Abstract
Purpose
Human cytomegalovirus (CMV) active infection (CMV infection) poses serious risks to CMV-seropositive heart transplant recipients. We evaluated the usefulness of simultaneous assessment of CMV-specific values for parameters of the humoral (antibodies) and cellular (CD4+ and CD8+ T-cells) immune responses in the identification of heart recipients at risk of developing CMV infection after transplantation.
Methods
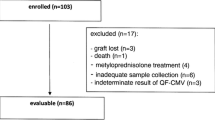
We prospectively studied 38 CMV-seropositive heart recipients. Anti-CMV antibody titers were assessed using enzyme-linked immunosorbent assays. CD4+ and CD8+ T-cell responses to overlapping peptide pools of the CMV proteins pp65 and immediate early protein-1 (IE1) were evaluated by flow cytometry. Immunological studies were performed before transplantation and at 30 days after transplantation. Patients with CMV infection were compared with heart recipients without CMV infection.
Results
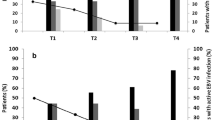
During the 6-month follow-up period, 13 (34.2%) patients developed CMV infection. At baseline, the mean anti-CMV-IgG antibody titer was lower in patients who developed CMV infection. This difference remained at 30 days after transplantation. One month after transplantation, the mean percentage of IE1-specific CD8+ T cells that are IFNg-positive (CD8/IFNg + IE1) was lower in CMV-infected patients. The predictive value of these variables at 30 days was increased when they were combined. Cox regression analysis revealed an association between the risk of developing CMV infection and the combination marker (low anti-CMV titer [<16,100] and low CD8/IFNg + IE1 percentages [<0.40%], relative hazard, 6.07; p = 0.019). The combination marker remained significant after adjustment for clinical variables.
Conclusions
This novel approach of a simultaneous assessment of specific anti-CMV antibody titers and CD8/IFNg + IE1 percentages might help identify heart transplant recipients with an increased risk of developing CMV infection.





Similar content being viewed by others
References
Gurgui M, Munoz P. Infection in heart transplantation. Enferm Infecc Microbiol Clin. 2007;25(9):587–97.
Pérez-Sola MJ, Castón JJ, Solana R, Rivero A, Torre-Cisneros J. Indirect effects of cytomegalovirus infection in solid organ transplant recipients. Enferm Infecc Microbiol Clin. 2008;26(1):38–47.
Avery RK. Prevention and treatment of cytomegalovirus infection and disease in heart transplant recipients. Curr Opin Cardiol. 1998;13(2):122–9.
Potena L, Holweg CT, Vana ML, et al. Frequent occult infection with Cytomegalovirus in cardiac transplant recipients despite antiviral prophylaxis. J Clin Microbiol. 2007;45(6):1804–10.
Kijpittayarit-Arthurs S, Eid AJ, Kremers WK, et al. Clinical features and outcomes of delayed-onset primary cytomegalovirus disease in cardiac transplant recipients. J Heart Lung Transplant. 2007;26(10):1019–24.
Li F, Kenyon KW, Kirby KA, et al. Incidence and clinical features of ganciclovir-resistant cytomegalovirus disease in heart transplant recipients. Clin Infect Dis. 2007;45(4):439–47.
Baldanti F, Lilleri D, Gerna G. Monitoring human cytomegalovirus infection in transplant recipients. J Clin Virol. 2008;41(3):237–41.
Kotton CN, Kumar D, Caliendo AM, et al. International consensus guidelines on the management of cytomegalovirus in solid organ transplantation. Transplantation. 2010;89(7):779–95.
Sarmiento E, Rodriguez-Molina JJ, Fernandez-Yañez J, et al. IgG monitoring to identify the risk for development of infection in heart transplant recipients. Transpl Infect Dis. 2006;8(1):49–53.
Sarmiento E, Lanio N, Gallego A, et al. Immune monitoring of anti cytomegalovirus antibodies and risk of cytomegalovirus disease in heart transplantation. Int Immunopharmacol. 2009;9(6):649–52.
Bunde T, Kirchner A, Hoffmeister B, et al. Protection from cytomegalovirus after transplantation is correlated with immediate early 1-specific CD8 T cells. J Exp Med. 2005;201(7):1031–6.
Sester U, Gärtner BC, Wilkens H, et al. Differences in CMV-specific T-cell levels and long-term susceptibility to CMV infection after kidney, heart and lung transplantation. Am J Transplant. 2005;5(6):1483–9.
Gerna G, Lilleri D, Fornara C, et al. Monitoring of human cytomegalovirus-specific CD4 and CD8 T-cell immunity in patients receiving solid organ transplantation. Am J Transplant. 2006;6(10):2356–64.
Egli A, Binet I, Binggeli S, et al. Cytomegalovirus-specific T-cell responses and viral replication in kidney transplant recipients. J Transl Med. 2008;6:29.
Mattes FM, Vargas A, Kopycinski J, et al. Functional impairment of cytomegalovirus specific CD8 T cells predicts high-level replication after renal transplantation. Am J Transplant. 2008;8(5):990–9.
Gamadia LE, Remmerswaal EB, Weel JF, Bemelman F, van Lier RA, Ten Berge IJ. Primary immune responses to human CMV: a critical role for IFN-gamma-producing CD4+ T cells in protection against CMV disease. Blood. 2003;101(7):2686–92.
La Rosa C, Limaye AP, Krishnan A, Longmate J, Diamond DJ. Longitudinal assessment of cytomegalovirus (CMV)-specific immune responses in liver transplant recipients at high risk for late CMV disease. J Infect Dis. 2007;195(5):633–44.
Opelz G, Döhler B, Ruhenstroth A. Cytomegalovirus prophylaxis and graft outcome in solid organ transplantation: a collaborative transplant study report. Am J Transplant. 2004;4(6):928–36.
Gerna G, Lilleri D, Chiesa A, et al. Virologic and immunologic monitoring of cytomegalovirus to guide preemptive therapy in solid-organ transplantation. Am J Transplant. 2011;11(11):2463–71.
Bunk S, Schaffert H, Schmid B, et al. Chlamydia pneumoniae-induced memory CD4+ T-cell activation in human peripheral blood correlates with distinct antibody response patterns. Clin Vaccine Immunol. 2010;17(5):705–12.
Dasari V, Smith C, Zhong J, Scott G, Rawlinson W, Khanna R. Recombinant glycoprotein B vaccine formulation with Toll-like receptor 9 agonist and immune-stimulating complex induces specific immunity against multiple strains of cytomegalovirus. J Gen Virol. 2011;92:1021–31.
Sabbaj S, Pass RF, Goepfert PA, Pichon S. Glycoprotein B vaccine is capable of boosting both antibody and CD4 T-cell responses to cytomegalovirus in chronically infected women. J Infect Dis. 2011;203(11):1534–41.
Bonaros N, Mayer B, Schachner T, et al. CMV-hyperimmune globulin for preventing cytomegalovirus infection and disease in solid organ transplant recipients: a meta-analysis. Clin Transplant. 2008;22:89–97.
Peggs KS, Thomson K, Samuel E, et al. Directly selected cytomegalovirus-reactive donor T cells confer rapid and safe systemic reconstitution of virus-specific immunity following stem cell transplantation. Clin Infect Dis. 2011;52:49–57.
Engstrand M, Lidehall AK, Totterman TH, Herrman B, Eriksson BM, Korsgren O. Cellular responses to cytomegalovirus in immunosuppressed patients: circulating CD8+ T cells recognizing CMVpp 65 are present but display functional impairment. Clin Exp Immunol. 2003;132(1):96–104.
Sester M, Sester U, Gärtner B, et al. Levels of virus-specific CD4 T cells correlate with cytomegalovirus control and predict virus-induced disease after renal transplantation. Transplantation. 2001;71(9):1287–94.
Gratama JW, Brooimans RA, van der Holt B, et al. Monitoring cytomegalovirus IE-1 and pp 65-specific CD4+ and CD8+ T-cell responses after allogeneic stem cell transplantation may identify patients at risk for recurrent CMV reactivations. Cytometry B Clin Cytom. 2008;74(4):211–20.
Kichner A, Hoffmeister B, Cherepnev-G G, et al. Dissection of the CMV specific T-cell response is required for optimized cardiac transplant monitoring. J Med Virol. 2008;80(9):1604–14.
Zhu J, Shearer GM, Marincola FM, et al. Discordant cellular and humoral immune responses to cytomegalovirus infection in healthy blood donors: existence of a Th1-type dominant response. Int Immunol. 2001;13(6):785–90.
Manuel O, Pang XL, Humar A, et al. An assessment of donor-to-recipient transmission patterns of human cytomegalovirus by analysis of viral genomic variants. J Infect Dis. 2009;199(11):1621–28.
Manuel O, Asberg A, Pang X, et al. Impact of genetic polymorphisms in cytomegalovirus glycoprotein B on outcomes in solid-organ transplant recipients with cytomegalovirus disease. Clin Infect Dis. 2009;49(8):1160–6.
Tu W, Potena L, Stepick-Biek P, et al. T-cell immunity to subclinical cytomegalovirus infection reduces cardiac allograft disease. Circulation. 2006;114(15):1608–15.
Nickel P, Bold G, Presber F, et al. High levels of CMV-IE-1-specific memory T cells are associated with less alloimmunity and improved renal allograft function. Transpl Immunol. 2009;20(4):238–42.
Amir AL, D'Orsogna LJ, Roelen DL, et al. Allo-HLA reactivity of virus-specific memory T cells is common. Blood. 2010;115(15):3146–57.
Ouwehand AJ, Balk AH, Baan CC, Vaessen LM, Jutte NH, Bos E, Claas FH, Weimar W, et al. Cytomegalovirus infection and allospecific cytotoxic activity of graft-infiltrating cells after heart transplantation. J Med Virol. 1994;42(2):175–81.
Ishibashi K, Tokumoto T, Shirakawa H, et al. Lack of antibodies against the antigen domain 2 epitope of cytomegalovirus (CMV) glycoprotein B is associated with CMV disease after renal transplantation in recipients having the same glycoprotein H serotypes as their donors. Transpl Infect Dis. 2011;13(3):318–23.
Acknowledgments
We thank members of the Department of Immunology, cardiologists and nursing staff of the Cardiology and Cardiovascular Surgery for their continued support and to the patients for their generous collaboration. N del Pozo performed ELISA tests for specific anti-varicella antibodies. L Valor performed the substudy of lymphoproliferative responses to PHA. M Jaramillo performed ELISA tests for serum IFNg concentration J Romero contributed to the graphic design and Thomas O’Boyle proofread the manuscript.
Funding Sources
This study was supported by grants from the Instituto de Salud Carlos III, Fondo de Investigacion Sanitaria (projects numbers 05/0839, 08/1430), and Grifols International, Barcelona, Spain awarded to Javier Carbone. Elizabeth Sarmiento had a grant from the ERA-EDTA Fellowship Programme administrated by the European Molecular Biology Organization.
Conflict of interest statement
None of the authors have a financial or proprietary interest in the subject matter or materials discussed, including, but not limited to, employment, consultancies, stock ownership, honoraria, and paid expert testimony.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Carbone, J., Lanio, N., Gallego, A. et al. Simultaneous Monitoring of Cytomegalovirus-Specific Antibody and T-cell levels in Seropositive Heart Transplant Recipients. J Clin Immunol 32, 809–819 (2012). https://doi.org/10.1007/s10875-012-9670-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10875-012-9670-7