Abstract
Purpose
To compare two different embryo culture methods and to determine whether grouping embryos based on quality following Day 3 improved outcomes.
Methods
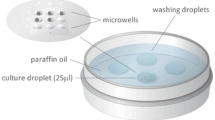
Two group embryo culture methods were compared in this study. All zygotes were individually cultured from Day 1 to Day 3. On Day 3, embryos were then cultured in group of 2–5 embryos per droplet until Day 5 or 6. The two group culture methods are: A, embryos were randomly grouped regardless of embryo quality; B, good and poor quality embryos were separately grouped. Blastocyst development rate, blastocyst utilization rate, implantation rate and pregnancy rate were detected.
Results
The group culture of Day 3 embryos, in which good or poor quality embryos were separately grouped, significantly promoted blastocyst development (61.2 %, 289/472) and blastocyst utilization rate (55.9 %, 264/472) in comparison with those embryos that were randomly grouped for culture regardless of embryo quality (44 %, 177/402 and 41.5 %, 167/402). There was no significant difference in the implantation rate and pregnancy rate between two group culture methods.
Conclusions
Grouping of embryos after Day 3 based on embryo quality may benefit blastocyst formation. This may be due to secretion of beneficial factors by good embryos, or removal of detrimental factors from poor embryos. No impacts on pregnancy or implantation outcomes were observed.
Similar content being viewed by others
References
Gardner DK, Lane M, Schoolcraft WB. Culture and transfer of viable blastocysts: a feasible proposition for human IVF. Hum Reprod. 2000;15:9–23.
Milki AA, Hinckley MD, Fisch JD, Dasig D, Behr B. Comparison of blastocyst transfer with day 3 embryo transfer in similar patient populations. Fertil Steril. 2000;73:126–9.
Dobson AT, Raja R, Abeyta MJ, Taylor T, Shen S, et al. Unique transcription through day 3 of human preimplantation development. Hum Mol Genet. 2004;13:1461–70.
Lane M, Gardner DK. Amino acids and vitamins prevent culture-induced metabolic perturbations and associated loss of viability of mouse blastocysts. Hum Reprod. 1998;13:991–7.
Menezo Y, Hazout A, Dumont M, Herbaut N, Nicollet B. Coculture of embryos on Vero cells and transfer of blastocysts in humans. Hum Reprod. 1992;1:101–6.
Quinn P, Margalit R. Beneficial effects of coculture with cumulus cells on blastocyst formation in a prospective trial with supernumerary human embryos. J Assist Reprod Genet. 1996;13:9–14.
Martin KL, Leese HJ. Role of glucose in mouse preimplantation embryo development. Mol Reprod Dev. 1995;40:436–43.
Gardner DK. Changes in requirements and utilization of nutrients during mammalian preimplantation embryo development and their significance in embryo culture. Theriogenology. 1998;49:83–102.
Gardner DK, Lane M, Calderon I, Leeton J. Environment of the preimplantation human embryo in vivo: metabolite analysis of oviduct and uterine fluids and metabolism of cumulus cells. Fertil Steril. 1996;65:349–53.
Wiemer WE, Anderson AR, Kyslinger ML, Weikert ML. Embryonic development and pregnancies following sequential culture in human tubal fluid and a modified simplex optimised medium containing amino acids. Reprod Biomed Online. 2002;5:323–7.
Canseco RS, Sparks AE, Person RE, Gwaszdauskas FC. Embryo density and medium volume effects on early murine embryo development. J Assist Reprod Genet. 1992;9:454–7.
Hoelker M, Rings F, Lund Q, Ghanem N, Phatsara C, Griese J, et al. Effect of the microenvironment and embryo density on developmental characteristics and gene expression profile of bovine preimplantative embryos cultured in vitro. Reproduction. 2009;137:415–25.
Rijnders PM, Jansen CAM. Influence of group culture and culture volume on the formation of human blastocysts: a prospective randomized study. Hum Reprod. 1999;14:2333–7.
Fujita T, Umeki H, Shimura H, Kugumiya K, Shiga K. Effect of group culture and embryo-culture conditioned medium on development of bovine embryos. J Reprod Dev. 2006;52:137–42.
Almagor M, Bejar C, Kafka I, Yaffe H. Pregnancy rates after communal growth of preimplantation human embryos in vitro. Fertil Steril. 1996;66(3):394–7.
Moessner J, Dodson WC. The quality of human embryo growth is improved when embryos are cultured in groups rather than separately. Fertil Steril. 1995;64(5):1034–5.
Hardy K, Spanos S. Growth factor expression and function in the human and mouse preimplantation embryo. J Endocrionol. 2002;172:221–36.
O’Neil C. Evidence for the requirement of autocrine growth factors for development of mouse preimplantation embryos in vitro. Biol Reprod. 1997;56:229–37.
Richter KS. The importance of growth factors preimplantation embryo development and in vitro culture. Curr Opin Obstet Gynecol. 2008;20:292–304.
Paria BC, Dey SK. Preimplantation embryo development in vitro: cooperative interactions among embryos and role of growth factors. Proc Natl Acad Sci USA. 1990;87:4756–60.
Reed ML, Woodward BJ, Swain JE. Single or group culture of mammalian embryos: the verdict of the literature. J Reprod Stem Cell Biotechnol. 2011;2:77–87.
Spindler RE, Crichton EG, Agca Y, Loskutoff N, Critser J, Gardner DK, et al. Improved felid embryo development by group culture is maintained with heterospecific companions. Theriogenology. 2006;66(1):82–92.
Spindler RE, Wildt DE. Quality and age of companion felid embryos modulate enhanced development by group culture. Biol Reprod. 2002;66(1):167–73.
Rebollar-Lazaro I, Matson P. The culture of human cleavage stage embryos alone or in groups: effect upon blastocyst utilization rates and implantation. Reprod Biol. 2010;10(3):227–34.
Spyropoulou I, Karamalego C, Bolton VN. A prospective randomized study comparing the outcome of in-vitro fertilization and embryo transfer following culture of human embryos individually or in groups before embryo transfer on day 2. Hum Reprod. 1999;14(1):76–9.
Palermo G, Joris H, Devroey P, Van Steirteghem AC. Pregnancies after intracytoplasmic injection of single spermtozoon into an oocyte. Lancet. 1992;340:17–8.
Gardner DK, Schoolcraft WB. In vitro culture of human blastocyst. In: Jansen R, Mortimer D, editors. Towards reproductive certainty: infertility and genetics beyond 1999. Carnforth: Parthenon Press; 1999. p. 378–88.
Gardner DK, Lane M. Embryo culture system. In: Trounson AO, Gardner DK, editors. Handbook of in vitro fertilization. 2nd ed. Boca Raton: CRC Press; 2000. p. 205–64.
Lana M, Gardnar DK. Effect of incubation volume and embryo density on the development and viability of mouse embryos in vitro. Hum Reprod. 1992;7:558–62.
Stoddart NR, Wild AE, Fleming TP. Stimulation of development in vitro by platelet-activating factor receptor ligands released by mouse preimplantation embryos. J Reprod Fertil. 1996;108:47–53.
Gardnar DK, Lana M, Spitzer A, Batt PA. Enhanced rates of cleavaged and development for sheep zygotes cultured to the blastocyst stage in the absence serum and somatic cells: amino acids, vitamins, and culturing embryos in groups stimulate development. Biol Reprod. 1994;50:390–400.
Palma GA, Clement-Stegewald A, Berg U, Brem G. Role of the embryo number in the development of in vitro produced bovine embryos. Theriogenology. 1992;37:271.
Ferry L, Mermillod P, Massip A, Dessy F. Bovine embryo culture in serum-poor oviduct conditioned medium need cooperation to reach the blastocyst stage. Theriogenology. 1994;42:445–53.
O’Doherty EM, Wade MG, Hill JL, Boland MP. Effects of culturing bovine oocytes either sigly or in groups on development to blastocysts. Theriogenology. 1997;48:161–9.
Ebner T, Shebl O, Moser M, Mayer RB, Arzt W, Tews G. Group culture of human zygotes is superior to individual culture in terms of blastulation, implantation and life birth. Reprod Biomed Online. 2010;21:762–8.
Gardner D, Lana M. Culture systems for the human embryo. In: Textbook of assisted reproductive technologies: laboratory and clinical perspectives, 2008;219–240. Eds Gardner D, Weissman A, Howles C, Shoham Z, Third edition. Informa Healthcare, London.
Kane MT, Morgan PM, Coonan C. Peptide growth factors and preimplantation development. Hum Reprod Update. 1997;3:137–57.
Summers MC, Biggers JD. Chemically defined media and the culture of mammalian preimplantation embryos: historical perspective and current issues. Hum Reprod Update. 2003;9:557–82.
Salahuddin S, Ookutsu S, Goto K, Nakanishi Y, Nagata Y. Effects of embryo density and co-culture of unfertilized oocytes on embryonic development of in-vitro fertilized mouse embryo. Hum Reprod. 1995;10:2382–5.
Lane M, Gardner DK. Differential regulation of mouse embryo development and viability by amino acids. J Reprod Fertil. 1997;109:153–64.
Kato Y, Tsunoda Y. Effects of the culture density of mouse zygotes on the development in vitro and in vivo. Theriogenology. 1994;41:1315–22.
Gardner DK, Lana M. Amino acids and ammonium regulate mouse embryo development in culture. Biol Reprod. 1993;48:377–85.
Johnson MH, Nasr-Esfahani MH. Radical solutions and culture problem: could free oxygen radicals be responsible for the impaired development of preimplantation mammalian embryos in vitro ? BioAssays. 1994;16:31–8.
Sinclair KD, Dunne LD, Maxfield EK, Maltin CA, Young LE, Wilmut I, et al. Fetal growth and development following temporary exposure of day 3 ovine embryos to an advanced uterine environment. Reprod Fertil Dev. 1998;10:263–9.
Montag M, Van der Venl H. Evaluation of pronuclear morphology as the only selection criterion for further embryo culture and transfer: results of a prospective multicentre study. Hum Reprod. 2001;16:2384–9.
Tesarik J, Greco E. The probability of abnormal preimplantation development can be predicted by a single static observation on pronuclear stage morphology. Hum Reprod. 1999;14:1318–23.
Balakier H, Cadesky K. The frequency and developmental capability of human embryos containing multinucleated blastomeres. Hum Reprod. 1997;12:800–4.
Kligman I, Benadiva C, Alikani M, Munne S. The presence of multinucleated blastomeres in human embryos is correlated with chromosomal abnormalities. Hum Reprod. 1996;11:1492–8.
Jun SH, Choi B, Shahine L, Westphal LM, Behr B, et al. Defining human embryo phenotypes by cohort-specific prognostic factors. PLoS One. 2008;3:1–7.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule Our study demonstrates that Day 3 embryos with good and poor quality may be separately grouped for culture to blastocyst stage. Some unknown factors derived from poor quality embryos (such as arrested embryos) may inhibit the surrounding embryo development. The strategy of the combination of individual culture to Day 3 and good and poor quality of embryos be separately grouped for culture to Day 5 or 6 is recommended for clinical use.
Rights and permissions
About this article
Cite this article
Tao, T., Robichaud, A., Mercier, J. et al. Influence of group embryo culture strategies on the blastocyst development and pregnancy outcome. J Assist Reprod Genet 30, 63–68 (2013). https://doi.org/10.1007/s10815-012-9892-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-012-9892-x