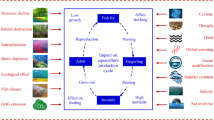
Abstract
This study evaluated the effects of three climate scenarios predicted by the Intergovernmental Panel on Climate Change for 2100, on the growth and physiology of the tambaqui (Colossoma macropomum). Juveniles of tambaqui were reared in four rooms (current conditions, and the climate scenarios B1, A1B and A2) for 150 days. Twelve fish were regularly collected for blood analyses and to have their weight and length measured. The amount of food consumed over the experimental period was recorded. Feed intake, weight gain and food conversion showed an increase in all climate scenarios (P < 0.05). Although these results were expected, the increases in weight gain in the more dramatic environmental situations were due to increases in feed intake. Increases in haematocrit, glucose, cortisol and triglycerides within the first 30 days of the experiment were observed. The present results indicate that climate changes can affect not only its feed intake, but also the growth of the tambaqui and that this fish species can recover blood parameters, suggesting an artificial acclimatization to the analysed environmental conditions after 30 days. Taken to nature, these characteristics of the tambaqui could cause ecological imbalances in the food web, in its frequency of occurrence and in its dominance.





Similar content being viewed by others
References
Almeida, O. T. & B. Almeida, 2006. Caracterização e análise financeira da indústria pesqueira. In Almeida, O. (ed), A Indústria Pesqueira na Amazônia. Ibama/ProVarzea, Manaus: 83–107.
Araújo-Lima, C. & M. Goulding, 1998. Os Frutos do Tambaqui: Ecologia, Conservação e Cultivo na Amazônia. Sociedade Civil Mamirauá/CNPq, Tefé/Am. 187p.
Auer, S. K., K. Salin, A. M. Rudolf, G. J. Anderson & N. B. Metcalfe, 2015. The optimal combination of standard metabolic rate and aerobic scope for somatic growth depends on food availability. Functional Ecology 29: 479–486.
Batista, V. D. S. & M. Petrere Junior, 2003. Characterization of the commercial fish production landed at Manaus, Amazonas State, Brazil. Acta Amazonica 33: 53–66.
Boyd, C. E. & C. S. Tucker, 1992. Water quality and pond soil analysis for aquaculture. Auburn University, Auburn.
Brett, J. R., 1969. Temperature and fish. Chesapeake Science 10: 275–276.
Brown, B. A., 1976. Hematology: Principles and procedures, 2nd ed. Lea & Febiger, Philadelphia.
Callaway, R., A. P. Shinn, S. E. Grenfell, J. E. Bron, G. Burnell, E. J. Cook, M. Crumlish, S. Culloty, K. Davidson, R. P. Ellis, K. J. Flynn, C. Fox, D. M. Green, G. C. Hays, A. D. Hughes, E. Johnston, C. D. Lowe, I. Lupatsch, S. Malham, A. F. Mendzil, T. Nickell, T. Pickerell, A. F. Rowley, M. S. Stanley, D. R. Tocher, J. F. Turnbull, G. Webb, E. Wootton & R. J. Shields, 2012. Review of climate change impacts on marine aquaculture in the UK and Ireland. Aquatic Conservation: Marine And Freshwater Ecosystems. Wiley Online Library (www.wileyonlinelibrary.com). doi:10.1002/aqc.2247.
Crockett, E. L., 1998. Cholesterol function in plasma membranes from ectotherms: membrane-specific roles in adaptation to temperature. American Zoologist 38: 291–304.
Donelson, J. M., P. L. Munday, M. I. McCormick, N. W. Pankhurst & P. M. Pankhurst, 2010. Effects of elevated water temperature and food availability on the reproductive performance of a coral reef fish. Marine Ecology Progress Series 401: 233–243.
Duffy, P. B., P. Brando, G. P. Asner & C. B. Field, 2015. Projections of future meteorological drought and wet periods in the Amazon. Proceedings of the National Academy of Sciences of the United States of America 112: 13172–13177.
Ferreira, M. W., F. G. Araújo, D. V. Costa, P. R. V. Logato, H. C. P. Figueiredo & L. D. S. Murgas, 2011. Influence of dietary oil sources on muscle composition and plasma lipoprotein concentrations in Nile Tilapia, Oreochromis niloticus. Journal of the World Aquaculture Society 42: 24–33.
Fivelstad, S. A., T. Olsen, G. Asgard, T. Bæverfjord, T. Vindheim Rasmussen & S. O. Stefansson, 2003. Long-term sub-lethal effects of carbon dioxide on Atlantic salmon smolts: ion regulation, haematology, element composition, nephrocalcinosis and growth parameters. Aquaculture. 215: 301–319.
Foss, A., B. Rosnes & V. Oiestad, 2003. Graded environmental hypercapnia in juvenile spotted wolffish (Anarhichas minor Olafsen): effects on growth, food conversion efficiency and nephrocalcinosis. Aquaculture 220: 607–617.
Gilman, S. E., M. C. Urban, J. Tewksbury, G. W. Gilchrist & R. D. Holt, 2010. A framework for community interactions under climate change. Trends in Ecology and Evolution 25: 325–331.
Gomes, L. C., B. Baldisserotto & J. A. Senhorini, 2000. Effect of stocking density on water quality, survival, and growth of larvae of matrinxã, Brycon cephalus (Characidae), in ponds. Aquaculture 183: 73–81.
Gomes, L. C., L. N. Simões & C. A. R. M. Araújo-Lima, 2010. Tambaqui (Colossoma macropomum). In Baldisserotto, B. & L. C. Gomes (eds), Espécies Nativas Para Piscicultura no Brasil, 2nd ed. Santa Maria, UFSM.
Goulding, M., 1980. The fishes and the forest; explorations in amazonian natural history. University California Press, Berkeley/Los Angeles, 280p.
Hasler, C. T., D. Butman, J. D. Jeffrey & C. D. Susk, 2016. Freshwater biota and rising pCO2? Ecology Letters 19: 98–108.
Houston, A. H. & A. Murad, 1995. Erythrodynamics in fish – recovery of the goldfish Carassius auratus from acute anemia. Canadian Journal of Zoology 73: 411–418.
Intergovernmental Panel on Climate Change (IPCC), 2007. Summary for policymakers. In Solomon, S., D. Qin, M. Manning, Z. Chen, M. Marquis, K. B. Averyt, M. Tignor & H. L. Miller (eds), Climate Change 2007: The Physical Science Basis. Contribution of Working Group I to the Fourth Assessment Report of the Intergovernmental Panel on Climate Change. Cambridge University Press, Cambridge.
Jordal, A. E. O., O. Lie & B. E. Torstensen, 2007. Complete replacement of dietary fish oil with a vegetable oil blend affects liver lipid and plasma lipoprotein levels in Atlantic salmon (Salmo salar L.). Aquaculture Nutrition 13: 114–130.
Kampen, E. J. & W. G. Zijlstra, 1964. Standardization of haemoglobinometry. Blibliotheca Haematologica 18: 68–72.
Lecklin, T. & M. Nikinmaa, 1998. Erythropoiesis in artic charr is not stimulated by anemia. Journal Fish Biology 53: 1169–1177.
Lermen, C. L., R. Lappe, M. Crestani, V. P. Vieira, C. R. Gioda, M. R. C. Schetinger, B. Baldisserotto, G. Moraes & V. M. Morsch, 2004. Effect of different temperature regimes on metabolic and blood parameters of silver catfish Rhamdia quelen. Aquaculture 239: 497–507.
Lima, L. C., L. P. Ribeiro, J. A. Malison, T. P. Barry & A. Heldj, 2006. Effects of temperature on performance characteristics and the cortisol stress response of surubim Pseudoplatystoma sp. Journal of the World Aquaculture Society 37: 89–95.
Luo, M., R. Guan, Z. Li & H. Jin, 2013. The effects of water temperature on the survival, feeding, and growth of the juveniles of Anguilla marmorata and A. bicolor pacifica. Aquaculture 400–401: 61–64.
Marengo, J. A., C. A. Nobre, J. Tomasella, M. D. Oyama, G. S. Oliveira, R. Oliveira, H. Camargo, L. Alves & I. F. Brown, 2008. The drought of Amazonia in 2005. Journal of Climate 21: 495–516.
Marengo, J. A., L. S. Borma, D. A. Rodriguez, P. Pinho, W. R. Soares & L. M. Alves, 2013. Recent extremes of drought and flooding in Amazonia: vulnerabilities and human adaptation. American Journal of Climate Change 2: 87–96.
Melzner, F., M. A. Gutowska, M. Langenbuch, S. Dupont, M. Lucassen, M. C. Thorndyke, M. Bleich & H. O. Pörtner, 2009. Physiological basis for high CO2 tolerance in marine ectothermic animals: pre-adaptation through lifestyle and ontogeny? Biogeosciences 6: 2313–2331.
Nikinmaa, M., 1990. Vertebrate red blood cells. Springer, Berlin/Heidelberg, New York.
Nowicki, J., G. M. Miller & P. L. Munday, 2012. Interactive effects of elevated temperature and CO2 on foraging behavior of juvenile coral reef fish. Journal of Experimental Marine Biology and Ecology 412: 46–51.
Oliveira, A. M., M. N. P. Silva, V. M. F. Almeida-Val & A. L. Val, 2012. Caracterização da atividade de piscicultura nas mesoregiões do estado do Amazonas, Amazônia brasileira. Revista Colombiana Ciência Animal 4: 154–162.
Parmesan, C. & G. Yohe, 2003. A globally coherent fingerprint of climate change impacts across natural systems. Nature 421: 37–42.
Pickering, A. D. & T. G. Pottinger, 1989. Stress responses and disease resistance in salmonid fish: effects of chronic elevation of plasma cortisol. Fish Physiology and Biochemistry 7: 253–258.
Piedras, S. R. N., P. R. R. Moraes & J. L. O. F. Pouey, 2004. Crescimento de juvenis de jundiá (Rhamdia quelen), de acordo com a temperatura da água. Boletim do Instituto de Pesca 30: 177–182.
Ross, R. M., W. F. Krise, L. A. Redell & R. M. Bennett, 2001. Effects of dissolved carbon dioxide on the physiology and behavior of fish in artificial streams. Environmental Toxicology 16: 84–95.
Saint-Paul, U., 1983. Investigations of the respiration of the neotropical fish, Colossoma macropomum (Serrasalmidae: the influence of weight and temperature on the routine oxygen consumption. Amazoniana 7: 433–443.
Saint-Paul, U. & G. M. Soares, 1987. Diurnal distribution and behavioral responses of fishes to extreme hypoxia in an Amazon floodplain lake. Environmental Biology of Fishes 20: 91–104.
Salazar, L. F., C. A. Nobre & M. D. Oyama, 2007. Climate change consequences on the biome distribution in tropical South America. Geophysical Research Letters 34: 1–6.
Santos, I. A. & J. Buchmann, 2010. Uma Revisão Qualitativa Enfatizando Aspectos Climáticos da Amazônia e da Região Nordeste do Brasil. Anuário do Instituto de Geociências 33: 9–23.
Silva, J. A. M., M. Pereira-Filho & M. I. Oliveira-Pereira, 2003. Valor energético de espécies vegetais importantes na alimentação do tambaqui. Acta Amazanica 33: 687–700.
Souza-Netto, J. G., 2012. Respostas fisiológicas de Colossoma macropomum (Cuvier 1818) ao aumento de temperatura e CO2. BADPI/INPA. Manaus/AM, Dissertação de Mestrado: 68p.
Tavares-Dias, M. & F. R. Moraes, 2004. Hematologia de peixes teleósteos. Villimpress, Ribeirão Preto.
Val, A. L., 2000. Organic phosphates in the red blood cells of fish. Comparative Biochemistry and Physiology 125: 417–435.
Val, A. L. & V. M. F. Almeida-Val, 1995. Fishes of the Amazon and Their Environment: Physiological and Biochemical Aspects. Springer-Verlang, Berlim.
Val, A. L., M. N. P. Silva & V. M. F. Almeida-Val, 2004. Estresse em peixes-Ajustes fisiológicos e distúrbios orgânicos. In Ranzani-Paiva, M. J. T., R. M. Takemoto & M. A. P. Lizama (eds), Sanidade de Organismos Aquáticos. São Paulo, Varela: 75–78.
Val, A. L., M. N. Paula-Silva, V. M. F. Almeida-Val & C. M. Wood, 2016. In vitro effects of increased temperature and decreased pH on blood oxygen affinity of 10 fish species of the Amazon. Journal of Fish Biology 89: 264–279.
Wendelaar Bonga, S. E., 1997. The stress response in fish. Physiological Reviews 77: 591–625.
Zar, J. K., 1984. Biostatistical Analysis, 2nd ed. Pretice-Hall, New Jersey.
Acknowledgements
The financial support provided by INCT ADAPTA - CNPq/FAPEAM is acknowledged. The authors also acknowledge the support of SEPROR/AM for helping with fish supply and Fernanda Garcia Dragan, Marcos Prado Lima and Maria de Nazaré Paula da Silva for the analyses and the fish management. AMO was a recipient of PhD fellowship from Brazilian CNPq and is now a recipient of Pos-Doc from Brazilian CAPES. ALV is a recipient of research fellowship from Brazilian CNPq.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest editors: Adalberto L. Val, Gudrun De Boeck & Sidinei M. Thomaz / Adaptation of Aquatic Biota of the Amazon
Rights and permissions
About this article
Cite this article
de Oliveira, A.M., Val, A.L. Effects of climate scenarios on the growth and physiology of the Amazonian fish tambaqui (Colossoma macropomum) (Characiformes: Serrasalmidae). Hydrobiologia 789, 167–178 (2017). https://doi.org/10.1007/s10750-016-2926-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10750-016-2926-0