Abstract
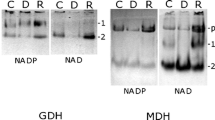
Methanol in low concentrations can stimulate the mixotrophic growth of some microscopic algae. The aim of the present work was to investigate the effect of methanol on the growth, photosynthesis, and respiration rate as well as free amino acid and soluble protein content in the cells of unicellular green alga Chlamydomonas reinhardtii. It was shown that 30–100 mM methanol induced an increase in C. reinhardtii biomass production compared to controls without the solvent. The packed cell volume was increased maximally by 35 % after growing for 6 days in the presence of 50 mM methanol. The effect was light-dependent, although the rate of photosynthesis changed insignificantly while the rate of respiration increased. The intracellular content of reduced nicotinamide coenzyme NAD(P)H also increased after methanol addition in the light. The intracellular content of free amino acids increased by 31 % as a result of 50 mM methanol addition, and their composition changed: glutamic acid, glutamine, alanine, serine, and tyrosine increased and methionine content decreased. The content of soluble protein also increased by 30 %, eliminating the possibility of proteolysis. Thus, methanol has a positive effect on nitrogen assimilation as indicated by the increase in the content of soluble proteins and free amino acids. This effect may be connected with methanol-induced stimulation of respiration and the light-dependent increase in NAD(P)H content. The results suggest that methanol not only is a carbon source for C. reinhardtii cells but also can take part in energy metabolism.






Similar content being viewed by others
References
Badger MR, Spalding MH (2000) CO2 acquisition, concentration and fixation in cyanobacteria and algae. In: Leegood RC, Sharkey TD, von Caemmerer S (eds) Photosynthesis: physiology and metabolism. Kluwer, Dordrecht, pp 369–397
Bishop NI, Senger H (1971) Preparation and photosynthetic properties of synchronous cultures of Scenedesmus. In: San Pietro A (ed) Methods in enzymology, 23A. Acad Press, NY, pp 53–66
Bölling C, Fiehn O (2005) Metabolite profiling of Chlamydomonas reinhardtii under nutrient deprivation. Plant Physiol 139:1995–2005
Chaabane L, Dammak L, Nikonenko VV, Bulvestre G, Auclair B (2007) The influence of absorbed methanol on the conductivity and on the microstructure of ion-exchange membranes. J Membrane Sci 298:126–135
Choi WY, Oh SH, Seo YC, Kim GB, Kang DH, Lee SY, Jung KH, Cho JS, Ahn JH, Choi GP, Lee HY (2011) Effects of methanol on cell growth and lipid production from mixotrophic cultivation of Chlorella sp. Biotechnol Bioprocess Eng 16:946–955
Cossins R (1964) The utilization of carbon-1 compounds by plants The metabolism of methanol-14C and its role in amino acid biosynthesis. Can J Biochem 44:1739–1802
Cournac L, Latouche G, Cerovic Z, Redding K, Ravenel J, Peltier G (2002) In vivo interactions between photosynthesis, mitorespiration, and chlororespiration in Chlamydomonas reinhardtii. Plant Physiol 129:1921–1928
Downie A, Miyazaki S, Bohnert H, John P, Coleman J, Parry M, Haslam R (2004) Expression profiling of the response of Arabidopsis thaliana to methanol stimulation. Phytochemistry 65:2305–2316
Fall R, Benson A (1996) Leaf methanol—the simplest natural product from plants. Trends Plant Sci 1:296–301
Fan J, Yan C, Andre C, Shanklin J, Schwender J, Xu C (2012) Oil accumulation is controlled by carbon precursor supply for fatty acid synthesis in Chlamydomonas reinhardtii. Plant Cell Physiol 53:1380–1390
Florencio FJ, Vega JM (1983) Separation, purification, and characterization of two isoforms of glutamine synthetase from Chlamydomonas reinhardtii. Z Naturforsch 38c:531–538
Gout E, Aubert S, Bligny R, Rébeillé F, Nonomura AR, Benson AA, Douce R (2000) Metabolism of methanol in plant cells Carbon-13 nuclear magnetic resonance studies. Plant Physiol 123:287–296
Hanson AD, Roje S (2001) Оne-carbon metabolism in higher plants. Annu Rev Plant Physiol Plant Mol Biol 52:119–137
Harris EH (2009) The Chalmydomonas sourcebook. Academic Press, San Diego, CA
Isidorov VA, Zenkevich IG, Ioffe BV (1985) Volatile organic compounds in the atmosphere of forest. Atmos Environ 19:1–8
Jacob DJ, Field BD, Li Q, Blake DR, de Gouw J, Warneke C, Hansel A, Wisthaler A, Singh HB, Guenther A (2005) Global budget of methanol: constraints from atmospheric observations. J Geophys Res 110, D08303. doi:10.1029/2004JD005172
Johnson X, Alric J (2013) Central carbon metabolism and electron transport in Chlamydomonas reinhardtii: metabolic constraints for carbon transport in partitioning between oil and starch. Eukaryot Cell 12:776–793
Kotzabasis K, Hatziathanasiou A, Bengoa-Ruigomez MV, Kentouri M, Divanach P (1999) Methanol as alternative carbon source for quicker efficient production of the microalgae Chlorella minutissima: role of the concentration and frequence of administration. J Biotechnol 70:357–362
Lea PJ, Miflin BJ (1974) An alternative route for nitrogen assimilation in higher plants. Nature 251:614–616
Lowry OH, Rosenbrough NJ, Farr AL, Randell RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
MacDonald R, Fall R (1993) Detection of substantial emissions of methanol from plants to the atmosphere. Atm Environ 27A:1709–1713
Moroney JV, Ynalvez RA (2007) Proposed carbon dioxide concentrating mechanism in Chlamydomonas reinhardtii. Eukaryot Cell 6:1251–1259
Navakoudis E, Ioannidis NE, Dörnemann D, Kotzabasis K (2007) Changes in the LHCII-mediated energy utilization and dissipation adjust the methanol-induced biomass increase. Biochim Biophys Acta 1767:948–955
Noctor G, Arisi A-CM, Jouanin L, Foyer CH (1999) Photorespiratory glycine enhances glutathione accumulation in both the chloroplastic and cytosolic compartments. J Exp Bot 50:1157–1167
Nonomura A, Benson A (1992) The path of carbon in photosynthesis: improved crop yields with methanol. Proc Natl Acad Sci U S A 89:9794–9798
Patra M, Salonen E, Terama E, Vattulainen I, Faller R, Lee BW, Holopainen J, Karttunen M (2006) Under the influence of alcohol: the effect of ethanol and methanol on lipid bilayers. Biophys J 90:1121–1135
Rai VK (2002) Role of amino acids in plant responses to stresses. Biol Plantarum 45:481–487
Renberg L, Johansson AI, Shutova T, Stenlund H, Aksmann A, Raven JA, Gardeström P, Moritzand T, Samuelsson G (2010) A metabolomic approach to study major metabolite changes during acclimation to limiting CO2 in Chlamydomonas reinhardtii. Plant Physiol 154:187–196
Sager R, Granick S (1953) Nutritional studies with Chlamydomonas reinhardii. Ann N Y Acad Sci 56:831–838
Sharkey T (1988) Estimating the rate of photorespiration in leaves. Physiol Plant 73:147–152
Siaut M, Cuine S, Cagnon C, Fessler B, Nguyen M, Carrier P, Beyly A, Beisson F, Triantaphylides C, Li-Beisson Y, Peltier G (2010) Oil accumulation in the model green alga Chlamydomonas reinhardtii: characterization, variability between common laboratory strains and relationship with starch reserves. BMC Biotechnol 11:7. doi:10.1186/1472-6750-11-7
Singh BK, Shaner DL (1995) Biosynthesis of branched chain amino acids: from test tube to field. Plant Cell 7:935–944
Spalding MH (2008) Microalgal carbon-dioxide-concentrating mechanisms: Chlamydomonas inorganic carbon transporters. J Exp Bot 59:1463–1473
Theodoridou A, Dörnemann D, Kotzabasis K (2002) Light dependent induction of strongly increased microalgal growth by methanol. Biochim Biophys Acta 1573:189–198
Umbarger HE (1978) Amino acid biosynthesis and its regulation. Annu Rev Biochem 47:533–606
Waffenschmidt S, Woessner JP, Beer K, Goodenough UW (1993) Isodityrosine cross-linking mediates insolubilization of cell walls in Chlamydomonas. Plant Cell 5:809–820
Acknowledgments
The authors are grateful to Alexander Teddy for language corrections.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stepanov, S.S., Zolotareva, E.K. Methanol-induced stimulation of growth, intracellular amino acids, and protein content in Chlamydomonas reinhardtii . J Appl Phycol 27, 1509–1516 (2015). https://doi.org/10.1007/s10811-014-0445-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10811-014-0445-9