Abstract
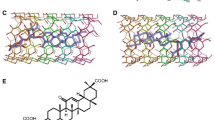
Complexation of morniflumate hydrochloride (MOR) with β-cyclodextrin (β-CD) in aqueous solution was studied. Structure of the MOR-β-CD inclusion complex was obtained by a combination of 1H NMR and molecular modeling studies. Computational models obtained by molecular mechanics and molecular dynamics were analyzed for their atom-accuracy. ROESY crosspeak intensities calculated from intermolecular interproton distances were compared with experimental intensities. The results demonstrate that comparison of calculated and experimental intensities is a good criterion to determine accuracy of the structure of CD complexes. Moreover, it is also demonstrated that ROESY experiment, with longer mixing time when initial rate approximation condition is not valid, can be used for quantitative purpose if intensity ratios, instead of absolute intensities, are used.








Similar content being viewed by others
References
Del Valle, E.M.M.: Cyclodextrins and their uses: a review. Process Biochem. 39, 1033–1046 (2004)
Schneider, H.-J.: Mechanisms of molecular recognition: investigations of organic host-guest complexes. Angew. Chem. Int. Ed. Engl. 30, 1417–1436 (1991)
Ariga, K., Kunitake, T.: Supramolecular Chemistry—Fundamentals and Applications Advanced Textbook. Springer, New York (2006)
Pérez-Cruz, F., Aguilera-Venegas, B., Lapier, M., Sobarzo-Sánchez, E., Villares, E.U., Olea-Azar, C.: Host-guest interaction between new nitrooxoisoaporphine and β-cyclodextrins: synthesis, electrochemical, electron spin resonance and molecular modeling studies. Spectrochim. Acta Mol. Biomol. Spectros. 102, 226–234 (2013)
Schneider, H.-J., Hacket, F., Rudiger, V., Ikeda, H.: NMR studies of cyclodextrins and cyclodextrin complexes. Chem. Rev. 98, 1755–1785 (1998)
Szejtli, J.: Past, present and future of cyclodextrin research. Pure Appl. Chem. 76, 1825–1845 (2004)
Pessine, F.B.T., Calderini, A., Alexandrino, G.L.: Review: cyclodextrin inclusion complexes probed by NMR techniques. In: Kim, D.H. (ed.) Magnetic Resonance Spectroscopy, pp. 237–264. InTec, Rijeka (2012)
Karpkird, T., Wanichweacharungruang, S.: Synthesis and photostability of methoxycinnamic acid modified cyclodextrins. J. Photochem. Photobiol., A 212, 56–61 (2010)
Dodziuk, H.: Cyclodextrins and Their Complexes. Chemistry, Analytical Methods, Applications. Wiley-VCH, London (2006)
Butts, C.P., Jones, C.R., Towers, E.C., Flynn, J.L., Appleby, L., Barron, N.J.: Interproton distance determinations by NOE—surprising accuracy and precision in a rigid organic molecule. Org. Biomol. Chem. 9, 177–184 (2011)
Ali, S.M., Shamim, S.: Complexation of (RS)-benzhexol with β-cyclodextrin: structure elucidation diastereomeric complexes by use of quantitative 1H–1H ROESY and computational methods. Monatsh. Chem. 146, 283–290 (2015)
Mura, P.: Analytical techniques for characterization of cyclodextrin complexes in aqueous solution: a review. J. Pharm. Biomed. Anal. 101, 238–250 (2014)
Demarco, P.V., Thakkar, A.L.: Cyclohepta-amylose inclusion complexes. A proton magnetic resonance study. J. Chem. Soc., Chem. Commun. 1, 2–4 (1970)
Leyva, E., Moctezuma, E., Strouse, J., García-Garibay, M.A.: Spectrometric and 2D NMR studies on the complexation of chlorophenols with cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 39, 41–46 (2001)
Benesi, H.A., Hildebrand, J.H.: A spectrophotometric investigation of the interaction of iodine with aromatic hydrocarbons. J. Am. Chem. Soc. 71, 2703–2707 (1949)
Scott, R.L.: Some comments on the Benesi-Hildebrand equation. Recl. Trav. Chim. Pays-Bas 75, 787–789 (1956)
Neuhaus, D., Williamson, M.P.: The Nuclear Overhauser Effect in Structural and Conformational Analysis. Wiley-VCH, London (1989)
Allinger, N.L.: Conformational analysis 130. MM2. A hydrocarbon force field utilizing V1 and V2 torsional terms. J. Am. Chem. Soc. 99, 8127–8134 (1977)
Zabel, V., Saenger, W., Mason, S.A.: Neutron diffraction study of the hydrogen bonding in β-cyclodextrin undecahydrate at 120 K: from dynamic flip-flops to static homodromic chains. J. Am. Chem. Soc. 108, 3664–3673 (1986)
Jingye, L., Deyue, Y., Qun, C.: Preparation and characterization of the crystalline inclusion complexes between cyclodextrins and poly(1,3-dioxolane). Sci. China, Ser. B 45, 73–83 (2002)
Kohler, J.E.H., Grczelschak-Mick, N.: The β-cyclodextrin/benzene complex and its hydrogen bond—a theoretical study using molecular dynamics, quantum mechanics and COSMO-RS. Beilstein J. Org. Chem. 9, 118–134 (2013)
Andersen, N.H., Eaton, H.L., Lai, X.: Quantitative small molecule NOESY. A practical guide for derivation of cross-relaxation rates and internuclear distances. Magn. Reson. Chem. 27, 515–528 (1989)
Jones, C.R., Butts, C.P., Harvey, J.N.: Accuracy in determining interproton distances using nuclear overhauser effect data from a flexible molecule. Beilstein J. Org. Chem. 7, 145–150 (2011)
Macura, S., Farmer II, B.T., Brown, L.R.: An improved method for the determination of cross-relaxation rates from NOE Data. J. Magn. Reson. 70, 493–499 (1986)
Acknowledgments
Morniflumate hydrochloride and β-cyclodextrin were very kindly provided by Amoli Organics Ltd, India, and Geertrui Haest, Cerestar Cargill, Belgium, respectively. Shazia Shamim is thankful to UGC, Government of India, for the financial assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ali, S.M., Shamim, S. Analysis of computational models of β-cyclodextrin complexes: structural studies of morniflumate hydrochloride and β-cyclodextrin complex in aqueous solution by quantitative ROESY analysis. J Incl Phenom Macrocycl Chem 83, 19–26 (2015). https://doi.org/10.1007/s10847-015-0534-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10847-015-0534-7