Abstract
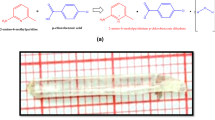
The crystal growth of acetanilide tetrachloromercurate(II), an inorganic–organic hybrid derivative has been achieved by solution growth through slow cooling method. The X-ray diffraction structural analysis of the hybrid material results that the compound exist in orthorhombic space group P212121 with lattice parameters; a = 13.111(2) Ǻ, b = 11.311(2) Ǻ, c = 8.355(6) Ǻ, α = β = γ = 90° and unit cell volume = 1436.24 Ǻ3. The fourier transform infrared spectroscopy profile shows that the C–C and C–N stretching modes of acetanilide ring and the observed spectra falls in mid-infrared range υ(526–2850) cm−1. The field emission scanning electron microscope image confirms that the hybrid material has a prismatic shape with an average granular size of ~25 nm. The energy dispersive X-ray spectroscopy analyzes the elemental proportions of the hybrid materials. Transmission electron microscopy image shows the narrow distribution of nano-spatial agglomeration of secondary interactions in inorganic–organic particles. The optical band gap (Eg = 3.75 eV) as calculated by linear fit profile of Tauc plot for allowed transition predicts that the hybrid material has potential applications in solar cells, electronic and opto-electronic devices.




Similar content being viewed by others
References
K.G. Sharp, Adv. Mater. 10, 1243–1248 (1998)
G. Kickelbick (ed.), Hybrid Materials: Synthesis, Characterization and Applications (Wiley-VCH, Weinheim, 2005)
Z. Linda, M.D. Hager, S.S. Ulrich, J.H. Matthew, M. Schmitt, J. Popp, B. Dietzek, Mater. Today 17, 57–69 (2014)
A.S. Mahadevi, G.N. Sastry, Chem. Rev. 116, 2775–2825 (2016)
T.K. Dobravc, A. Meden, F. Perdih, New J. Chem. 39, 4265–4277 (2015)
S.I. Stupp, C.P. Liam, Chem. Mater. 26, 507–518 (2013)
C. Sanchez, B. Julian, P. Belleville, M. Popall, J. Mater. Chem. 15, 3559–3592 (2005)
P.G. Romero, C. Sanchez (eds.), Functional Hybrid Materials (Wiley-VCH, Weinheim, 2004)
G. Kickelbick (ed.), Introduction to Hybrid Materials Hybrid Materials: Synthesis, Characterization, and Applications (Wiley-VCH, Weinheim, 2007)
S. Bikram, T. Atul, K. Mukesh, J. Dinesh, S.K. Verma, Ind. J. Appl. Res. 5, 9 (2015)
K. Mukesh, S.K. Verma, S. Bikram, T. Atul, K. Ajit, J. Dinesh, Chem. Sci. Tran. 4, 2 (2015)
T. Roisnel, J.R. Carvajal, Proceedings of the Seventh European Powder Diffraction Conference EPDIC 7, ed. by R. Delhez, E. J. Mittenmeijer (Trans Tech Publications, Uetikon-Zuerich, 2001), pp. 118–123
R. Carvajal, J. Phys. B 192, 55 (1993)
S. Kawazoe, K. Sakamoto, Y. Awamura, T. Maruyama, T. Suzuki, K. Onda, T. Takasu, Beta-form crystal of acetanilide derivative, European Patent EP1932838A2, 18 June 2008
L.X. Yang, Y.J. Zhu, H. Tong, Z.H. Liang, L. Li, L. Zhang, J. Solid State Chem. 180, 2095–2101 (2007)
S. Bag, C.R. Raj, J. Mater. Chem. A 2, 17848–17856 (2014)
R.S. Dey, S. Hajra, R.K. Sahu, C.R. Raj, M.K. Panigrahi, Chem. Commun. 48, 1787–1789 (2012)
C.A. Schneider, W.S. Rasband, K.W. Eliceiri, Nat. Methods 9, 671 (2012)
K. Wooseok, J. Li, J. Am. Chem. Soc. 130, 8114 (2008)
M.J. Schulz, A.D. Kelkar, M.J. Sundaresan (eds.), Nanoengineering of Structural, Functinal and Smart Materials (Taylor & Francis, Boca Raton, 2005)
F. Bourguiba, A. Dhahri, T. Tahri, K. Taibi, J. Dhabri, E.K. Hill, Bull. Mater. Sci. 39, 1765–1776 (2016)
Acknowledgement
The corresponding author (Dinesh Jasrotia) is thankful to University Grants Commission (UGC) for research funding under UGC-Major Research Project No. 42-777 of 2013.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Singh, B., Thakur, A., Kumar, M. et al. Structural and optical properties of inorganic–organic hybrid material of acetanilide tetrachloromercurate(II). J Mater Sci: Mater Electron 28, 10007–10011 (2017). https://doi.org/10.1007/s10854-017-6758-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-017-6758-0