Abstract
Purpose
Magnetic resonance imaging (MRI) with late gadolinium enhancement is commonly performed in patients with non-ischemic LV ventricular tachycardia/ventricular premature depolarizations (non-ischemic LV-VT/VPDs) to define VT substrate prior to catheter ablation. We investigated the prevalence of abnormal voltage and VT localized to areas of the myocardium not reported to have late gadolinium enhancement (LGE) on routine pre-procedural MRI and sought to determine if quantitative MRI analysis could reduce this discordance.
Methods
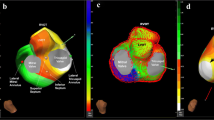
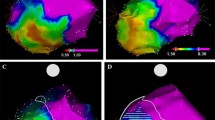
Patients with non-ischemic LV-VT/VPD who underwent LV endocardial mapping with VT/VPD ablation and either septal or free wall MRI-voltage discordance were studied. Electroanatomic maps were analyzed post-procedure for areas of electrogram-defined scar and VT localized to areas without reported LGE. Discordant segments were then analyzed offline using delayed signal intensity of >2 and >5 standard deviations above normal myocardium.
Results
Of 90 consecutive patients, 32 (36%) patients with septal (n = 16), free wall (n = 14) or both (n = 2) MRI-voltage + mismatch were identified. All discordant segments demonstrated unipolar voltage abnormalities with 12 patients (6 septal and 6 free wall) also showing low bipolar voltage but no LGE at signal intensity >5 standard deviations. Eleven patients (5 septum, 6 free wall) had VT localized to discordant areas. Ninety-three percent of patients in the septal group (26/48 segments) and 89% of patients in the free wall group (9/13 segments) had a concordant response established by using a signal intensity cutoff of >2 standard deviations.
Conclusions
MRI-voltage discordance was identified in 36% of patients with non-ischemic LV-VT/VPD who underwent VT ablation. In 12% of patients, VT was targeted in areas of abnormal voltage not suggested by routine qualitative MRI. Quantitative MRI analysis using a lower signal intensity threshold increased the sensitivity for detecting areas of clinically relevant VT substrate.



Similar content being viewed by others
References
Neilan TG, Farhad H, Mayrhofer T, Shah RV, Dodson JA, Abbasi SA, et al. Late gadolinium enhancement among survivors of sudden cardiac arrest. JACC Cardiovasc Imaging. 2015;8:414–23.
Poyhonen P, Kivisto S, Holmstrom M, Hanninen H. Quantifying late gadolinium enhancement on CMR provides additional prognostic information in early risk-stratification of nonischemic cardiomyopathy: a cohort study. BMC Cardiovasc Disord. 2014;14:110.
aus dem Siepen F, Buss SJ, Messroghli D, Andre F, Lossnitzer D, Seitz S, et al. T1 mapping in dilated cardiomyopathy with cardiac magnetic resonance: quantification of diffuse myocardial fibrosis and comparison with endomyocardial biopsy. Eur Heart J Cardiovasc Imaging. 2015;16:210–6.
Gulati A, Jabbour A, Ismail TF, Guha K, Khwaja J, Raza S, et al. Association of fibrosis with mortality and sudden cardiac death in patients with nonischemic dilated cardiomyopathy. JAMA. 2013;309:896–908.
Bogun FM, Desjardins B, Good E, Gupta S, Crawford T, Oral H, et al. Delayed-enhanced magnetic resonance imaging in nonischemic cardiomyopathy: utility for identifying the ventricular arrhythmia substrate. J Am Coll Cardiol. 2009;53:1138–45.
Wu KC, Weiss RG, Thiemann DR, Kitagawa K, Schmidt A, Dalal D, et al. Late gadolinium enhancement by cardiovascular magnetic resonance heralds an adverse prognosis in nonischemic cardiomyopathy. J Am Coll Cardiol. 2008;51:2414–21.
Betensky BP, Deyell MW, Tzou WS, Zado ES, Marchlinski FE. Sinus rhythm electrocardiogram identification of basal-lateral ischemic versus nonischemic substrate in patients with ventricular tachycardia. J Interv Card Electrophysiol. 2012;35:311–21. discussion 321.
Valles E, Bazan V, Marchlinski FE. ECG criteria to identify epicardial ventricular tachycardia in nonischemic cardiomyopathy. Circ Arrhythm Electrophysiol. 2010;3:63–71.
Marchlinski FE, Callans DJ, Gottlieb CD, Zado E. Linear ablation lesions for control of unmappable ventricular tachycardia in patients with ischemic and nonischemic cardiomyopathy. Circulation. 2000;101:1288–96.
Hutchinson MD, Gerstenfeld EP, Desjardins B, Bala R, Riley MP, Garcia FC, et al. Endocardial unipolar voltage mapping to detect epicardial ventricular tachycardia substrate in patients with nonischemic left ventricular cardiomyopathy. Circ Arrhythm Electrophysiol. 2011;4:49–55.
Cano O, Hutchinson M, Lin D, Garcia F, Zado E, Bala R, et al. Electroanatomic substrate and ablation outcome for suspected epicardial ventricular tachycardia in left ventricular nonischemic cardiomyopathy. J Am Coll Cardiol. 2009;54:799–808.
Haqqani HM, Tschabrunn CM, Tzou WS, Dixit S, Cooper JM, Riley MP, et al. Isolated septal substrate for ventricular tachycardia in nonischemic dilated cardiomyopathy: incidence, characterization, and implications. Heart Rhythm. 2011;8:1169–76.
Betensky BP, Kapa S, Desjardins B, Garcia FC, Callans DJ, Dixit S, et al. Characterization of trans-septal activation during septal pacing: criteria for identification of intramural ventricular tachycardia substrate in nonischemic cardiomyopathy. Circ Arrhythm Electrophysiol. 2013;6:1123–30.
Santangeli P, Hamilton-Craig C, Dello Russo A, Pieroni M, Casella M, Pelargonio G, et al. Imaging of scar in patients with ventricular arrhythmias of right ventricular origin: cardiac magnetic resonance versus electroanatomic mapping. J Cardiovasc Electrophysiol. 2011;22:1359–66.
Marra MP, Leoni L, Bauce B, Corbetti F, Zorzi A, Migliore F, et al. Imaging study of ventricular scar in arrhythmogenic right ventricular cardiomyopathy: comparison of 3D standard electroanatomical voltage mapping and contrast-enhanced cardiac magnetic resonance. Circ Arrhythm Electrophysiol. 2012;5:91–100.
Rashid S, Rapacchi S, Vaseghi M, Tung R, Shivkumar K, Finn JP, et al. Improved late gadolinium enhancement MR imaging for patients with implanted cardiac devices. Radiology. 2014;270:269–74.
Polin GM, Haqqani H, Tzou W, Hutchinson MD, Garcia FC, Callans DJ, et al. Endocardial unipolar voltage mapping to identify epicardial substrate in arrhythmogenic right ventricular cardiomyopathy/dysplasia. Heart Rhythm. 2011;8:76–83.
Spears DA, Suszko AM, Dalvi R, Crean AM, Ivanov J, Nanthakumar K, et al. Relationship of bipolar and unipolar electrogram voltage to scar transmurality and composition derived by magnetic resonance imaging in patients with nonischemic cardiomyopathy undergoing VT ablation. Heart Rhythm. 2012;9:1837–46.
Desjardins B, Yokokawa M, Good E, Crawford T, Latchamsetty R, Jongnarangsin K, et al. Characteristics of intramural scar in patients with nonischemic cardiomyopathy and relation to intramural ventricular arrhythmias. Circ Arrhythm Electrophysiol. 2013;6:891–7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Data is collected in a registry approved by the University of Pennsylvania Investigational Review Board and informed consent is obtained on all patients prior to procedures.
Financial support
Supported in part by The Richard T. and Angela Clark Innovation Fund in Cardiac Electrophysiology and The F. Harlan Batrus Research Fund.
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Betensky, B.P., Dong, W., D’Souza, B.A. et al. Cardiac magnetic resonance imaging and electroanatomic voltage discordance in non-ischemic left ventricle ventricular tachycardia and premature ventricular depolarizations. J Interv Card Electrophysiol 49, 11–19 (2017). https://doi.org/10.1007/s10840-017-0228-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10840-017-0228-8