Abstract
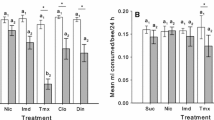
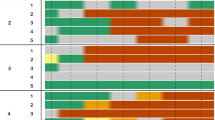
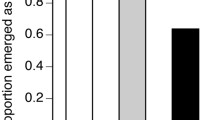
A wide application of systemic pesticides and detection of their residues in bee-collected pollen and nectar at sublethal concentrations led to the emergence of concerns about bees’ chronic exposure and possible sublethal effects on insect pollinators. Therefore, special attention was given to reducing unintentional intoxications under field conditions. The sensitivity of winter bees throughout their long lifespan to residual exposure of pesticides is not well known, since most previous studies only looked at the effects on summer bees. Here, we performed various laboratory bioassays to assess the effects of clothianidin on the survival and behavior of winter bees. Oral lethal and sublethal doses were administered throughout 12-day. The obtained LD50 values at 48, 72, 96 h and 10 days were 26.9, 18.0, 15.1 and 9.5 ng/bee, respectively. Concentrations <20 µg/kg were found to be sublethal. Oral exposure to sublethal doses was carried out for 12-day and, the behavioral functions were tested on the respective 13th day. Although slight reductions in the responses at the concentrations 10 and 15 µg/kg were observed, all tested sublethal concentrations had showed non-significant effects on the sucrose responsiveness, habitation of the proboscis extension reflex and olfactory learning performance. Nevertheless, chronic exposure to 15 µg/kg affected the specificity of the early long-term memory (24 h). Since the tested concentrations were in the range of field-relevant concentrations, our results strongly suggest that related-effects on winter and summer bees’ sensitivity should also be studied under realistic conditions.






Similar content being viewed by others
References
Aliouane Y, El Hassani AK, Gary V, Armengaud C, Lambin M, Gauthier M (2009) Sub-chronic exposure of honeybees to sublethal doses of pesticides: effects on behavior. Environ Toxicol Chem 28:113–122
Behrends A, Scheiner R (2010) Learning at old age: a study on winter bees. Front Behav Neurosci 4:15
Blacquière T, Smagghe G, Gestel CAM, Mommaerts V (2012) Neonicotinoids in bees a review on concentrations side-effects and risk assessment. Ecotoxicology 21:973–992
Brown LA, Ihara M, Buckingham SD, Matsuda K, Sattelle DB (2006) Neonicotinoid insecticides display partial and super agonist actions on native insect nicotinic acetylcholine receptors. J Neurochem 99:608–615
Cano Lozano V, Bonnard E, Gauthier M, Richard D (1996) Mecamylamine- induced impairment of acquisition and retrieval of olfactory conditioning in the honeybee. Behav Brain Res 81:215–222
Cano Lozano V, Armengaud C, Gauthier M (2001) Memory impairment induced by cholinergic antagonists injected into the mushroom bodies of the honeybee. J Comp Physiol A 187:249–254
Ceuppens B, Eeraerts M, Vleugels T, Cnops G, Roldan-Ruiz I, Smagghe G (2015) Effects of dietary lambda-cyhalothrin exposure on bumblebee survival, reproduction, and foraging behavior in laboratory and greenhouse. J Pest Sci 88:777–783
Cox DR (1972) Regression models and life tables. Biometrics 38:67–77
Cutler GC, Scott-Dupree CD (2007) Exposure to clothianidin seed-treated canola has no long-term impact on honey bees. J Econ Entomol 100:765–772
Cutler GC, Scott-Dupree CD, Sultan M, McFarlane AD, Brewer L (2014) A large- scale field study examining effects of exposure to clothianidin seed-treated canola on honey bee colony health, development, and overwintering success. PeerJ 2:e652
Dacher M, Lagarrigue A, Gauthier M (2005) Antennal tactile learning in the honeybee: effect of nicotinic antagonists on memory dynamics. Neuroscience 130:37–50
Dechaume-Moncharmont FX, Decourtye A, Hennequet-Hantier C, Pons O, Pham- Delégue M-H (2003) Statistical analysis of the honeybee survival after chronic exposure to insecticides. Environ Toxicol Chem 22:3088–3094
Decourtye A, Devillers J (2010) Ecotoxicity of neonicotinoid insecticides to bees. In: Thany SH (ed) Insect nicotinic acetylcholine receptors, 1st edn. Springer, New York, pp 85–95
Decourtye A, Lacassie E, Pham-Delegue MH (2003) Learning performances of honeybees (Apis mellifera L.) are differentially affected by imidacloprid according to the season. Pest Manag Sci 59(3):269–278
Decourtye A, Armengaud C, Renou M, Devillers J, Cluzeau S, Gauthier M, Pham- Delegue MH (2004) Imidacloprid impairs memory and brain metabolism in the honeybee (Apis mellifera L.). Pestic Biochem Phys 78:83–92
Decourtye A, Henry M, Desneux N (2013) Environment: overhaul pesticide testing on bees. Nature 497(7448):188
Desneux N, Decourtye A, Delpuech JM (2007) The sublethal effects of pesticides on beneficial arthropods. Ann Rev Entomol 52:81–106
EFSA European Food Safety Authority (2013) Conclusion on the peer review of the pesticide risk assessment for bees for the active substance clothianidin. EFSA J 11:3066
El Hassani AK, Dacher M, Gary V, Lambin M, Gauthier M, Armengaud C (2008) Effects of sublethal doses of acetamiprid and thiamethoxam on the behavior of the honeybee (Apis mellifera). Arch Environ Contam Toxicol 54:653–661
Fauser-Misslin A, Sadd BM, Neumann P, Sandrock C (2014) Influence of combined pesticide and parasite exposure on bumblebee colony traits in the laboratory. J Appl Ecol 51:450–459
Gauthier M, Dacher M, Thany SH, Niggebrugge C, Deglise P, Kljucevic P, Armengaud C, Grünewald B (2006) Involvement of alpha-bungarotoxin- sensitive nicotinic receptors in long-term memory formation in the honeybee (Apis mellifera). Neurobiol Learn Mem 86:164–174
Gill RJ, Ramos-Rodriguez O, Raine NE (2012) Combined pesticide exposure severely affects individual- and colony-level traits in bees. Nature 491:105–108
Girolami V, Mazzon L, Squartini A, Mori N, Marzaro M, Dibernardo A, Greatti M, Giorio C, Tapparos A (2009) Translocation of neonicotinoid insecticides from coated seeds to seedling guttation drops: a novel way of intoxication for bees. J Econ Entomol 102:1808–1815
Guez D, Suchail S, Gauthier M, Maleszka R, Belzunces LP (2001) Contrasting effects of imidacloprid on habituation in 7- and 8-day-old honeybees (Apis mellifera). Neurobiol Learn Mem 76:183–191
Guez D, Belzunces LP, Maleszka R (2003) Effects of imidacloprid metabolites on habituation in honeybees suggest the existence of two subtypes of nicotinic receptors differentially expressed during adult development. Pharmacol Biochem Behav 75:217–222
Halm MP, Rortais A, Arnold G, Tasei JN, Rault S (2006) New risk assessment approach for systemic insecticides: the case of honey bees and imidacloprid (Gaucho). Environ Sci Technol 40:2448–2454
Hammer M, Menzel R (1995) Learning and memory in the honeybee. J Neurosci 15:1617–1630
Han P, Niu CY, Lei CL, Cui JJ, Desneux N (2010a) Use of an innovative T-tube maze assay and the proboscis extension response assay to assess sublethal effects of GM products and pesticides on learning capacity of the honey bee Apis mellifera L. Ecotoxicology 19:1612–1619
Han P, Niu CY, Lei CL, Cui JJ, Desneux N (2010b) Quantification of toxins in a Cry1Ac+ CpTI cotton cultivar and its potential effects on the honey bee Apis mellifera L. Ecotoxicology 19:1452–1459
Jeschke P, Nauen R (2008) Neonicotinoids-from zero to hero in insecticide chemistry. Pest Manag Sci 64:1084–1098
Joachimsmeier I, Pistorius J, Schenke D, Kirchner WH (2012) Guttation and risk for honey bee colonies (Apis mellifera L.): use of guttation drops by honey bees after migration of colonies—a field study. Julius-Kuhn-Archiv 437, 2012. In: 11th international symposium of the ICP-BR Bee Protection Group, Wageningen, 2–4 Nov 2011. doi: 10.5073/jka.2012.437.016
Krupke CH, Hunt GJ, Eitzer BD, Andino G, Given K (2012) Multiple routes of pesticide exposure for honey bees living near agricultural fields. PLoS One 7:e29268
Lambin M, Armengaud C, Raymond S, Gauthier M (2001) Imidacloprid- induced facilitation of the proboscis extension reflex habituation in the honeybee. Arch Insect Biochem Physiol 48:129–134
Laurino D, Manino A, Patetta A, Porporato M (2013) Toxicity of neonicotinoid insecticides on different honey bee genotypes. Bull Insectol 66:119–126
Maus C, Curé G, Schmuck R (2003) Safety of imidacloprid seed dressings to honey bees: a comprehensive overview and compilation of the current state of knowledge. Bull Insectol 56:51–57
McCabe SI, Hartfelder K, Santana WC, Farina WM (2007) Odor discrimination in classical conditioning of proboscis extension in two stingless bee species in comparison to Africanized honeybees. J Comp Physiol A 193:1089–1099
Ramirez-Romero R, Chaufaux J, Pham-delègue M (2005) Effects of Cry1Ab protoxin, deltametrhrin and imidacloprid on the foraging activity and the learning performances of the honeybee Apis mellifera, a comparative approach. Apidologie 36:601–611
Rondeau G, Sánchez-Bayo F, Tennekes HA, Decourtye A, Ramírez-Romero R, Desneux N (2014) Delayed and time-cumulative toxicity of imidacloprid in bees, ants and termites. Sci Rep 4:5566
Rundlöf M, Andersson GKS, Bommarco R, Fries I, Hederström V, Herbertsson L, Jonsson O, Klatt BK, Pedersen TR, Yourstone J et al (2015) Seed coating with a neonicotinoid insecticide negatively affects wild bees. Nature 521:77–80
Samson-Robert O, Labrie G, Chagnon M, Fournier V (2014) Neonicotinoid- Contaminated puddles of water represent a risk of intoxication for honey bees. PLoS One 9:e108443
Sánchez-Bayo F, Hyne RV (2014) Detection and analysis of neonicotinoids in river waters - development of a passive sampler for three commonly used insecticides. Chemosphere 99:143–151
Sánchez-Bayo F, Goulson D, Pennacchio F, Nazzi F, Goka K, Desneux N (2016) Are bee diseases linked to pesticides?– A brief review. Environ Int 89–90:7–11
Schmuck R, Schöning R, Stork A, Schramel O (2001) Risk posed to honeybees (Apis mellifera L, Hymenoptera) by an imidacloprid seed dressing of sunflowers. Pest Manag Sci 57:225–238
Schneider CW, Tautz J, Grünewald B, Fuchs S (2012) RFID Tracking of sub-lethal effects of two neonicotinoid insecticides on the foraging behavior of Apis mellifera. PLoS One 7(1):e30023
Seehuus SC, Norberg K, Gimsa U, Krekling T, Amdam GV (2006) Reproductive protein protects functionally sterile honey bee workers from oxidative stress. Proc Natl Acad Sci USA 103:962–967
Suchail S, Guez D, Belzunces LP (2001) Discrepancy between acute and chronic toxicity induced by imidacloprid and its metabolites in Apis mellifera. Environ Toxicol Chem 20:2482–2486
Suchail S, Debrauwer L, Belzunces LP (2004) Metabolism of imidacloprid in Apis mellifera. Pest Manag Sci 60:291–296
Tennekes HA, Sánchez-Bayo F (2013) The molecular basis of simple relationships between exposure concentration and toxic effects with time. Toxicology 309:39–51
Thany SH, Gauthier M (2005) Nicotine injected into the antennal lobes induces a rapid modulation of sucrose threshold and improves short-term memory in the honeybee Apis mellifera. Brain Res 1039:216–219
Williamson SM, Wright GA (2013) Exposure to multiple cholinergic pesticides impairs olfactory learning and memory in honeybees. J Exp Biol 216:1799–1807
Winston ML (1987) The biology of the honey bee. Harvard University Press, Cambridge
Xu T, Dyer DG, McConnell LL, Bondarenko S, Allen R, Heinemann O (2015) Clothianidin in agricultural soils and uptake into corn pollen and canola nectar after multi-year seed treatment applications. Environ Toxicol Chem. Accepted doi:10.1002/etc.3281
Acknowledgments
We thank the team of behavioral biology and biology education at the Ruhr-University for helpful discussions. We are also grateful to Dr. Pia Aumeier for her taking care of the study apiary. Suggestions and comments by anonymous reviewers and the editor to improve the manuscript are gratefully acknowledged.
Funding
This study was not supported by any commercial company.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Alkassab, A.T., Kirchner, W.H. Impacts of chronic sublethal exposure to clothianidin on winter honeybees. Ecotoxicology 25, 1000–1010 (2016). https://doi.org/10.1007/s10646-016-1657-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-016-1657-3