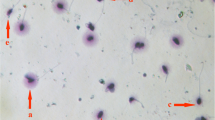
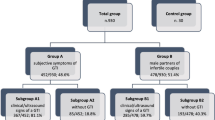
Abstract
Purpose
To verify the prevalence of semen bacterial contamination and whether the contamination could decrease sperm quality.
Methods
Spermiogram, semen culture, and sperm transmission electron microscopy (TEM) analysis were performed. TEM data were elaborated using a mathematical formula that calculates a fertility index (FI)—able to define patients as fertile or infertile—and the percentage of sperm apoptosis, immaturity and necrosis. We aligned the amino acid sequence of beta-tubulin with protein of the most frequent species isolated from semen.
Results
Patients were divided according to the contaminating species; in each group, we observed fertile individuals, in whom the semen quality was similar to that of controls and infertile men whose sperm quality was significantly decreased, in terms of motility, FI, apoptosis and necrosis. Partial homology between β-tubulin and bacterial proteins was observed.
Conclusion
Sperm bacterial contamination is quite frequent and could contribute to the deterioration of the sperm quality of infertile men.

Similar content being viewed by others
References
Pellati D, Mylonakis I, Bertoloni G, Fiore C, Andrisani A, Ambrosini G, et al. Genital tract infections and infertility. Eur J Obstet Gynecol Reprod Biol 2008;140:3–11. doi:10.1016/j.ejogrb.2008.03.009.
Henkel R, Schill WB. Sperm separation in patients with urogenital infections. Andrologia 1998;30:91–7.
Urata K, Narahara H, Tanaka Y, Egashira T, Takayama F, Miyakawa I. Effect of endotoxin-induced reactive oxygen species on sperm motility. Fertil Steril 2001;76:163–6. doi:10.1016/S0015-0282(01)01850-7.
Sanocka-Maciejewska D, Ciupińska M, Kurpisz M. Bacterial infection and semen quality. J Reprod Immunol 2005;67:51–6. doi:10.1016/j.jri.2005.06.003.
Purvis K, Christiansen E. Infection in the male reproductive tract. Impact, diagnosis and treatment in relation to male infertility. Int J Androl 1993;16:1–13. doi:10.1111/j.1365-2605.1993.tb01146.x.
Diemer T, Huwe P, Ludwig M, Schroeder-Printzen I, Michelmann HW, Schiefer HG, et al. Influence of autogenous leucocytes and Escherichia coli on sperm motility parameters in vitro. Andrologia 2003;35:100–5. doi:10.1046/j.1439-0272.2003.00523.x.
Diemer T, Huwe P, Michelmann HW, Mayer F, Schiefer HG, Weidner W. Escherichia coli-induced alterations of human spermatozoa. An electron microscopy analysis. Int J Androl 2000;23:178–86. doi:10.1046/j.1365-2605.2000.00224.x.
Mehta RH, Sridhar H, Vijay Kumar BR, Anand Kumar TC. High incidence of oligozoospermia and teratozoospermia in human semen infected with the aerobic bacterium Streptococcus faecalis. RBM Online 2002;5:17–21.
Qiang H, Jiang MS, Lin JY, He WM. Influence of enterococci on human sperm membrane in vitro. Asian J Androl 2007;9:77–81. doi:10.1111/j.1745-7262.2007.00219.x.
Gdoura R, Kchaou W, Chaari C, Znazen A, Keskes L, Rebai T, et al. Ureaplasma urealyticum, Ureaplasma parvum, Mycoplasma hominis and Mycoplasma genitalium infections and semen quality of infertile men. BMC Infect Dis 2007;7:129. doi:10.1186/1471-2334-7-129.
Fraczek M, Kurpisz M. Inflammatory mediators exert toxic effects of oxidative stress on human spermatozoa. J Androl 2007;28:325–33. doi:10.2164/jandrol.106.001149.
Ombelet W, Bosmans E, Janssen M, Cox A, Vlasselaer J, Gyselaers W, et al. Semen parameters in a fertile versus subfertile population: a need for change in the interpretation of semen testing. Hum Reprod 1997;12:987–93. doi:10.1093/humrep/12.5.987.
World Health Organization. WHO laboratory manual for the examination of human semen and semen-cervical mucus interactions. 4thth ed. Cambridge, United Kingdom: Cambridge University Press; 1999.
Baccetti B, Bernieri G, Burrini AG, Collodel G, Crisà N, Mirolli M, et al. Notulae seminologicae. 5. Mathematical evaluation of interdependent submicroscopic sperm alterations. J Androl. 1995;16:356–71.
Collodel G, Moretti E. Morphology and meiotic segregation in spermatozoa from men of proven fertility. J Androl 2008;29:106–14. doi:10.2164/jandrol.107.002998.
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, et al. Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 1997;25:3389–402. doi:10.1093/nar/25.17.3389.
Haidl G. Macrophages in semen are indicative of chronic epididymal infection. Arch Androl 1990;25:5–11. doi:10.3109/01485019008987587.
Tremellen K. Oxidative stress and male infertility—a clinical perspective. Hum Reprod Update 2008;14:243–58. doi:10.1093/humupd/dmn004.
Fraczek M, Szumala-Kakol A, Jedrzejczak P, Kamieniczna M, Kurpisz M. Bacteria trigger oxygen radical release and sperm lipid peroxidation in in vitro model of semen inflammation. Fertil Steril 2007;88:1076–85. doi:10.1016/j.fertnstert.2006.12.025.
Potts JM, Sharma R, Pasqualotto F, Nelson D, Hall G, Agarwal A. Association of ureaplasma urealyticum with abnormal reactive oxygen species levels and absence of leukocytospermia. J Urol 2000;163:1775–8. doi:10.1016/S0022-5347(05)67540-4.
Villegas J, Schulz M, Soto L, Sanchez R. Bacteria induce expression of apoptosis in human spermatozoa. Apoptosis 2005;10:105–10. doi:10.1007/s10495-005-6065-8.
Fanibunda SE, Velhal SM, Raghavan VP, Bandivdekar AH. CD4 independent binding of HIV gp120 to mannose receptor on human spermatozoa. J Acquir Immune Defic Syndr 2008;48:389–97.
Fujita K, Yokota T, Oguri T, Fujime M, Kitagawa R. In vitro adherence of Staphylococcus saprophyticus, Staphylococcus epidermidis, Staphylococcus haemolyticus, and Staphylococcus aureus to human ureter. Urol Res 1992;6:399–402. doi:10.1007/BF00294495.
Nallapareddy SR, Singh KV, Sillanpää J, Garsin DA, Höök M, Erlandsen SL, et al. Endocarditis and biofilm-associated pili of Enterococcus faecalis. J Clin Invest 2006;116:2582–4. doi:10.1172/JCI29021.
Baccetti BM, Bruni E, Capitani S, Collodel G, Mancini S, Piomboni P, et al. Studies on varicocele III: ultrastructural sperm evaluation and 18, X and Y aneuploidies. J Androl 2006;27:94–101. doi:10.2164/jandrol.05081.
Collodel G, Baccetti B, Capitani S, Moretti E. Necrosis in human spermatozoa. I. Ultrastructural features and FISH study in semen from patients with uro-genital infections. J Submicrosc Cytol Pathol 2005;37:67–73.
Grassmé H, Jendrossek V, Gulbins E. Molecular mechanisms of bacteria induced apoptosis. Apoptosis 2001;6:441–5. doi:10.1023/A:1012485506972.
Eggert-Kruse W, Rohr G, Ströck W, Pohl S, Schwalbach B, Runnebaum B. Anaerobes in ejaculates of subfertile men. Hum Reprod Update 1995;1:462–78. doi:10.1093/humupd/1.5.462.
Kiessling AA, Desmarais BM, Yin HZ, Loverde J, Eyre RC. Detection and identification of bacterial DNA in semen. Fertil Steril 2008;90:1744–56. doi:10.1016/j.fertnstert.2007.08.083.
Acknowledgements
Research supported by Piano di Ateneo per la Ricerca (PAR) grant of University of Siena, 2005 (G.C.), 2006 (N.F.), Italy.
Author information
Authors and Affiliations
Corresponding author
Additional information
Capsule the presence of bacteria in semen may decrease sperm motility and increase apoptosis and necrosis particularly in infertile men.
Rights and permissions
About this article
Cite this article
Moretti, E., Capitani, S., Figura, N. et al. The presence of bacteria species in semen and sperm quality. J Assist Reprod Genet 26, 47–56 (2009). https://doi.org/10.1007/s10815-008-9283-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10815-008-9283-5