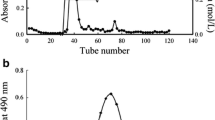
Abstract
Two extracellular polysaccharides, designated as WPA and WPB, were isolated from the fungus Aspergillus aculeatus using Q-Sepharose fast flow and Sephacryl S-300 column chromatography. WPA composed of mannose and galactose in a molar ratio of 3.9:1.0, and WPB mainly contained mannose. The molecular weight of WPA and WPB was about 28.1 kDa and 21.0 kDa, respectively. On the basis of methylation and NMR analysis, the possible main chain of WPA was [→5)-β-D-Galf-(1 → 2,6)-α-D-Manp(1→], and WPB was mainly [→2,6)-α-D-Manp(1→], both with [α-D-Manp(1 → 2)-α-D-Manp(1 → 2)-α-D-Manp(1→] substituted at C-2 of [→2,6)-α-D-Manp(1→]. Meanwhile, WPA displayed a stronger anti-proliferative effect than WPB on HeLa, MCF-7 and MGC-803 cells in vitro. WPA and WPB could arrest HeLa cells in G2/M phase and induce HeLa cells apoptosis. Thus, our study provides evidence that WPA and WPB may be taken as potential candidates for treating cervical carcinoma.






Similar content being viewed by others
References
Seviour R.J., Stasinopoulos S.J., Auer D.P.F., Gibbs P.A.: Production of pullulan and other exopolysaccharides by filamentous fungi. Crit. Rev. Biotechnol. 12, 279–298 (1992)
Bae J.S., Jang K.H., Jin H.K.: Effects of natural polysaccharides on the growth and peritoneal carcinomatosis of human gastric adenocatcinoma in a nude mouse model. Cancer Lett. 235, 60–68 (2006)
Chen G.C., Zhang P.Y., Huang T.T., Yu W.Q., Lin J., Li P., Chen K.S.: Polysaccharides from Rhizopus nigricans mycelia induced apoptosis and G2/M arrest in BGC-823 cells. Carbohydr. Polym. 97, 800–808 (2013)
Pei J.J., Wang Z.B., Ma H.L., Yan J.K.: Structural features and antitumor activity of a novel polysaccharide from alkaline extract of Phellinus linteus mycelia. Carbohydr. Polym. 115, 472–477 (2015)
Meng L., Sun S.S., Li R., Shen Z.P., Wang P., Jiang X.L.: Antioxidant activity of polysaccharides produced by Hirsutella sp. and relation with their chemical characteristics. Carbohyd. Polym. 117, 452–457 (2015)
Zhang B.Z., Inngjerdingen K.T., Zou Y.F., Rise F., Michaelsen T.R., Yan P.S., Paulsenm B.S.: Characterization and immunomodulating activities of exo-polysaccharides from submerged cultivation of Hypsizigus marmoreus. Food Chem. 163, 120–128 (2014)
Sun Y., Sun T.W., Wang F., Zhang J., Li C., Chen X.N., Li Q., Sun S.B.: A polysaccharide from the fungi of Huaier exhibits anti-tumor potential and immunomodulatory effects. Carbohydr. Polym. 92, 577–582 (2013)
Gómez-Miranda, B, Prieto, A, Leal, J.A., Ahrazem, O, Jiménez-Barbero, J, Bernabé, M. Differences among the cell wall galactomannans from Aspergillus wentii and Chaetosartorya chrysella and that of Aspergillus fumigatus. Glycoconj. J. 20, 239–246(2004)
Choma A., Wiater A., Komaniecka I., Paduch R., Pleszczyńska M., Szczodrak J.: Chemical characterization of a water insoluble (1 → 3)-α-D-glucan from an alkaline extract of Aspergillus wentii. Carbohydr. Polym. 91, 603–608 (2013)
Jin X.C., Zhao S.J.: Extraction optimization and bioactivities of an extracellular polysaccharide produced by Aspergillus fumigatus. Int. J. Biol. Macromol. 68, 13–17 (2014)
Sun H.H., Mao W.J., Chen Y., Guo S.D., Li H.Y., Qi X.H., Chen Y.L., Xu J.: Isolation, chemical characteristics and antioxidant properties of the polysaccharides from marine fungus Penicillium sp. F23–2. Carbohydr. Polym. 78, 117–124 (2009)
Wang C.Y., Mao W.J., Chen Z.Q., Zhu W.M., Chen Y.L., Zhao C.Q., Li N., Yan M.X., Liu X., Guo T.T.: Purification, structural characterization and antioxidant property of an extracellular polysaccharide from Aspergillus terreus. Process Biochem. 48, 1395–1401 (2013)
Matthaei J.H., Jones O.W., Martin R.G., Nirenberg M.W.: Characteristics and composition of RNA coding units. P. Natl. Acad. Sci USA. 48, 666–667 (1962)
Dubois M., Gilles K.A., Hamilton J.K., Rebers P.A., Smith F.: Colorimetric method for determination of sugars and related substances. Anal. Chem. 28, 350–356 (1956)
Bitter T., Muir H.M.: A modified uronic acid carbazole reaction. Anal. Biochem. 4, 330–334 (1962)
Bradford M.M.: A rapid and sensitive method for the quantization of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976)
Hakomori S.: A rapid permethylation of glycolipid, and polysaccharide catalyzed by methylsulfinyl carbanion in dimethyl sulfoxide. J. Biochem. 55, 205–208 (1964)
Mosman T.: Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. Immunol. Methods. 65, 55–63 (1983)
Ikuta K., Shibata N., Blake J.S., Dahl M.V., Nelson R.D., Hisamichi K., Kobayashi H., Suzuki S., Okawa Y.: NMR study of the galactomannans of Trichophyton mentagrophytes and Trichophyton rubrum. Biochem. J. 323, 297–305 (1997)
Ahrazem O., Prieto A., Leal J.A., Jiménez-Barbero J., Bernabé M.: Fungal cell-wall galactomannans isolated from Geotrichum spp. and their teleomorphs, Dipodascus and Galactomyces. Carbohydr. Res. 337, 2347–2351 (2002)
Bernabé M., Salvachúa D., Jiménez-Barbero J., Leal J.A., Prieto A.: Structures of wall heterogalactomannans isolated from three genera of entomopathogenic fungi. Fungal Biol. 115, 862–870 (2011)
Prieto, A., Leal, J.A., Povera, A., Jiménez-Barbero, J., Gόmez-Miranda, B., Domenech, K., A hrazem, O., Bernabé, M. Structure of complex cell wall polysaccharides isolated from Trichoderma and Hypocrea species. Carbohyd. Res. 304, 281–291 (1997)
Giménez-Abián M.I., Bernabé M., Leal J.A., Jiménez-Barbero J., Prieto A.: Structure of a galactomannan isolated from the cell wall of the fungus Lineolata rhizophorae. Carbohydr. Res. 342, 2599–2603 (2007)
Domenech J., Prieto A., Gómez-Miranda B., Leal J.A., Ahrazem O., Jiménez-Barbero J., Bernabé M.: Structure of fungal polysaccharides isolated from the cell-wall of three strains of Verticillium fungicola. Carbohydr. Polym. 50, 209–212 (2002)
Chen, Y, Mao W.J., Wang, B.F., Zhou, L.N., Gu, Q.Q., Chen, Y.L., Zhao, C.Q., Li, N, Wang, C.Y., Shan, J.M., Yan, M.X., Lin, C. Preparation and characterization of an extracellular polysaccharide produced by the deep-sea fungus Penicillium griseofulvum. Bioresour. Technol. 132, 178–181 (2013)
Omarsdottir S., Petersen B.O., Paulsen B.S., Togola A., Duus J.D., Olafsdottir E.S.: Structural characterisation of novel lichen heteroglycans by NMR spectroscopy and methylation analysis. Carbohydr. Res. 341, 2449–2455 (2006)
He M.W., Yang X.B., Jiao Y.B., Tiao L.M., Zhao Y.: Characterization of antioxidant and antiproliferative acidic polysaccharides from Chinese wolfberry fruits. Food Chem. 133, 978–989 (2012)
Xie J.H., Liu X., Shen M.Y., Nie S.P., Zhang H., Li C., Gong D.M., Xie M.Y.: Purification, physicochemical characterisation and anticancer activity of a polysaccharide from Cyclocarya paliurus leaves. Food Chem. 136, 1453–1460 (2013)
Zhu Q.Q., Jiang Y.M., Lin S., Wen L.R., Wu D., Zhao M.M., Chen F., Jia Y.X., Yang B.: Structural identification of (1 → 6)-α-D-Glucan, a key responsible for the health benefits of Longan, and evaluation of antitumor activity. Biomacromolecules. 14, 1999–2003 (2013)
Khammanit R., Chantakru S., Kitiyanant Y., Saikhun J.: Effect of serum starvation and chemical inhibitors on cell cycle synchronization of canine dermal fibroblasts. Theriogenology. 70, 27–34 (2008)
Zhang E.M., Li X.L., Zhang S.F., Chen L.Q., Zheng X.X.: Cell cycle synchronization of embryonic stem cells: Effect of serum deprivation on the differentiation of embryonic bodies in vitro. Biochem. Bioph. Res. Co. 333, 1171–1177 (2005)
Zong A.Z., Cao H.Z., Wang F.S.: Antitumor polysaccharides from natural resources: A review of recent research. Carbohydr. Polym. 90, 1395–1410 (2012)
Ren L., Perera C., Hemar Y.: Antitumor activity of mushroom polysaccharides: A review. Food Funct. 3, 1118–1130 (2012)
Nie S.P., Zhang H., Li W.J., Xie M.Y.: Current development of polysaccharides from Ganoderma: isolation, structure and bioactivities. Bioactive carbohydrates and Dietary Fibre. 1, 10–20 (2013)
Mei Y.X., Zhu H., Hu Q.M., Liu Y.Y., Zhao S.M., Peng N., Liang Y.X.: A novel polysaccharide from mycelia of cultured Phellinus linteus displays antitumor activity through apoptosis. Carbohydr. Polym. 124, 90–97 (2015)
Li J.E., Cui S.W., Nie S.P., Xie M.Y.: Structure and biological activities of a pectic polysaccharide from Mosla chinensis Maxim. cv. Jiangxiangru. Carbohyd. Polym. 105, 276–284 (2014)
Zhu Z.Y., Pang W., Li Y.Y., Ge X.R., Chen L.J., Liu X.C., Lv Q., Dong G.L., Liu A.J., Zhang Y.M.: Effect of ultrasonic treatment on structure and antitumor activity of mycelial polysaccharides from Cordyceps gunnii. Carbohydr. Polym. 114, 12–20 (2014)
Yu J., Sun R.L., Zhao Z.Q., Wang Y.Y.: Auricularia polytricha polysaccharides induce cell cycle arrest and apoptosis in human lung cancer A549 cell. Int. J. Biol. Macromol. 68, 67–71 (2014)
Zhang M., Cheung P.C.K., Chiu L.C.M., Wong E.Y.L., Ooi V.E.C.: Cell-cycle arrest and apoptosis induction in human breast carcinoma MCF-7 cells by carboxymethylated β-glucan from the mushroom sclerotia of Pleurotus tuber-regium. Carbohydr. Polym. 66, 455–462 (2006)
Jin X.C., Ning Y.: Extraction optimization and bioactivity of polysaccharides from Aspergillus fumigatus AF1. Carbohydr. Polym. 96, 411–416 (2013)
Li X., Jiao L.L., Zhang X., Tian W.M., Chen S., Zhang L.P.: Structure of polysaccharides from mycelium and culture medium of Phellinus nigricans using submerged fermentation. Sci. China Ser. C Life Sci. 51, 513–519 (2008)
Acknowledgments
This work was supported by financial support from Hebei University, Natural Science Foundation of Hebei province in China (B2015201069), Youth Fund Project of Hebei Education Department (QN2014131), and Bureau of Baoding city science and technology project (14ZF083).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Electronic supplementary material
Supplementary Fig. S1
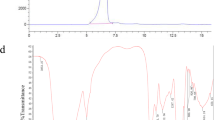
NMR spectra of WPA and WPB. (a) 1H-1H COSY spectrum, (b) 1H-13C HMQC spectrum, (c) 1H-1H NOESY spectrum and (d) 1H-13C HMBC spectrum. A: [→2)-α-D-Manp(1→], B: [→2)-α-D-Manp(1→], C: [→2,6)-α-D-Manp(1→], D: [α-D-Manp(1→], E: [→5)-β-D-Galf(1→] (PDF 313 kb)
Supplementary Fig. S2
Cell cycle synchronization of HeLa cells with or without 10 % FBS for 24 h. (PDF 27 kb)
Rights and permissions
About this article
Cite this article
Li, H., Liu, X., Xu, Y. et al. Structure and antitumor activity of the extracellular polysaccharides from Aspergillus aculeatus via apoptosis and cell cycle arrest. Glycoconj J 33, 975–984 (2016). https://doi.org/10.1007/s10719-016-9717-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-016-9717-8