Abstract
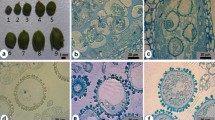
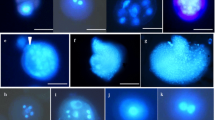
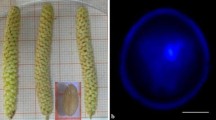
Microspore culture technique is widely used in Brassicas for development of haploids and doubled haploids (DH) in breeding programmes and genetic studies. Indian cauliflower, a group of vegetable Brassica; with tolerance to higher temperature and moisture, has been selected in India for last 200 years. In India, cauliflowers are grown throughout the year in different climatic zones based on their temperature adaptation. For the first time, we report microspore embryogenesis in different maturity groups of Indian cauliflowers. The main objective of our study was to optimize protocol for efficient microspore embryogenesis in different types of cauliflower cultivated in India. We studied different factors like bud size, developmental stage of the microspores, culture density and heat shock treatments for efficient microspore embryogenesis in four different genotypes representing four different maturity groups. Among different factors, the bud size and microspore developmental stage was found to be more critical for successful microspore embryogenesis. The optimum bud size for high frequency microspore embryogenesis for the two early maturity groups (September–October maturity) was 4.0–4.5 mm, whereas, it was 4.5–5.0 mm for the cultivars grown for November onwards harvesting. These optimized bud sizes contained the highest percent of viable microspores and maximum number of microspores at late uninucleate to early binucleate stage. The microspore density of 6–8 × 104 cells per mL was found most suitable for efficient microspore embryogenesis. A post-culture incubation of microspores at 30.0 °C/32.5 °C for 24 h followed by maintenance at 25 °C was found optimum for induction of microspore embryos in different maturity group of Indian cauliflower. The flow cytometry analysis of 646 microspore derived plants revealed that more than 60 % plants were spontaneous diploids and less than 15 % were haploids. However, the genotypes from different groups showed varied response in different culture density and temperature treatments. Thus, it is necessary to optimize genotype specific protocol for efficient microspore embryogenesis before its routine application. The protocol developed in this study will facilitate doubled haploids based breeding and genetic studies of all types of cauliflower grown in India.



Similar content being viewed by others
References
Abraha E, Bechyn M, Klıma M, Vyvadilova M (2008) Analysis of factors affecting embryogenesis in microspore cultures of Brassica carinata. Agricultura Tropica et Subtropica 41:53–59
Agarwal PK, Agarwal P, Custers JBM, Liu CM, Bhojwani SS (2006) PCIB, an antiauxin, enhances microspore embryogenesis in microspore culture of Brassica juncea. Plant Cell Tissue Organ Cult 86:201–210
Ali MM, Mian MAK, Custers JBM, Khuram MMH (2008) Microspore culture and the performance of microspore derived doubled haploid in Brassica juncea (L.). Bangladesh J Agric Res 33:571–578
Barro F, Martin A (1999) Response of different genotypes of Brassica carinata to microspore culture. Plant Breed 118:79–81
Chan J, Pauls KP (2007) Brassica napus Rop GTPases and their expression in microspore cultures. Planta 225:469–484
Chanana NP, Dhawan V, Bhojwani SS (2005) Morphogenesis in isolated microspore cultures of Brassica juncea. Plant Cell Tissue Organ Cult 83:169–177
Cousin A, Nelson MN (2009) Twinned microspore-derived embryos of canola (Brassica napus L.) are genetically identical. Plant Cell Rep 28:831–835
Custers JBM (2003) Microspore culture in rapeseed (Brassica napus L.). In: Maluszynski M, Kasha KJ, Forster BP, Szarejko I (eds) Doubled haploid production in crop plants: a manual. Kluwer, Dordrecht, pp 185–194
Dey SS, Sharma SR, Parkash C, Barwal RN, Bhatia R (2011a) Genetic divergence in snowball cauliflower (Brassica oleracea var. botrytis L.). Indian J Plant Genetic Resour 24:48–51
Dey SS, Sharma SR, Parkash C, Barwal RN, Bhatia R (2011b) Superior Ogura based CMS lines with better combining ability improve yield and earliness in cauliflower (Bassica oleracea var. botrytis L.). Euphytica 182:187–197
Dias JCS (1999) Effect of activated charcoal on Brassica oleracea microspore culture embryogenesis. Euphytica 108:65–69
Duijs JG, Voorrips RE, Visser DL, Custers JBM (1992) Microspore culture is successful in most crop types of Brassica oleracea L. Euphytica 60:45–55
Dunwell JM (2010) Haploids in flowering plants: origins and exploitation. Plant Biotechnol J 8:377–424
Esteves P, Clermont I, Marchand S, Belzile F (2014) Improving the efficiency of isolated microspore culture in six-row spring barley: II-exploring novel growth regulators to maximize embryogenesis and reduce albinism. Plant Cell Rep 33:871–879
Ferrie AMR, Caswell KL (2011) Isolated microspore culturete chniques and recent progress for haploid and doubled haploid plant production. Plant Cell Tissue Org Cult 104:301–309
Ferrie AMR, Mollers C (2011) Haploids and doubled haploids in Brassica spp. for genetic and genomic research. Plant Cell Tissue Organ Cult 104:375–386
Forster BP, Thomas WTB (2005) Doubled haploids in genetics andplant breeding. Plant Breed Rev 25:57–88
Gamborg OL, Miller RA, Ojima K (1968) Nutrient requirements of suspension cultures of soybean root cells. Exp Cell Res 50:151–158
Gu HH, Hagberg P, Zhou WJ (2004) Cold pretreatment enhances microspore embryogenesis in oilseed rape (Brassica napus L.). Plant Growth Regul 42:137–143
Gu H, Zhao Z, Sheng X, Yu H, Wang J (2014) Efficient doubled haploid production in microspore culture of loose-curd cauliflower (Brassica oleracea var. botrytis). Euphytica 195:467–475
Guo YD, Pulli S (1996) High-frequency embryogenesis in Brassica campestris microspore culture. Plant Cell Tissue Organ Cult 46:219–225
Halkjaer M, Ringgaard S (1997) Microspore culture in B. oleracea L. In: COST-824 gametic embryogenesis workshop, Book of Abstracts. Sjusjoen, p. 32
Hansen M and Elameen E (1997) Application of microspore culture in Norwegian cabbage breeding programme. In: COST-824 gametic embryogenesis workshop, Book of Abstracts. Sjusjoen, p. 14
Huang B, Bird S, Kemble R, Simmonds D, Keller WA, Miki B (1990) Effects of culture density, conditioned medium and feeder cultures on microspore embryogenesis in Brassica napus. Plant Cell Rep 8:594–597
Kott LS, Polsoni L, Ellis B, Beversdorf WD (1988) Autotoxicity in isolated microspore cultures of Brassica napus. Can J Bot 66:1665–1670
Lemonnier-Le Penhuizic C, Chatelet C, Kloareg B, Potin P (2001) Carrageenan oligosaccharides enhance stress-induced microspore embryogenesis in Brassica oleracea var italica. Plant Sci 160:1211–1220
Li JW, Si SW, Cheng JY, Li JX, Liu JQ (2013) Thidiazuron and silver nitrate enhanced gynogenesis of unfertilized ovule cultures of Cucumis sativus. Biol Plant 57:164–168
Prem D, Gupta K, Sarkar G, Agnihotri A (2008) Activated charcoal induced high frequency microspore embryogenesis and efficient doubled haploid production in Brassica juncea. Plant Cell Tissue Organ Cult 93:269–282
Prem D, Solís MT, Bárány I, Rodríguez-Sanz H, Risueño MC, Testillano PS (2012) A new microspore embryogenesis system under low temperature which mimics zygotic embryogenesis initials, expresses auxin and efficiently regenerates doubled-haploid plants in Brassica napus. BMC Plant Biol 12:127
Roberts AR (2007) The use of bead beating to prepare suspensions of nuclei for flow cytometry from fresh leaves, herbarium leaves, petals and pollen. Cytometry A 71:1039-1044
Shumilina DV, Shmykova NA, Bondareva Suprunova TP (2015) Effect of genotype and medium culture content on microspore derived embryo formation in Chinese cabbage (Brassica rapa ssp. Chinensis cv. Lastochka). Biology Bulletin 42:302–309
Simmonds DH, Keller WA (1999) Significance of preprophase strands of microtubules in the induction of microspore embryogenesis of Brassica napus. Planta 208:383–391
Supena EDJ, Suharsono S, Jacobsen E, Custers JBM (2006) Successful development of a shed-microspore culture protocol for doubled haploid production in Indonesian hot pepper (Capsicum annuum L.). Plant Cell Rep 25:1–10
Supena EDJ, Winarto B, Riksen T, Dubas E, van Lammeren A, Offringa R, Boutilier K, Custers JBM (2008) Regeneration of zygotic-like microspore-derived embryos suggests an important role for the suspensor in early embryo patterning. J Exp Bot 59:803–814
Swarup V, Chatterjee SS (1972) Origin and improvement of Indian cauliflower. Econ Bot 26:381–393
Takahata Y, Keller WA (1991) High frequency embryogenesis and plant regeneration in isolated microspore culture of Brassica oleracea L. Plant Sci 74(2):235–242
Takahata Y, Takani Y, Kaizuma N (1993) Determination of microspore population to obtain high frequency embryogenesis in broccoli (Brassica oleracea L.). Plant Tissue Cult Lett 10:49–53
Takahira J, Cousin A, Nelson MN, Cowling WA (2011) Improvement in efficiency of microspore culture to produce doubled haploid canola (Brassica napus L.) by flow cytometry. Plant Cell Tissue Organ Cult 104:51–59
Telmer CA, Simmonds DH, Newcomb W (1992) Determination of developmental stage to obtain high frequencies of embryogenic microspores in Brassica napus. Physiol Plant 84:417–424
Tian H, Yao CY, Sun MX (2004) High frequency conversion of microspore-derived embryos of Brassica napus cv. Topas by supplemental calcium and vitamins. Plant Cell Tissue Organ Cult 76:159–165
Touraev A, Vincente O, Heberle-Bors E (1997) Initiation of microspore embryogenesis by stress. Trends Plant Sci 2:297–302
Wan GL, Naeem MS, Geng XX, Xu L, Li B, Jilani G, Zhou WJ (2011) Optimization of microspore embryogenesis and plant regeneration protocols for Brassica napus. Int J Agric Biol 13:83–88
Wang TT, Li HX, Zhang J, Ouyang B, Lu Y, Ye Z (2009) Initiation and development of microspore embryogenesis in recalcitrant purple flowering stalk (Brassica campestris ssp. chinensis var. purpuIrea Hort) genotypes. Sci Hort 121:419–424
Winarto B, Teixeira da Silva JA (2011) Microspore culture protocol for Indonesian Brassica oleracea. Plant Cell Tiss Organ Cult 107:305–315
Yang H, Xue-ling Y, Hui F, Hong L, Ya-nan R (2014) Improved efficiency of microspore culture of Brassica campestris ssp. Pekinensis (Chinese cabbage). Afr J Agric Res 9:2111–2118
Yeung EC (1995) Structural and developmental patterns in somatic embryogenesis. In: Thorpe TA (ed) In vitro embryogenesis in plants. Kluwer, Dordrecht, pp 205–247
Zhang W, Fu Q, Dai X, Bao M (2008) The culture of isolated microspores of ornamental kale (Brassica oleracea var. acephala) and the importance of genotype to embryo regeneration. Sci Hort 117:69–72
Acknowledgments
We are highly thankful to Science and Engineering Research Board, Department of Science and Technology, Govt. of India for financial assistance in carrying out this research work. We are also thankful to Dr. Sashi Bhusan, CSIR-IHBT, Palampur for his support and help for FCM analysis. We are also grateful to Prof. Wallace A. Cowling and Mrs. Anouska Cousin, University of Western Australia for technical help and suggestions during the experiments.
Funding was provided by Science and Engineering Research Board (Grant No. SB/FT/LS-236/2012).
Author information
Authors and Affiliations
Corresponding author
Additional information
R. Bhatia and S. S. Dey have contributed equally to the work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bhatia, R., Dey, S.S., Sood, S. et al. Optimizing protocol for efficient microspore embryogenesis and doubled haploid development in different maturity groups of cauliflower (B. oleracea var. botrytis L.) in India. Euphytica 212, 439–454 (2016). https://doi.org/10.1007/s10681-016-1775-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10681-016-1775-2