Abstract
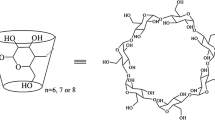
Coumarin-appended cyclophanes bearing positively or negatively charged side chains were synthesized as a water-soluble host (1a or 1b, respectively). Host 1a and 1b showed fluorescence bands with fluorescence maxima at 404 nm originated from coumarin moiety. As a host for guest molecules by using macrocyclic cavity, cationic host 1a binds anionic guests such as 6-p-toluidinonaphthalene-2-sulfonate (TNS), 6-anilinonaphthalene-2-sulfonate (2,6-ANS), and 8-anilinonaphthalene-1-sulfonate (1,8-ANS) more strongly than anionic host 1b, reflecting intermolecular electrostatic interactions. In addition, both host 1a and 1b showed protein surface recognition and fluorescence response toward myoglobin, a small and globular protein. The fluorescence intensity originating from the hosts decreased upon the addition of myoglobin, reflecting the formation of 1a- and 1b-myoglobin complexes. On the other hand, such fluorescence response of 1a and 1b was almost negligible for other proteins such as egg white albumin, bovine serum albumin, human albumin, concanavaline A, fibrinogen, γ-globlin, peanut agglutinin, trypsin, and lysozyme.






Similar content being viewed by others
References
Rebek Jr, J.: Host–guest chemistry of calixarene capsules. Chem. Commun. 8, 637–643 (2000)
Biros, S.M., Rebek Jr, J.: Structure and binding properties of water-soluble cavitands and capsules. Chem. Soc. Rev. 36, 93–104 (2007)
Berryman, O.B., Sather, A.C., Lledó, A., Rebek Jr, J.: Switchable catalysis with a light-responsive cavitand. Angew. Chem. Int. Ed. 50, 9400–9403 (2011)
Vögtle, F., Seel, C., Windscheif, P.-M.: In: Vögtle, F (ed), Comprehensive supramolecular chemistry, Vol. 2, Chap. 7. Pergamon, Oxford, pp. 211–265 (1996)
Seward, E., Hopkins, R.B., Sauerer, W., Tam, S.-W., Diederich, F.: Redox-dependent binding ability of a flavin cyclophane in aqueous solution: hydrophobic stacking versus cavity-inclusion complexation. J. Am. Chem. Soc. 112, 1783–1790 (1990)
Ariga, K., Nakanishi, T., Terasaka, Y., Tsuji, H., Sakai, D., Kikuchi, J.: Piezoluminescence at the air–water interface through dynamic molecular recognition driven by lateral pressure application. Langmuir 21, 976–981 (2005)
Hamuro, Y., Calama, M.C., Park, H.S., Hamilton, A.D.: A Calixarene with four peptide loops: an antibody mimic for recognition of protein surfaces. Angew. Chem. Int. Ed. 36, 2680–2683 (1997)
Lin, Q., Park, H.S., Hamuro, Y., Lee, C.S., Hamilton, A.D.: Protein surface recognition by synthetic agents: design and structural requirements of a family of artificial receptors that bind to cytochrome C. Biopolymers 47, 285–297 (1998)
Chinta, J.P., Acharya, A., Kumar, A., Rao, C.P.: Spectroscopy and microscopy studies of the recognition of amino acids and aggregation of proteins by Zn(II) complex of lower rim naphthylidene conjugate of calix[4]arene. J. Phys. Chem. B 113, 12075–12083 (2009)
Geraci, C., Consoli, G.M., Granata, G., Galante, E., Palmigiano, A., Pappalardo, M., Di Puma, S.D., Spadaro, A.: First self-adjuvant multicomponent potential vaccine candidates by tethering of four or Eight MUC1 antigenic immunodominant PDTRP units on a calixarene platform: synthesis and biological evaluation. Bioconjugate Chem. 24, 1710–1720 (2013)
Marra, A., Moni, L., Pazzi, D., Corallini, A., Bridi, D., Dondoni, A.: Synthesis of sialoclusters appended to calix[4]arene platforms via multiple azide-alkyne cycloaddition. New inhibitors of hemagglutination and cytopathic effect mediated by BK and influenza A viruses. Org. Biomol. Chem. 6, 1396–1409 (2008)
Aleandri, S., Casnati, A., Fantuzzi, L., Mancini, G., Rispoli, G., Sansone, F.: Incorporation of a calixarene-based glucose functionalised bolaamphiphile into lipid bilayers for multivalent lectin recognition. Org. Biomol. Chem. 11, 4811–4817 (2013)
Chinta, P., Rao, C.P.: Triazole linked lower rim glycosyl appended 1,3-calix[4]arene conjugates: synthesis, characterization, and their interaction with jacalin. Carbohydr. Res. 369, 58–62 (2013)
Hayashida, O., Uchiyama, M.: Multivalent macrocyclic hosts: histone surface recognition, Guest binding, and delivery by cyclophane-based resorcinarene oligomers. J. Org. Chem. 72, 610–616 (2007)
Hayashida, O., Ogawa, N., Uchiyama, M.: Surface recognition and fluorescence sensing of histone by dansyl-appended cyclophane-based resorcinarene trimer. J. Am. Chem. Soc. 129, 13698–13705 (2007)
Gordo, S., Martos, V., Santos, E., Menéndez, M., Bo, C., Giralt, E., de Mendoza, J.: Stability and structural recovery of the tetramerization domain of p53-R337H mutant induced by a designed templating ligand. Proc. Natl. Acad. Sci. USA. 105, 16426–16431 (2008)
McGovern, R.E., Fernandes, H., Khan, A.R., Power, N.P., Crowley, P.B.: Protein camouflage in cytochrome c–calixarene complexes. Nat. Chem. 4, 527–533 (2012)
Patra, D., Ozdemir, F., Miranda, O.R., Samanta, B., Sanyal, A., Rotello, V.M.: Formation and size tuning of colloidal microcapsules via host–guest molecular recognition at the liquid–liquid interface. Langmuir 25, 13852–13854 (2009)
Bindman, N.A., van der Donk, W.A.: A General method for fluorescent labeling of the N-termini of lanthipeptides and its application to visualize their cellular localization. J. Am. Chem. Soc. 135, 10362–10371 (2013)
Lohar, S., Safin, D.A., Sengupta, A., Chattopadhyay, A., Matalobos, J.S., Babashkina, M.G., Robeyns, K., Mitoraj, M.P., Kubisiak, P., Garcia, Y., Das, D.: Ratiometric sensing of lysine through the formation of the pyrene excimer: experimental and computational studies. Chem. Commun. 51, 8536–8539 (2015)
Kumar, A., Ghosh, M.K., Choi, C.H., Kim, H.S.: Selective fluorescence sensing of salicylic acids using a simple pyrenesulfonamide receptor. RSC Adv. 5, 23613–23621 (2015)
Chen, W., Fang, Q., Yang, D., Zhang, H., Song, X., Foley, J.: Selective, highly sensitive fluorescent probe for the detection of sulfur dioxide derivatives in aqueous and biological environments. Anal. Chem. 87, 609–616 (2015)
Song, H.Y., Ngai, M.H., Song, Z.Y., MacAry, P.Y., Hoble, J., Lear, M.J.: Practical synthesis of maleimides and coumarin-linked probes for protein and antibody labelling via reduction of native disulfides. Org. Biomol. Chem. 7, 3400–3406 (2009)
Elbert, W., Breitenbach, S., Neftel, A., Hahn, J.: 4-Methyl-7-methoxycoumarin as a fluorescent label for high-performance liquid chromatographic analysis of dicarboxylic acids. J. Chromatogr. A 328, 111–120 (1985)
Chowdhury, P.K., Halder, M., Sanders, L., Arnold, R.A., Liu, Y., Armstrong, D.W., Kundu, S., Hargrove, M.S., Song, X., Petrich, J.W.: The complex of apomyoglobin with the fluorescent dye coumarin 153. Photochem. Photobiol. 79, 440–446 (2004)
Odashima, K., Itai, A., Iitaka, Y., Koga, K.: Biomimetic studies using artificial systems. 3. Design, synthesis, and inclusion complex forming ability of a novel water-soluble paracyclophane possessing diphenylmethane skeletons. J. Org. Chem. 50, 4478–4484 (1985)
Hayashida, O., Kaku, Y.: Synthesis of dabsyl-appended cyclophanes and their heterodimer formation with pyrene-appended cyclophanes. J. Org. Chem. 78, 10437–10442 (2013)
Pashkova, A., Chen, H.S., Rejtar, T., Zang, X., Giese, R., Andreev, V., Moskovets, E., Karger, B.L.: Coumarin tags for analysis of peptides by MALDI-TOF MS and MS/MS. 2. alexa fluor 350 tag for increased peptide and protein identification by LC-MALDI-TOF/TOF MS. Anal. Chem. 77, 2085–2096 (2005)
Slavik, J.: 1-Anilino-8-naphthalene sulfonate anion-protein binding depends primarily on ion pair formation. Biochem. Biophys. Acta 694, 1–25 (1982)
Benesi, H.A., Hildebrand, J.H.: A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J. Am. Chem. Soc. 71, 2703–2707 (1949)
Nesbitt, C.A., Yeung, K.K.C.: In-capillary enrichment, proteolysis and separation using capillary electrophoresis with discontinuous buffers: application on proteins with moderately acidic and basic isoelectric points. Analyst 134, 65–71 (2009)
Acknowledgments
The present work is partially supported by Grant-in-Aid (No. 24550166) from the Ministry of Education, Culture, Science, Sports and Technology of Japan.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hayashida, O., Harada, Y. & Kojima, M. Synthesis of coumarin-appended cyclophanes and evaluation of their complexation with myoglobin. J Incl Phenom Macrocycl Chem 83, 111–117 (2015). https://doi.org/10.1007/s10847-015-0546-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-015-0546-3