Abstract
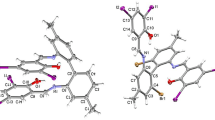
The complexes, Ba (HQS) (H2O)4 (HQS = 8-hydroxyquinoline-5-sulfonic acid) (1) and Ag (HIQS) (H2O) (Ferron = 7-iodo-8-hydroxyquinoline-5-sulfonic acid) (2) have been synthesized and characterized by X-ray diffraction analysis and spectroscopic studies. In compound 1, Ba2+ ion has a nine-coordinate monocapped antiprismatic geometry. In compound 2, Ag+ has distorted tetrahedral coordination and Ag···I interactions generate the supramolecular architectures. The complexes have been characterized by FT-IR and UV–Visible measurements. In both the structures, the inversion-related organic ligands are stacked over one another leading to three-dimensional networks.
Graphical Abstract








Similar content being viewed by others
References
Barbara RS, Price CP, Jayasankar A, Matzger AJ, Hornedo NR (2004) Adv Drug Deliv Rev 56:241–274
Dunitz JD, Bernstein J (1995) Acc Chem Res 28:193–200
Hughes LJ, Stezowski JJ, Hughes RE (1979) J Am Chem Soc 101:7655–7657
Nangia A, Desiraju GR (1999) Chem Commun 605–606
Davies JA, Hockensmith CA, Kukushkin VY, Kukushkin YN (1996) Synthetic coordination chemistry: principles and practice. World Scientific, London
Kaschak DM, Johnson SA, Hooks DE, Kim HN, Ward MD, Mallouk TE (1998) J Am Chem Soc 120:10887–10894
Nakato T, Furumi Y, Okuhara T (1998) Chem Lett 611–612
Solin SA (1997) Annu Rev Mater Sci 27:89–94
Cote AP, Shimizu GKH (2003) Coord Chem Rev 245:49–64
Cai J (2004) Coord Chem Rev 248:1061–1083
Onoda A, Yamado Y, Doi M, Ueyama N (2001) Inorg Chem 40:516–521
Papaefstathiou GS, MacGillivray LR (2002) Angew Chem Int Ed 41:2070–2073
Eddoudi M, Moler DB, Li H, Chen B, Reineke TM, O’Keeffe M, Yaghi OM (2001) Acc Chem Res 34:319–330
Shimizu GKH, Enright GD, Ratcliffe CI, Rego GS, Reid JL, Ripmeester JA (1998) Chem Mater 10:3282–3283
Bambury RE (1979) Burger’s Medicinal Chemistry, Part II. In: Wolff ME (ed) Wiley, New York
Batten SR, Robson R (1998) Angew Chem Int Ed Engl 37:1460–1494
Wu H, Dong XW, Ma JF (2006) Acta Crystallogr Sec E 62:m385–m387
Sheldrick GM (1997) SHELXS-97 and SHELXL-97. University of Gottingen, Germany
Lin-Vien D, Colthup NB, Fateley WG, Grasselli JC (1991) The handbook of infrared and raman characteristic frequencies of organic molecules. Academic Press, San Diego
Abd El Wahed MG, El Manakhly KA, El Kososy N (1995) Mater Chem Phys 41:117–122
Gonzaler-Baro AC, Baran EJ (2001) J Braz Chem Soc 12(2):208–214
Cai J, Chen CH, Liao CZ, Feng XI, Chen XM (2001) Acta Crystallogr Sec B 57:520–530
Dalrymple SA, Shimizu GKH (2001) Chem Eur J 8:3010–3015
Muthiah PT, Francis S, Bocelli G, Cantoni A (2003) Acta Crystallogr Sec E 59:m1164–m1167
Francis S, Muthiah PT, Bocelli G, Cantoni A (2003) Acta Crystallogr Sec E 59:m87–m90
Francis S, Muthiah PT, Butcher RJ (2004) Acta Crystallogr Sec E 60:m62–m64
Liu HY, Wu H, Ma JF (2006) Acta Crystallogr Sec E 62:m1036–m1037
Zhang F, Li YZ, Gao X, Chen HL, Liu QT, Odani A, Yamauchi O (2004) Chem Lett 33:556–557
Raj SB, Muthiah PT, Rychlewska U, Warzajtis B, Bocelli G, Olla R (2003) Acta Crystallogr Sec E 59:m46–m49
Balasubramani K, Muthiah PT, Bocelli G, Cantoni A (2005) J Coord Chem 58:1689–1694
Othman AH, Goh SC, Fun HK, Sivakumar K (1996) Acta Crystallogr Sec E 52:2760–2763
Smith G, Cloutt BA, Lynch DE, Byriel KA, Kennard CHL (1998) Inorg Chem 37:3236–3242
Ino I, Wu LP, Munakata M, Maekawa M, Suenaga Y, Kuroda-Sowa T, Kitamori Y (2001) Inorg Chem 39:2146–2151
Hunter CA (1994) Chem Soc Rev 23:101–109
Muthiah PT, Murugesan P (2006) J Coord Chem 591:1167–1172
Raj SB, Muthiah PT, Bocelli G, Olla R (2002) Acta Crystallogr Sec E 58:m513–m516
Francis S, Muthiah PT, Bocelli G, Cantoni A (2003) Acta Crystallogr Sec E 59:m1154–m1156
Raj SB, Muthiah PT, Bocelli G, Righi L (2001) Acta Crystallogr Sec E 57:m591–m594
Petit S, Coquerel G, Perez G (1993) New J Chem 17:187–192
Rao HY, Tao J, Ng SW (2003) Acta Crystallogr Sec E 59:m859–m860
Francis S, Muthiah PT, Rychlewska U, Warzajtis B (2004) Acta Crystallogr Sec E 60:m137–m139
Francis S, Muthiah PT, Rychlewska U, Warzajtis B (2003) Acta Crystallogr Sec E 59:m1160–m1163
Balasubramani K, Francis S, Muthiah PT, Bocelli G, Cantoni A (2004) Acta Crystallogr Sec E 60:m1576–m1579
Francis S, Muthiah PT, Bocelli G, Cantoni A (2003) Acta Crystallogr Sec E 59:m1157–m1159
Acknowledgment
S. F. thanks the Council of Scientific and Industrial Research, New Delhi, India for the award of Senior Research Fellowship [Ref. No. 9/475(123) 2004- EMRI]. M. H. thanks the Council of Scientific and Industrial Research, New Delhi, India for the award of Senior Research Fellowship [Ref. No. 9/475(123) 2004- EMRI].
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Balasubramani, K., Hemamalini, M., Francis, S. et al. Supramolecular Organization in Tetra Aqua (μ-8-Hydroxyquinoline-5-sulfonate) Barium (II) and Ag···I Interactions in a Pseudopolymorphic Form of (7-Iodo-8-hydroxyquinoline-5-sulfonate) Silver (I) Monohydrate. J Chem Crystallogr 40, 316–322 (2010). https://doi.org/10.1007/s10870-009-9653-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10870-009-9653-6