Abstract
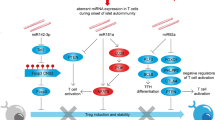
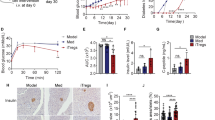
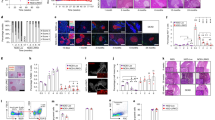
Type-1 diabetes (TID) is an autoimmune disease in which the body’s own immune cells attack islet β cells, the cells in the pancreas that produce and release the hormone insulin. Mir-26a has been reported to play functions in cellular differentiation, cell growth, cell apoptosis, and metastasis. However, the role of microRNA-26a (Mir-26a) in autoimmune TID has never been investigated. In our current study, we found that pre-Mir-26a (LV-26a)-treated mice had significantly longer normoglycemic time and lower frequency of autoreactive IFN-γ-producing CD4+ cells compared with an empty lentiviral vector (LV-Con)-treated non-obese diabetic (NOD) mice. Mir-26a suppresses autoreactive T cells and expands Tregs in vivo and in vitro. Furthermore, in our adoptive transfer study, the groups receiving whole splenocytes and CD25-depleted splenocytes from LV-Con-treated diabetic NOD mice develop diabetes at 3 to 4 weeks of age. In comparison, mice injected with undepleted splenocytes obtained from LV-26a-treated reversal NOD mice develop diabetes after 6–8 weeks. And depletion of CD25+ cells in the splenocytes of reversed mice abrogates the delay in diabetes onset. In conclusion, Mir-26a suppresses autoimmune diabetes in NOD mice in part through promoted regulatory T cells (Tregs) expression.





Similar content being viewed by others
References
Rowe, P.M. 1998. Treating type 1 diabetes by regulating autoimmunity. Lancet 352: 966.
O’Connell, R.M., D.S. Rao, A.A. Chaudhuri, and D. Baltimore. 2010. Physiological and pathological roles for microRNAs in the immune system. Nature Reviews Immunology 10: 111–122.
Lu, J., G. Getz, E.A. Miska, et al. 2005. MicroRNA expression profiles classify human cancers. Nature 435: 834–838.
Zhang, B., X.X. Liu, J.R. He, et al. 2011. Pathologically decreased miR-26a antagonizes apoptosis and facilitates carcinogenesis by targeting MTDH and EZH2 in breast cancer. Carcinogenesis 32: 2–9.
Chen, L., J. Zheng, Y. Zhang, et al. 2011. Tumor-specific expression of microRNA-26a suppresses human hepatocellular carcinoma growth via cyclin-dependent and -independent pathways. Molecular Therapy 19: 1521–1528.
Leeper, N.J., A. Raiesdana, Y. Kojima, et al. 2011. MicroRNA-26a is a novel regulator of vascular smooth muscle cell function. Journal of Cellular Physiology 226: 1035–1043.
Liu, B., X. Wu, B. Liu, et al. 2012. MiR-26a enhances metastasis potential of lung cancer cells via AKT pathway by targeting PTEN. Biochimica et Biophysica Acta 1822: 1692–1704.
Zhang R, Tian A, Wang J, Shen X, Qi G, Tang Y. 2014. miR26a Modulates T17/T Balance in the EAE Model of Multiple Sclerosis by Targeting IL6. Neuromolecular Medicine.
Xie F, Chai J, Zhang Z, Hu Q, Ma T. 2015. MicroRNA 26a Prolongs Skin Allograft Survival and Promotes Regulatory T Cells Expansion in mice. Transplant International.
Wing, K., and S. Sakaguchi. 2010. Regulatory T cells exert checks and balances on self tolerance and autoimmunity. Nature Immunology 11: 7–13.
Petzold, C., J. Riewaldt, D. Watts, T. Sparwasser, S. Schallenberg, and K. Kretschmer. 2013. Foxp3(+) regulatory T cells in mouse models of type 1 diabetes. Journal of Diabetes Research 2013: 940710.
Zoka, A., G. Barna, A. Somogyi, G. Muzes, A. Olah, Z. Al-Aissa, O. Hadarits, K. Kiss and G. Firneisz. 2014. Extension of the CD4Foxp3CD25 regulatory T-cell subpopulation in type 1 diabetes mellitus. Autoimmunity: 1–9.
Abdi, R., P. Fiorina, C.N. Adra, M. Atkinson, and M.H. Sayegh. 2008. Immunomodulation by mesenchymal stem cells: a potential therapeutic strategy for type 1 diabetes. Diabetes 57: 1759–1767.
Fiorina, P., M. Jurewicz, A. Augello, et al. 2009. Immunomodulatory function of bone marrow-derived mesenchymal stem cells in experimental autoimmune type 1 diabetes. Journal of Immunology 183: 993–1004.
Anderson, M.S., and J.A. Bluestone. 2005. The NOD mouse: a model of immune dysregulation. Annual Review of Immunology 23: 447–485.
Bach, J.F. 1994. Insulin-dependent diabetes mellitus as an autoimmune disease. Endocrine Reviews 15: 516–542.
Tisch, R., and H. McDevitt. 1996. Insulin-dependent diabetes mellitus. Cell 85: 291–297.
Andre, I., A. Gonzalez, B. Wang, J. Katz, C. Benoist, and D. Mathis. 1996. Checkpoints in the progression of autoimmune disease: lessons from diabetes models. Proceedings of the National Academy of Sciences of the United States of America 93: 2260–2263.
Brusko, T.M., C.H. Wasserfall, M.J. Clare-Salzler, D.A. Schatz, and M.A. Atkinson. 2005. Functional defects and the influence of age on the frequency of CD4+ CD25+ T-cells in type 1 diabetes. Diabetes 54: 1407–1414.
Pop, S.M., C.P. Wong, D.A. Culton, S.H. Clarke, and R. Tisch. 2005. Single cell analysis shows decreasing FoxP3 and TGFbeta1 coexpressing CD4+ CD25+ regulatory T cells during autoimmune diabetes. Journal of Experimental Medicine 201: 1333–1346.
Tritt, M., E. Sgouroudis, E. D’Hennezel, A. Albanese, and C.A. Piccirillo. 2008. Functional waning of naturally occurring CD4+ regulatory T-cells contributes to the onset of autoimmune diabetes. Diabetes 57: 113–123.
You, S., M. Belghith, S. Cobbold, et al. 2005. Autoimmune diabetes onset results from qualitative rather than quantitative age-dependent changes in pathogenic T-cells. Diabetes 54: 1415–1422.
Fontenot, J.D., M.A. Gavin, and A.Y. Rudensky. 2003. Foxp3 programs the development and function of CD4+ CD25+ regulatory T cells. Nature Immunology 4: 330–336.
Golshayan, D., S. Jiang, J. Tsang, M.I. Garin, C. Mottet, and R.I. Lechler. 2007. In vitro-expanded donor alloantigen-specific CD4+ CD25+ regulatory T cells promote experimental transplantation tolerance. Blood 109: 827–835.
Mottet, C., H.H. Uhlig, and F. Powrie. 2003. Cutting edge: cure of colitis by CD4+ CD25+ regulatory T cells. Journal of Immunology 170: 3939–3943.
Tang, Q., K.J. Henriksen, M. Bi, et al. 2004. In vitro-expanded antigen-specific regulatory T cells suppress autoimmune diabetes. Journal of Experimental Medicine 199: 1455–1465.
Hall, B.M., G. Tran, and S.J. Hodgkinson. 2009. Alloantigen specific T regulatory cells in transplant tolerance. International Immunopharmacology 9: 570–574.
O’Connell, P.J., S. Yi, E.M. Carrington, and A.M. Lew. 2010. Role of regulatory T cells in xenotransplantation. Current Opinion in Organ Transplantation 15: 224–229.
Borsellino, G., M. Kleinewietfeld, D. Di Mitri, et al. 2007. Expression of ectonucleotidase CD39 by Foxp3+ Treg cells: hydrolysis of extracellular ATP and immune suppression. Blood 110: 1225–1232.
Kobie, J.J., P.R. Shah, L. Yang, J.A. Rebhahn, D.J. Fowell, and T.R. Mosmann. 2006. T regulatory and primed uncommitted CD4 T cells express CD73, which suppresses effector CD4 T cells by converting 5′-adenosine monophosphate to adenosine. Journal of Immunology 177: 6780–6786.
Mira, E., B. Leon, D.F. Barber, et al. 2008. Statins induce regulatory T cell recruitment via a CCL1 dependent pathway. Journal of Immunology 181: 3524–3534.
Murphy, T.J., C.N. Ni, Y. Zang, J.A. Mannick, and J.A. Lederer. 2005. CD4+ CD25+ regulatory T cells control innate immune reactivity after injury. Journal of Immunology 174: 2957–2963.
Kim, K.D., J. Zhao, S. Auh, et al. 2007. Adaptive immune cells temper initial innate responses. Nature Medicine 13: 1248–1252.
Mahajan, D., Y. Wang, X. Qin, et al. 2006. CD4+ CD25+ regulatory T cells protect against injury in an innate murine model of chronic kidney disease. Journal of the American Society of Nephrology 17: 2731–2741.
Author information
Authors and Affiliations
Corresponding author
Additional information
Hui Ma and Shoutao Zhang contributed equally to this work.
Rights and permissions
About this article
Cite this article
Ma, H., Zhang, S., Shi, D. et al. MicroRNA-26a Promotes Regulatory T cells and Suppresses Autoimmune Diabetes in Mice. Inflammation 39, 1–9 (2016). https://doi.org/10.1007/s10753-015-0215-0
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10753-015-0215-0