Abstract
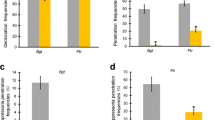
Bread wheat (BW) and durum wheat (DW) are both strongly affected by Septoria tritici blotch caused by the hemibiotrophic fungus Zymoseptoria tritici. However, only the BW-Z. tritici pathosystem has been well studied so far. Here, we compared compatible interactions between Z. tritici and both BW and DW species at the cytological, biochemical and molecular levels. Fungal infection process investigations showed close spore germination and leaf penetration features in both interactions, although differences in the patterns of these events were observed. During the necrotrophic phase, disease severity and sporulation levels were associated in both interactions with increases of the two cell-wall degrading enzyme activities endo-β-1,4-xylanase and endo-β-1,3-glucanase as well as protease. An analysis of plant defense responses during the first five days post inoculation revealed inductions of GLUC, Chi4, POX and PAL and a repression of LOX gene expressions in both wheat species, although differences in kinetics and levels of induction or repression were observed. In addition, peroxidase, catalase, glucanase, phenylalanine ammonia-lyase and lipoxygenase activities were induced in both wheat species, while only weak accumulations of hydrogen peroxide and polyphenols were detected at the fungal penetration sites. Our study revealed overall a similarity in Z. tritici infection process and triggered wheat defense pathways on both pathosystems.







Similar content being viewed by others
References
Adhikari, T. B., Balaji, B., Breeden, J., & Goodwin, S. B. (2007). Resistance of wheat to Mycosphaerella graminicola involves early and late peaks of gene expression. Physiological and Molecular Plant Pathology, 71, 55–68.
Berraies, S., Gharbi, M. S., Rezgui, S., & Yahyaoui, A. (2014). Estimating grain yield losses caused by septoria leaf blotch on durum wheat in Tunisia. Chilean Journal of Agricultural Research, 74(4), 432–437.
Bradford, M. M. (1976). A rapid and sensitive method for quantitation of microgram quantities of protein utilizing the principle of protein-dye-binding. Analytical Biochemistry, 72, 248–254.
Cohen, L., & Eyal, Z. (1993). The histology of processes associated with the infection of resistant and susceptible wheat cultivars with Septoria tritici. Plant Pathology, 42, 737–743.
Cohen-Kupiec, R., & Chet, I. (1998). The molecular biology of chitin digestion. Current Opinion of Biotechnology, 9, 270–277.
Deller, S., Hammond-Kosack, K. E., & Rudd, J. J. (2011). The complex interactions between host immunity and non-biotrophic fungal pathogens of wheat leaves. Journal of Plant Physiology, 168, 63–71.
Diani, Z., Ouarraqi, E. M., Aissam, S., Hsissou, D., & Modafar, C. E. (2009). Induction of early oxidative events in soft wheat leaves inoculated with Septoria tritici and their relationship to resistance of Moroccan cultivars. International Journal of Agriculture and Biology, 11, 351–359.
Douaiher, M. N., Nowak, E., Durand, R., Halama, P., & Reignault, P. (2007). Correlative analysis of Mycosphaerella graminicola pathogenicity and cell wall degrading enzymes produced in vitro: the importance of xylanases and polygalacturonases. Plant Pathology, 56, 79–86.
Duncan, K. E., & Howard, R. J. (2000). Cytological analysis of wheat infection by the leaf blotch pathogen Mycosphaerella graminicola. Mycological Research, 104, 1074–1082.
El Hadrami, I., & Baaziz, M. (1995). Somatic embryogenesis and analysis of peroxidases in Phoenix dactylifera L. Biologia Plantarum, 37, 197–203.
El Hadrami, I., Ramos, T., El Bellaj, M., El Idrissi-Tourane, A., & Macheix, J. J. (1997). A sinapic derivative as an induced defense compound of date palm against Fusarium oxysporum f. Sp. albedinis, the agent causing bayoud disease. Journal of Phytopathology, 145, 329–333.
Eyal, Z. (1999). The septoria tritici and stagonospora nodorum blotch diseases of wheat. European Journal of Plant Pathology, 105, 629–641.
Farsad, L. K., Mardi, M., & Ebrahimi, M. A. (2013). Quantitative expression analysis of candidate genes for Septoria Tritici blotch resistance in wheat (Triticum aestivum L.). Progress in Biological Sciences, 3(1), 72–78.
Goodwin, S. B., Ben M’Barek, S., Dhillon, B., Wittenberg, A. H. J., Crane, C. F., Hane, J. K., et al. (2011). Finished genome of the fungal wheat pathogen Mycosphaerella graminicola reveals dispensome structure, chromosome plasticity, and stealth pathogenesis. PLoS Genetics, 7(6), e1002070.
Gouache, D., Bensadoun, A., Brunc, F., Pagéd, C., Makowskie, D., & Wallachf, D. (2013). Modelling climate change impact on Septoria Tritici blotch (STB) in France: accounting for climate model and disease model uncertainty. Agricultural and Forest Meteorology, 170, 242–252.
Hellweg, M. (2003). Molecular biological and biochemical studies of proteolytic enzymes of the cereal pathogen Fusarium graminearum. Bremen, Deutschland: Westfälischen Wilhelms-Universität Münster, Dissertation des Doktorgrades.
Jones, J. D., & Dang, J. L. (2006). The plant immune system. Nature, 444, 323–329.
Kema, G. H. J., Yu, D., Rijkenberg, F. H. J., Shaw, M. W., & Baayen, R. P. (1996). Histology of the pathogenesis of Mycosphaerella graminicola in wheat. Phytopathology, 86, 777–786.
Keon, J., Antoniw, J., Carzaniga, R., Deller, S., Ward, J. L., Baker, J. M., et al. (2007). Transcriptional adaptation of Mycosphaerella graminicola to programmed cell death (PCD) of its susceptible wheat host. Molecular Plant-Microbe Interactions, 20(2), 178–193.
Lee, W.-S., Rudd, J. J., Hammond-Kosack, K. E., & Kanyuka, K. (2014). Mycosphaerella graminicola LysM effector-mediated stealth pathogenesis subverts recognition through both CERK1 and CEBiP homologues in wheat. Molecular Plant-Microbe Interactions, 27(3), 236–243.
Marshall, R., Kombrink, A., Motteram, J., Loza-Reyes, E., Lucas, J., Hammond-Kosack, K. E., et al. (2011). Homologs from the fungus Mycosphaerella graminicola reveals novel functional properties and varying contributions to virulence on wheat. Plant Physiology, 156(2), 756–769.
Mauch, F., Mauch-Mani, B., & Boller, T. (1988). Antifungal hydrolases in pea tissue. II. Inhibition of fungal growth by combinations of chitinase and a β-1,3-glucanase. Plant Physiology, 88, 936–942.
Palmer, C., & Skinner, W. (2002). Mycosphaerella graminicola: latent infection, crop devastation and genomics. Molecular Plant Pathology, 3, 63–70.
Pfaffl, M. W., Horgan, G. W., & Dempfle, L. (2002). Relative expression software tool (REST©) for group-wise comparison and statistical analysis of relative expression results in real-time PCR. Nucleic Acids Research, 30(9), 36.
Quaedvlieg, W., Kema, G. H. J., Groenewald, J. Z., Verkley, G. J. M., Seifbarghi, S., Razavi, M., et al. (2011). Zymoseptoria gen. nov.: a new genus to accommodate Septoria-like species occurring on graminicolous hosts. Persoonia, 26, 57–69.
Randoux, B., Renard-Merlier, D., Mulard, G., Mulard, G., Duyme, F., Sanssené, J., et al. (2010). Distinct defenses induced in wheat against powdery mildew by acetylated and nonacetylated oligogalacturonides. Phytopathology, 100(13), 1552–1563.
Ray, S., Anderson, J. M., Urmeev, F. I., & Goodwin, S. B. (2003). Rapid induction of a protein disulfide isomerase and defense related genes in wheat in response to the hemibiotrophic fungal pathogen Mycosphaerella graminicola. Plant Molecular Biology, 53, 701–714.
Rohel, E. A., Payne, A. C., Fraaije, B. A., & Hollomon, D. W. (2001). Exploring infection of wheat and carbohydrate metabolism in Mycosphaerella graminicola transformants with differentially regulated green fluorescent protein expression. Molecular Plant-Microbe Interactions, 14, 1–7.
Rudd, J. J., Kanyuka, K., Hassani-Pak, K., Derbyshire, M., Andogabo, A., & Devonshire, J. (2015). Transcriptome and metabolite profiling the infection cycle of Zymoseptoria tritici on wheat (Triticum aestivum) reveals a biphasic interaction with plant immunity involving differential pathogen chromosomal contributions, and a variation on the hemibiotrophic lifestyle definition. Plant Physiology. doi:10.1104/pp.114.255927.
Shetty, N. P., Kristensen, B. K., Newman, M.-A., Møller, K., Gregersen, P. L., & Jørgensen, H. J. L. (2003). Association of hydrogen peroxide with restriction of Septoria tritici in resistant wheat. Physiological and Molecular Plant Pathology, 62, 333–346.
Ryals, J. A., Neuenschwander, U. H., Willits, M. G., Molina, A., Steiner, H. Y., & Hunt, M. D. (1996) Systemic acquired resistance. Plant Cell 8, 1809–1819.
Shetty, N. P., Mehrabi, R., Lütken, H., Haldrup, A., Kema, G. H. J., Collinge, D. B., et al. (2007). Role of hydrogen peroxide during the interaction between the hemibiotrophic fungal pathogen Septoria tritici on wheat. New Phytologist, 174, 637–647.
Shetty, N. P., Jensen, J. D., Knudsen, A., Finnie, C., Geshi, N., Blennow, A., et al. (2009). Effects of β-1,3-glucan from Septoria tritici on structural defense responses in wheat. Journal of Experimental Botany, 60(15), 4287–4300.
Siah, A., Deweer, C., Duyme, F., Sanssené, J., Durand, R., Halama, P., et al. (2010). Correlation of in planta endo-beta-1,4-xylanase activity with the necrotrophic phase of the hemibiotrophic fungus Mycosphaerella graminicola. Plant Pathology, 59, 661–670.
Tayeh, C., Randoux, B., Bourdon, N., & Reignault, P. (2013). Lipid metabolism is differentially modulated by salicylic acid and heptanoyl salicylic acid during the induction of resistance in wheat against powdery mildew. Journal of Plant Physiology, 170, 1620–1629.
Tayeh, C., Randoux, B., Tisserant, B., Khong, G., Jacques, P., & Reignault, P. (2015). Are ineffective defense reactions potential target for induced resistance during the compatible wheat-powdery mildew interaction? Plant Biology and Biochemistry, 96, 9–19.
Tian, S. M., Weinert, J., & Zhao, Q. H. (2009). Correlation between cell wall-degrading enzymes in wheat leaves infected by Septoria tritici and disease severity. Canadian Journal of Plant Pathology, 31, 387–392.
Weber, G. F. (1922). Septoria diseases of cereals. II. Septoria diseases of wheat. Phytopathology, 12(12), 537–585.
Yang, F., Li, W., & Jørgensen, H. J. L. (2013). Transcriptional reprogramming of wheat and the hemibiotrophic pathogen Septoria tritici during two phases of the compatible interaction. PloS One, 8(11), 1–15.
Acknowledgment
Lamia Somai-Jemmali was supported by Campus France for internships in Institut Supérieur d’Agriculture (Lille, France) and Université du Littoral Côte d’Opale (Calais, France).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Somai-Jemmali, L., Randoux, B., Siah, A. et al. Similar infection process and induced defense patterns during compatible interactions between Zymoseptoria tritici and both bread and durum wheat species. Eur J Plant Pathol 147, 787–801 (2017). https://doi.org/10.1007/s10658-016-1043-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10658-016-1043-2