Abstract
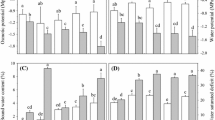
Shock-like water stress using hydroponics and gradual water deficit in soil are the two widely used treatments to analyze transcriptional response of many crops to drought. In this study, we investigated the effects of shock drought (ShD) (0, 0.5, 1, 4, and 8 h) and slow drought (SDD) [soil water content (SWC) of 35 and 50 %] on the expression of well-known drought-responsive genes supplemented with physiological changes in barley. Two barley cultivars with contrasting leaf relative water content (RWC) and water loss rate (WLR) values were selected as Martı (MR; 60 % RWC and 0.046 gh−1 g−1 DW) and Erginel90 (ER; 38.3 % RWC and 0.350 gh−1 g−1 DW) under 38 % of SWC condition. According to the results, 0.5 h ShD was the critical time point for stress perception in leaves defined by the increase in WLR, ion leakage and H2O2 concentration. Expressions of antioxidant-related genes (Cu–Zn/SOD, HvCAT2, HvGST6, HvAPX) were rapidly induced in MR at 8 h shock, while only slightly upregulated in ER. We have also observed higher induction of expressions of HvBAS1, HvMT-2, HvABA7 and a photosynthesis-related gene HvLHCB during ShD compared to SDD. Contrarily, transcription factors (TFs), HvWRKY12 and HvDRF1 were expressed with lower values during shock-dehydration. Slow-drought treatments in both cultivars were characterized with high leaf RWCs and osmotic adjustment with low cell membrane damage, suggesting that barley maintains a basal tolerance to long-term water deficit. Our results confirmed that type of water stress treatment is crucial to measure gene expression, and a shock-like dehydration method should be the treatment of choice in evaluating barley plants with different physiological characteristics for water tolerance.




Similar content being viewed by others
References
Abebe T, Melmaiee K, Berg V, Wise RP (2010) Drought response in the spikes of barley: gene expression in the lemma, palea, awn, and seed. Funct Integr Genomics 10:191–205
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress and signal transduction. Annu Rev Plant Biol 55:373–399
Atienza SG, Faccioli P, Perrotta G, Dalfino G, Zschiesche W, Humbeck K, Stanca AM, Cattivelli L (2004) Large scale analysis of transcripts abundance in barley subjected to several single and combined abiotic stress conditions. Plant Sci 167:1359–1365
Babu RC, Zhang JX, Blum A, Ho THD, Wu R, Nguyen HT (2004) HVA1, a LEA gene from barley confers dehydration tolerance in transgenic rice (Oryza sativa L.) via cell membrane protection. Plant Sci 166:855–862
Bajji M, Kinet J, Lutts S (2002) The use of the electrolyte leakage method for assessing cell membrane stability as a water stress tolerance test in durum wheat. Plant Growth Regul 36:61–70
Balazadeh S, Kwasniewski M, Caldana C, Mehrnia M, Zanor MI, Xue G-P, Mueller-Roeber B (2011) ORS1, an H2O2-responsive NAC transcription factor, controls senescence in Arabidopsis thaliana. Mol Plant 4:346–360
Bandurska H, Niedziela J, Chadzinikolau T (2013) Separate and combined responses to water deficit and UV-B radiation. Plant Sci 213:98–105
Bedada G, Westerbergh A, Müller T, Galkin, E, Bdolach E, Moshelion M, Fridman E, Schmid KJ (2014) Transcriptome sequencing of two wild barley (Hordeum spontaneum L.) ecotypes differentially adapted to drought stress reveals ecotype-specific transcripts. BMC Genomics 15:995
Bustin SA, Benes V, Garson JA, Hellemans J, Huggett J, Kubista M, Mueller R, Nolan T, Pfaffl MW, Shipley GL, Vandesompele J, Wittwer CT (2009) The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 55(4):611–622
Cattivelli L, Rizza F, Badeck FW, Mazzucotelli E, Mastrangelo AM, Francia E, Marè C, Tondelli A, Stanca AM (2008) Drought tolerance improvement in crop plants: an integrated view from breeding to genomics. Field Crop Res 105:1–14
Chaves MM, Maroco JP, Pereira JS (2003) Understanding plant responses to drought from genes to the whole plant. Funct Plant Biol 30:239–264
Chen W, Chao G, Singh KB (1996) The promoter of a H2O2-inducible, Arabidopsis glutathione S-transferase gene contains closely linked OBF- and OBP1-binding sites. Plant J 10:955–966
Cushman JC, Bohnert HJ (2000) Genomic approaches to plant stress tolerance. Curr Opin Plant Biol 3:117–124
de Mezer M, Turska-Taraska A, Kaczmarek Z, Glowacka K, Swarcewicz B, Rorat T (2014) Differential physiological and molecular response of barley genotypes to water deficit. Plant Physiol Biochem 80:234–248
Ergen ZN, Thimmapuram J, Bohnert HJ, Budak H (2009) Transcriptome pathways unique to dehydration tolerant relatives of modern wheat. Funct Integr Genomics 9:377–396
Faralli M, Lektemur C, Rosellini D, Gürel F (2015) Effects of heat shock on salinity tolerance in barley (Hordeum vulgare L.): plant growth and stress-related gene transcription. Biol Plant 59(3):537–546
Fujita Y, Fujita M, Shinozaki K, Yamaguchi-Shinozaki K (2011) ABA-mediated transcriptional regulation in response to osmotic stress in plants. J Plant Res 124:509–525
Furlan AL, Eliana B, Carmen TM, Castro S, Dietz K-J (2014) Antioxidant enzyme activities and gene expression patterns in peanut nodules during a drought and rehydration cycle. Funct Plant Biol 41:704–713
Gadjev I, Stone JM, Gechev TS (2008) Programmed cell death in plants: new insights into redox regulation and the role of hydrogen peroxide. Int Rev Cell Mol Biol 270:87–144
Gechev TS, Hille J (2005) Hydrogen peroxide as a signal controlling plant programmed cell death. J Cell Biol 168:17–20
Grillo S, Leone A (1996) Physical stresses in plants: genes and their products for tolerance. Springer, Berlin
Grzesiak M, Filek M, Barbasz A, Kreczmer B, Hartikainen H (2013) Relationships between polyamines, ethylene, osmoprotectants and antioxidant enzymes activities in wheat seedlings after shortterm PEG- and NaCl-induced stresses. Plant Growth Regul 69:177–189
Guo P, Baum M, Grando S, Ceccarelli S, Bai G, Li R, von Korff M, Varshney RK, Graner A, Valkoun J (2009) Differentially expressed genes between drought-tolerant and drought-sensitive barley genotypes in response to drought stress during the reproductive stage. J Exp Bot 60:3531–3544
Habte E, Müller LM, Shtaya M, Davis SJ, von Korff M (2014) Osmotic stress at the barley root affects expression of circadian clock genes in the shoot. Plant Cell Environ 37:1321–1337
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Calif Agric Exp Stn Circ 347:1–32
Janska A, Hodek J, Svoboda P, Zamecnik J, Prasil IT, Vlasakova E, Milella L, Ovesna J (2013) The choice of reference gene set for assessing gene expression in barley (Hordeum vulgare L.) under low temperature and drought stress. Mol Genet Genomics 288(11):639–649
Kocheva KV, Georgiev GI, Kochev VK (2014) An improvement of the diffusion model for assessment of drought stress in plant tissues. Physiol Plant 150:88–94
Li J, Wang X, Zhang Y, Jia H, Bi Y (2011) cGMP regulates hydrogen peroxide accumulation in calcium-dependent salt resistance pathway in Arabidopsis thaliana roots. Planta 234:709–722
McDonald AJS, Davies WJ (1996) Keeping in touch: responses of the whole plant to deficits in water and nitrogen supply. Adv Bot Res 22:229–300
Miller G, Suzuki N, Ciftci-Yilmaz S, Mittler R (2010) Reactive oxygen species homeostasis and signalling during drought and salinity stresses. Plant Cell Environ 33:453–467
Mir RR, Zaman-Allah M, Sreenivasulu N, Trethowan R (2012) Integrated genomics, physiology and breeding approaches for improving drought tolerance in crops. Theor Appl Genet 125:625–645
Ozturk ZN, Talame V, Deyholos M, Michalowski CB, Galbraithe DW, Gözükırmızı N, Tuberosa R, Bohnert HJ (2002) Monitoring large-scale changes in transcript abundance in drought- and salt-stressed barley. Plant Mol Biol 48:551–573
Ramanjulu S, Bartels D (2002) Drought- and desiccation-induced modulation of gene expression in plants. Plant Cell Environ 25:141–151
Rezaei MK, Shobbar ZS, Shahbazi M, Abedini R, Zare S (2013) Glutathione S-transferase (GST) family in barley: identification of members, enzyme activity, and gene expression pattern. J Plant Physiol 170:1277–1284
Richards SL, Wilkins KA, Swarbreck SM, Anderson AA, Habib N, Smith AG, McAinsh M, Devies JM (2014) The hydroxyl radical in plants: from seed to seed. J Exp Bot 66:37–46
Ristic Z, Jenks MA (2002) Leaf cuticle and water loss in maize lines differing in dehydration avoidance. J Plant Physiol 159:645–651
Rollins JA, Habte E, Templer SE, Colby T, Schmidt J, von Korff M (2013) Leaf proteome alterations in the context of physiological and morphological responses to drought and heat stress in barley (Hordeum vulgare L.). J Exp Bot 64:3201–3212
Rushton PJ, Somssich IE, Ringler P, Shen QJ (2010) WRKY transcription factors. Trends Plant Sci 15:247–258
Shao HB, Chu LY, Jaleel CA, Manivannan P, Panneerselvam R, Shao MA (2009) Understanding water deficit stress-induced changes in the basic metabolism of higher plants—biotechnologically and sustainably improving agriculture and the ecoenvironment in arid regions of the globe. Crit Rev Biotechnol 29:131–151
Suprunova T, Krugman T, Fahima T, Chen G, Shams I, Korol A, Nevo E (2004) Differential expression of dehydrin genes in wild barley, Hordeum spontaneum, associated with resistance to water deficit. Plant Cell Environ 27:1297–1308
Talame V, Ozturk NZ, Bohnert HJ, Tuberosa R (2007) Barley transcript profiles under dehydration shock and drought stress treatments: a comparative analysis. J Exp Bot 58:229–240
Teulat B, Monneveux P, Wery J, Borries J, Souyris I, Charrier A, This D (1997) Relationships between relative water content and growth parameters under water stress in barley: a QTL study. New Phytol 137:99–107
Walia H, Wilson C, Wahid A, Condamine P, Cui X, Close TJ (2006) Expression analysis of barley (Hordeum vulgare L.) during salinity stress. Funct Integr Genomics 6:143–156
Xu Z, Zhou G, Shimizu H (2010) Plant responses to drought and rewatering. Plant Signal Behav 5:649–654
Xue GP, Loveridge CW (2004) HvDRF1 is involved in abscisic acid-mediated gene regulation in barley and produces two forms of AP2 transcriptional activators, interacting preferable with a CT-rich element. Plant J 37:326–339
Yang S, Huang C, Wu Z (2006) Stomatal movement in response to long distance-communicated signals initiated by heat shock in partial roots of Commelina communis L. Sci China Ser C Life Sci 49:18–25
Acknowledgments
This work was supported by The Scientific Research Projects Coordination Unit of Istanbul University, Project Number BAP 4712. The authors thank to the staff of Dr. Maria von Korff’s lab for experimental advice on stress applications and qRT-PCR.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Table S1
Primer sequences used for gene expression analysis by semiquantitative RT-PCR and qRT-PCR (DOC 42 kb)
Table S2
Percent change in relative water content (RWC) and water loss rate (WLR, gh−1 g−1 dry weight) in ten barley cultivars at the end of 10 days of dehydration. Values are given as mean ± SD (n = 5). Different letters indicate significant differences at P ≤ 0.05 using least square means (DOCX 13 kb)
Table S3
Bivariate correlation coefficients (R) between physiological parameters and gene expressions of HvBAS1, HvMT2 and HvABA7 during shock-like dehydration (ShD) treatment, 0–8 h period, in MR and ER genotypes. IL: Ion leakage, Osmol: osmolality. Significance (*P < 0.05, **P < 0.01) (DOCX 17 kb)
Rights and permissions
About this article
Cite this article
Gürel, F., Öztürk, N.Z., Yörük, E. et al. Comparison of expression patterns of selected drought-responsive genes in barley (Hordeum vulgare L.) under shock-dehydration and slow drought treatments. Plant Growth Regul 80, 183–193 (2016). https://doi.org/10.1007/s10725-016-0156-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10725-016-0156-0