusion
Autosomal recessive hyper-IgE syndrome is a combined immunodeficiency disease characterized by high morbidity and mortality rate. The different genetic background and environmental factors may explain the more severe phenotypes seen in our series. DOCK8 defect is the most common identified genetic cause. Patients with no identified genetic etiology are likely to carry mutations in the regulatory elements of genes tested or in novel genes that are yet to be discovered.





Similar content being viewed by others
References
Freeman AF, Holland SM. The hyper IgE syndrome. Immunol Allergy Clin North Am. 2008; 28(2):277–viii.
Grimbacher B, Holland SM, Gallin JI, Greenberg F, Hill SC, Malech HL, Miller JA, O'Connell AC, Dent B, Puck JM. Hyper-IgE syndrome with recurrent infections-an autosomal dominant multisystem disorder. N Engl J Med. 1999;340:692–702.
Minegishi Y, Saito M, Tsuchiya S, Tsuge I, Takada H, Hara T, Kawamura N, Ariga T, Pasic S, Stojkovic O, Metin A, Karasuyama H. Dominant-negative mutations in the DNA-binding domain of STAT3 cause hyper-IgE syndrome. Nature. 2007;448:1058–62.
Renner ED, Puck JM, Holland SM, Schmitt M, Weiss M, Frosch M, Bergmann M, Davis J, Belohradsky BH, Grimbacher B. Autosomal recessive hyperimmunoglobulin E syndrome: a distinct disease entity. J Pediatr. 2004;144:939.
Minegishi Y, Saito M, Morio T, Watanabe K, Agematsu K, Tsuchiya S, Takada H, Hara T, Kawamura N, Ariga T, Kaneko H, Kondo N, Tsuge I, Yachie A, Sakiyama Y, Iwata T, Bessho F, Ohishi T, Joh K, Imai K, Kogawa K, Shinohara M, Fujieda M, Wakiguchi H, Pasic S, Abinun M, Ochs HD, Renner ED, Jansson A, Belohradsky BH, Metin A, Shimizu N, Mizutani S, Miyawaki T, Nonoyama S, Karasuyama H. Human tyrosine kinase 2 deficiency reveals its requisite roles in multiple cytokine signals involved in innate and acquired immunity. Immunity. 2006;25(5):745–55.
Zhang Q, Su HC. hyper immunoglobulin E in pediatrics. Current Openion in Pediatrics. 2011.
Zhang Q, Davis JC, Lamborn IT, Freeman AF, Jing H, Favreau AJ, Matthews HF, Davis J, Turner ML, Uzel G, Holland SM, Su HC: Combined Immunodeficiency Associated with DOCK8 Mutations. N Engl J Med 2009; 19; 361(21): 2046–2055.
Engelhardt KR, McGhee S, Winkler S, Sassi A, Woellner C, Lopez-Herrera G, Chen A, Kim HS, Lioret MG, Schulze I, et al. Large deletions and point mutations involving DOCK8 in the autosomal recessive form of the Hyper-IgE syndrome. J Allergy ClinImmunol. 2009;124(6):1289.
Lee W, Huang J, Lin S, Yeh K, Chen L, Hsieh M, Huang Y, Kuo H, Yang KD, Yu H, Jaing T, Yang C: Clinical Aspects and Genetic Analysis of Taiwanese Patients with the Phenotype of Hyper-Immunoglobulin E Recurrent Infection Syndromes [HIES]. J ClinImmunol 2010 Dec 1[online].
Su HC. Dedicator of cytokinesis 8 [DOCK8] deficiency. Current Opinion in Allergy and Clinical Immunology. 2010;10:515–20.
Zhang Q, Davis JC, Dove CG, Su HC. Genetic, clinical and laboratory markers for Dock8 immunodeficiency syndrome. Disease Markers. 2010;29:131–9.
Randall KL, Lambe T, Johnson AL, Treanor B, Kucharska E, Domaschenz H, et al. Dock8 mutations cripple B cell immunological synapses, germinal centers and long-lived antibody production. Nat Immunol. 2009;10(12):1283–91.
Fung I, Russell SM, Oliaro J. Interplay of polarity proteins and GTPases in T-Lymphocyte function. Clin Dev Immunol. 2012;2012:417485.
Lambe T, Crawford G, Johnson AL, Crockford TL, Bouriez-Jones T, Smyth AM, et al. DOCK8 is essential for T-cell survival and the maintenance of CD8+ T-cell memory. Eur J Immunol. 2011;41(12):3423–35.
Harada Y, Tanaka Y, Terasawa M, Pieczyk M, Habiro K, Katakai T, et al. DOCK8 is a Cdc42 activator critical for interstitial dendritic cell migration during immune responses. Blood. 2012;119(19):4451–61.
El Mouzan MI, Al Salloum AA, Al Herbish AS, Qurachi MM, Al Omar AA. Consanguinity and major genetic disorders in Saudi children: a community-based cross-sectional study. Ann Saudi Med. 2008;28(3):169–73.
Grimbacher B, Schäffer AA, Holland SM, Davis J, Gallin JI, Malech HL, et al. Genetic linkage of hyper-IgE syndrome to chromosome 4. Am J Hum Genet. 1999;65(3):735–44.
Sanal O, Jing H, Ozgur T, Ayvaz D, Strauss-Albee DM, Ersoy-Evans S, et al. Additional diverse findings expand the clinical presentation of DOCK8 deficiency. J Clin Immunol. 2012;32(4):698–708.
Al-Herz W, Ragupathy R, Massaad MJ, Al-Attiyah R, Nanda A, Engelhardt KR, Grimbacher B, Notarangelo L, Chatila T, Geha RS. Clinical, immunologic and genetic profiles of DOCK8-deficient patients in Kuwait. Clin Immunol. 2012;143(3):266–72.
Dasouki M, Okonkwo KC, Ray A, Folmsbeel CK, Gozales D, Keles S, Puck JM, Chatila T: Deficient T Cell Receptor Excision Circles [TRECs] In autosomal recessive hyper IgE syndrome caused by DOCK8 mutation: Implications for pathogenesis and potential detection by newborn screening. Clinical immunology. 2011;article in press.
Al Khatib S, Keles S, Garcia-Lloret M, Karakoc-Aydiner E, Reisli I, Artac H, Camcioglu Y, Cokugras H, Somer A, Kutukculer N, Yilmaz M, Ikinciogullari A, Yegin O, Yüksek M, Genel F, Kucukosmanoglu E, Baki A, Bahceciler NN, Rambhatla A, Nickerson DW, McGhee S, Barlan IB, Chatila T: Defects along the T[H]17 differentiation pathway underlie genetically distinct forms of the hyper IgE syndrome. JACI 2009;124(2):342–8, 348.e1-5
Gatz SA, Benninghoff U, Schütz C, Schulz A, Hönig M, Pannicke U, Holzmann KH, Schwarz K, Friedrich W. Curative treatment of autosomal-recessive hyper-IgE syndrome by hematopoietic cell transplantation. Bone Marrow Transplantation. 2011;46:552–6.
Bittner TC, Pannicke U, Renner ED, Notheis G, Hoffmann F, Belohradsky BH, Wintergerst U, Hauser M, Klein B, Schwarz K, Schmid I, Albert MH. Successful long-term correction of autosomal recessive hyper-IgE syndrome due to DOCK8 deficiency by hematopoietic stem cell transplantation. KlinPadiatr. 2010;222(6):351–5.
McDonald DR, Massaad MJ, Johnston A, Keles S, Chatila T, Geha RS, Pai SY. Successful engraftment of donor marrow after allogeneic hematopoietic cell transplantation in autosomal-recessive hyper-IgE syndrome caused by dedicator of cytokinesis 8 deficiency. J Allergy Clin Immunol. 2010;126(6):1304–5.
Barlogis V, Galambrun C, Chambost H, Lamoureux-Toth S, Petit P, Stephan JL et al.: Successful allogenic hematopoietic stem cell transplantation for DOCK8 deficiency. J Allergy Clin Immunol. 2011; 128(2):420–22.e2.
Metin A, Tavil B, Azik F, Azkur D, Ok-Bozkaya I, Kocabas C, Tunc B, Uckan D. Successful bone marrow transplantation for DOCK8 deficient hyper IgE syndrome. Pediatr Transplant. 2012;16(4):398–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hamoud Al-Mousa and Abbas Hawwari share senior co-authorship.
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Esm 1
(DOCX 17.8 kb)
Fig. S1
The family pedigrees. Note the high rate of death. (PPT 148 kb)
Fig. S2
Schematic representation of the wild-type variant 1 DOCK8 protein and the expected effect of the indicated mutations on the structure of the protein. Yellow and red boxes indicate the locations of the main domains of the DOCK8 protein. (PPT 72 kb)
Rights and permissions
About this article
Cite this article
Alsum, Z., Hawwari, A., Alsmadi, O. et al. Clinical, Immunological and Molecular Characterization of DOCK8 and DOCK8-like Deficient Patients: Single Center Experience of Twenty Five Patients. J Clin Immunol 33, 55–67 (2013). https://doi.org/10.1007/s10875-012-9769-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-012-9769-x