Abstract
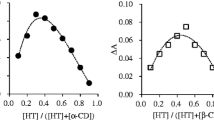
Inclusion complexes of fluconazole (HFlu) with β-cyclodextrin (β-CD) and hydroxypropyl- β-cyclodextrin (HP-β-CD) had been prepared by the coprecipitation method. The 1:1 stoichiometry of complexation for both inclusion complexes was achieved in the binary system by phase solubility and fluorimetric study. The inclusion complexes were also characterized by DSC-TGA analyses and ESI–MS spectra. The molecular assemblies were proposed that a triazolyl ring of HFlu was inserted into the narrower end of the CD cavity and the 2, 4-difluorophenyl ring into the wider end to generate 1-D chain structure on the basis of physical characters. The comparative work indicated that the higher stability was obviously observed for HFlu-HP-β-CD than for HFlu-β-CD, and provided a crucial clue to find out the dependency: cavity size, van der Waals and hydrophobic interactions as well as hydrogen bonding.









Similar content being viewed by others
References
Rekharsky, M.V., Inoue, Y.: Complexation thermodynamics of cyclodextrins. Chem. Rev. 98, 1875–1917 (1998)
Liu, Y., Han, B.H., Zhang, H.Y.: Spectroscopic studies on molecular recognition of modified cyclodextrins. Curr. Org. Chem. 8, 35–46 (2004)
Brewster, M.E., Loftsson, T.: Cyclodextrins as pharmaceutical solubilizers. Adv. Drug Deliver. Rev. 59, 645–666 (2007)
Uekama, K.: Design and evaluation of cyclodextrin-based drug formulation. Chem. Pharm. Bull. 52, 900–915 (2004)
Loftsson, T., Duchêne, D.: Cyclodextrins and their pharmaceutical applications. Int. J. Pharm. 329, 1–11 (2007)
Maestrelli, F., Mura, P., Casini, A., Mincione, F., Scozzafava, A., Supuran, C.T.: Cyclodextrin complexes of sulfonamide carbonic anhydrase inhibitors as long-lasting topically acting anti-glaucoma agents. J. Pharm. Sci. 91, 2211–2219 (2002)
Carrier, R.L., Miller, L.A., Ahmed, I.: The utility of cyclodextrins for enhancing oral bioavailability. J. Control. Rel. 123, 78–99 (2007)
Uekama, K., Hirayama, F., Irie, T.: Cyclodextrin drug carrier systems. Chem. Rev. 98, 2045–2076 (1998)
Davis, M.E., Brewster, M.E.: Cyclodextrin-based pharmaceutics: past, present and future. Nat. Rev. Drug Discov. 3, 1023–1035 (2004)
Szente, L., Szejtli, J.: Cyclodextrins as food ingredients. Trends Food Sci. Tech. 15, 137–142 (2004)
Centini, M., Maggiore, M., Casolaro, M., Andreassi, M., Facino, R.M., Anselmi, C.: Cyclodextrins as cosmetic delivery systems. J. Incl. Phenom. Macrocycl. Chem. 57, 109–112 (2007)
Peeters, J., Neeskens, P., Tollenaere, J.P., Van Remoortere, P., Brewster, M.E.: Characterization of the interaction of 2-hydroxypropyl-β-cyclodextrin with itraconazole at pH 2, 4, and 7. J. Pharm. Sci. 91, 1414–1422 (2002)
Song, L.X., Bai, L., Xu, X.M., He, J., Pan, S.Z.: Inclusion complexation, encapsulation interaction and inclusion number in cyclodextrin chemistry. Coord. Chem. Rev. 253, 1276–1284 (2009)
Jadhav, P., Petkar, B., Pore, Y., Kulkarni, A., Burade, K.: Physicochemical and molecular modeling studies of cefixime-l-arginine-cyclodextrin ternary inclusion compounds. Carbohyd. Polym. 98, 1317–1325 (2013)
Loftsson, T., Hreinsdóttir, D., Másson, M.: The complexation efficiency. J. Incl. Phenom. Macrocycl. Chem. 57, 545–552 (2007)
Messner, M., Kurkov, S.V., Jansook, P., Loftsson, T.: Self-assembled cyclodextrin aggregates and nanoparticles. Int. J. Pharm. 387, 199–208 (2010)
Odds, F.C., Cheesman, S.L., Abbott, A.B.: Antifungal effects of fluconazole (UK 49858), a new triazole antifungal, in vitro. J. Antimicrob. Chemoth. 18, 473–478 (1986)
Ling, Y., Zhang, L., Li, J., Fan, S.S., Du, M.: Thiocyanate-induced conformational transformation of a flexible fluconazole ligand in Cd(II) coordination polymers. CrystEngComm 12, 604–611 (2010)
Upadhyay, S.K., Kumar, G.: NMR and molecular modeling studies on the interaction of fluconazole with β-cyclodextrin. Chem. Central J. 3, 9 (2009)
Yurtdas, G., Demirel, M., Genc, L.: Inclusion complexes of fluconazole with β-cyclodextrin: physicochemical characterization and in vitro evaluation of its formulation. J. Incl. Phenom. Macrocycl. Chem. 70, 429–435 (2011)
Higuchi, T., Connors, K.: Phase-solubility techniques. In: Reilly, C.N. (ed.) Advances in Analytical Chemistry and Instrumentation, pp. 117–212. Wiley-Interscience, New York (1965)
Liu, Y., Song, Y., Chen, Y., Yang, Z.X., Ding, F.: Spectrophotometric study on the controlling factor of molecular selective binding of dyes by bridged bis(beta-cyclodextrin)s with diseleno-bis(benzoyl) linkers. J. Phys. Chem. B 109, 10717–10726 (2005)
Djedaini, F., Lin, S.Z., Perly, B., Wouessidjewe, D.: High-field nuclear magnetic resonance techniques for the investigation of a β-cyclodextrin: indomethacin inclusion complex. J. Pharm. Sci. 79, 643–646 (1990)
Orgoványi, J., Pöppl, L.: H-Otta, K., Lovas, G.A.; Thermoanalytical method for studying the guest content in cyclodextrin inclusion complexes. J. Therm. Anal. Calorim. 81, 261–266 (2005)
Inkmann, E., Holzgrabe, U.: H-1 and C-13 nuclear magnetic resonance studies of the sites of protonation in itraconazole and fluconazole. J. Pharm. Biomed. Anal. 20, 297–307 (1999)
Schneider, H.J., Hacket, F., Rudiger, V., Ikeda, H.: NMR studies of cyclodextrins and cyclodextrin complexes. Chem. Rev. 98, 1755–1785 (1998)
Qian, L., Guan, Y., Xiao, H.: Preparation and charcterization of inclusion complexes of a cationic β-cyclodextrin polymer with butylparaben or triclosan. Int. J. Pharm. 357, 244–251 (2008)
Zoppetti, G., Puppini, N., Pizzutti, M., Fini, A., Giovani, T., Comini, S.: Inclusion complex characterization between progesterone and hydroxypropyl β-cyclodextrin in aqueous solution by NMR study. J. Incl. Phenom. Macrocycl. Chem. 57, 283–288 (2007)
Cowins, J., Abimbola, O., Ananaba, G., Wang, X.Q., Khan, I.: Preparation and characterization of β-sitosterol/β-cyclodextrin crystalline inclusion complexes. J. Incl. Phenom. Macrocycl. Chem. 83, 141–148 (2015)
Ali, S.M., Shamim, S.: Analysis of computational models of β-cyclodextrin complexes: structural studies of morniflumate hydrochloride and β-cyclodextrin complex in aqueous solution by quantitative ROESY analysis. J. Incl. Phenom. Macrocycl. Chem. 83, 19–26 (2015)
Al-Marzouqi, A.H., Elwy, H.M., Shehadi, I., Adem, A.: Physicochemical properties of antifungal drug-cyclodextrin complexes prepared by supercritical carbon dioxide and by conventional techniques. J. Pharm. Biomed. Anal. 49, 227–233 (2009)
Acknowledgments
This work was supported by the Foundation of Guangdong Provincial Department of Science and Technology (2012A080800002 and S2013010012018).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, J., Zhang, S., Zhou, Y. et al. Inclusion complexes of fluconazole with β-cyclodextrin and 2-hydroxypropyl-β-cyclodextrin in aqueous solution: preparation, characterization and a structural insight. J Incl Phenom Macrocycl Chem 84, 209–217 (2016). https://doi.org/10.1007/s10847-016-0598-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-016-0598-z