Abstract
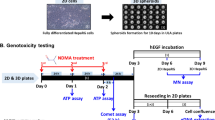
Sublethal exposure to environmental genotoxicants may impact genome integrity in affected organisms. It is therefore necessary to develop tools to measure the extent and longevity of genotoxicant-induced DNA damage, and choose appropriate model organisms for biomonitoring. To this end, markers of DNA damage were measured in zebrafish larvae and adults following exposure to model genotoxicants (benzo[a]pyrene and ethyl methanesulfonate). Specifically, we assessed primary DNA damage and the existence of potentially persistent genomic alterations through application of the comet assay, quantitative random amplified polymorphic DNA (qRAPD) and amplified fragment length polymorphism (AFLP) assays. Furthermore, expression of genes involved in DNA repair, oxidative stress response and xenobiotic metabolism was evaluated as well. Additionally, the AFLP method was applied to adult specimens 1 year after larval exposure to the genotoxicants to evaluate the longevity of the observed DNA alterations. Large numbers of DNA alterations were detected in larval DNA using the comet assay, qRAPD and AFLP, demonstrating that zebrafish larvae are a sensitive model for revealing genotoxic effects. Furthermore, some of these genomic alterations persisted into adulthood, indicating the formation of stable genomic modifications. qRAPD and AFLP methods proved to be highly sensitive to genotoxic effects, even in cases when the comet assay indicated a lack of significant damage. These results thus support the use of zebrafish larvae as a sensitive model for monitoring the impact of genotoxic insult and give evidence of the longevity of genomic modifications induced by genotoxic agents.






Similar content being viewed by others
References
Aina R, Palin L, Citterio S (2006) Molecular evidence for benzo(a)pyrene and naphthalene genotoxicity in Trifolium repens L. Chemosphere 65:666–673
Akcha F, Burgeot T, Budzinski H, Pfohl-Leszkowicz A, Narbonne J-F (2000) Induction and elimination of bulky benzo[a]pyrene-related DNA adducts and 8-oxodGuo in mussels Mytilus galloprovincialis exposed in vivo to B[a]P-contaminated feed. Mar Ecol Prog Ser 205:195–206
Atienzar FA, Jha AN (2004) The random amplified polymorphic DNA (RAPD) assay to determine DNA alterations, repair and transgenerational effects in B(a)P exposed Daphnia magna. Mutat Res 552:125–140
Atienzar FA, Jha AN (2006) The random amplified polymorphic DNA (RAPD) assay and related techniques applied to genotoxicity and carcinogenesis studies: a critical review. Mutat Res 613:76–102
Atienzar FA, Cordi B, Donkin ME, Evenden AJ, Jha AN, Depledge MH (2000) Comparison of ultraviolet-induced genotoxicity detected by random amplified polymorphic DNA with chlorophyll fluorescence and growth in a marine macroalgae, Palmaria palmate. Aquat Toxicol 50:1–12
Atienzar FA, Venier P, Jha AN, Depledge MH (2002) Evaluation of the random amplified polymorphic DNA (RAPD) assay for the detection of DNA damage and mutations. Mutat Res 521:151–163
Azqueta A, Slyskova J, Langie SA, O’Neill Gaivão I, Collins A (2014) Comet assay to measure DNA repair: approach and applications. Front Genet 5:1–8
Bagley MJ, Anderson SL, May B (2001) Choice of methodology for assessing genetic impacts of environmental stressors: polymorphism and reproducibility of RAPD and AFLP fingerprints. Ecotoxicology 10:239–244
Boettcher M, Kosmehl T, Braunbeck T (2011) Low-dose effects and biphasic effect profiles: is trenbolone a genotoxicant? Mutat Res 723:152–157
Bowditch BM, Albright DG, Williams JGK, Braun MJ (1993) Use of randomly amplified polymorphic DNA markers in comparative genome studies. Method Enzymol 224:294–309
Brevik A, Lindeman B, Brunborg G, Duale N (2012) Paternal Benzo[a]pyrene exposure modulates microRNA expression patterns in the developing mouse embryo. Int J Cell Biol. doi:10.1155/2012/407431
Cambier S, Gonzalez P, Durrieu G, Bourdineaud JP (2010) Cadmium induced genotoxicity in zebrafish at environmentally relevant doses. Ecotox Environ Safe 73:312–319
Castaño A, Becerril C (2004) In vitro assessment of DNA damage after short- and long-term exposure to benzo(a)pyrene using RAPD and the RTG-2 fish cell line. Mutat Res 552:141–151
Collins AR (2004) The Comet assay for DNA damage and repair: principles, applications, and limitations. Appl Biochem Biotech 26:249–261
Çulcu T, Sözen E, Tüylü BA (2010) Determination of genotoxicants induced DNA damage by using RAPD-PCR in human peripheral blood lymphocytes. Fresen Environ Bull 19:2205–2209
Domingues I, Oliveira R, Lourenço J, Grisolia CK, Mendo S, Soares AMVM (2010) Biomarkers as a tool to assess effects of chromium (VI): comparison of responses in zebrafish early life stages and adults. Comp Biochem Physiol 152:338–345
Fang X, Thornton C, Scheffler BE, Willett KL (2013) Benzo[a]pyrene decreases global and gene specific DNA methylation during zebrafish development. Environ Toxicol Phar 36:40–50
Fraser L, Pareek CS, Strzeżek J (2008) Identification of amplified fragment length polymorphism markers associated with freezability of boar semen—a preliminary study. Med Weter 64:646–649
Geffroy B, Ladhar C, Cambier S, Treguer-Delapierre M, Brèthes D, Bourdineaud J-P (2012) Impact of dietary gold nanoparticles in zebrafish at very low contamination pressure: the role of size, concentration and exposure time. Nanotoxicology 6:144–160
Häfeli N, Schwartz P, Burkhardt-Holm P (2011) Embryotoxic and genotoxic potential of sewage system biofilm and river sediment in the catchment area of a sewage treatment plant in Switzerland. Ecotox Environ Safe 74:1271–1279
Hande MP, Boei JJWA, Natarajan AT (1996) Induction and persistence of cytogenetic damage in mouse splenocytes following whole-body X-irradiation analysed by fluorescence in situ hybridization. II. Micronuclei. Int J Radiat Biol 70:375–383
Helleday T (2003) Pathways for mitotic homologous recombination in mammalian cells. Mutat Res 532:103–115
Hoeijmakers JHJ (2001) Genome maintenance mechanisms for preventing cancer. Nature 411:366–374
Hsu TH, Lin KH, Gwo JC (2008) Genetic integrity of black sea bream (Acanthopagrus schlegeli) sperm following cryopreservation. J Appl Ichthyol 24:456–459
Hsu T, Tsai H-T, Huang K-M, Luan M-C, Hsieh C-R (2010) Sublethal levels of cadmium down-regulate the gene expression of DNA mismatch recognition protein MutS homolog 6 (MSH6) in zebrafish (Danio rerio) embryos. Chemosphere 81:748–754
ISO (1996) International Organization for Standardization. Water quality—determination of the acute lethal toxicity of substances to a freshwater fish [Brachydanio rerio Hamilton-Buchanan (Teleostei, Cyprinidae)]. ISO 7346-3: flow-through method. http://www.iso.org
Jarvis RB, Knowles JF (2003) DNA damage in zebrafish larvae induced by exposure to low-dose rate γ radiation: detection by the alkaline comet assay. Mutat Res 541:63–69
Jin X, Chen Q, Tang S-S, Zou J-J, Chen K-P, Zhang T, Xiao X-L (2009) Investigation of quinocetone-induced genotoxicity in HepG2 cells using the comet assay, cytokinesis-block micronucleus test and RAPD analysis. Toxicol In Vitro 23:1209–1214
Kisseljova NP, Kisseljov FL (2005) DNA demethylation and carcinogenesis. Biochemistry (Moscow) 70:743–752
Kosmehl T, Krebs F, Manz W, Braunbeck T, Hollert H (2007) Differentiation between bioavailable and total hazard potential of sediment-induced DNA fragmentation as measured by the Comet assay with zebrafish embryos. J Soils Sediments 7:377–387
Kosmehl T, Hallare AV, Braunbeck T, Hollert H (2008) DNA damage induced by genotoxicants in zebrafish (Danio rerio) embryos after contact exposure to freeze-dried sediment and sediment extracts from Laguna Lake (The Philippines) as measured by the comet assay. Mutat Res 650:1–14
Labra M, Grassi F, Imazio S, Di Fabio T, Citterio S, Sgorbati S, Agradi E (2004) Genetic and DNA methylation changes induced by potassium dichromate in Brassica napus L. Chemosphere 54:1049–1058
Labra M, Bernasconi M, Grassi F, De Mattia F, Sgorbati S, Airoldi R, Citterio S (2007) Toxic and genotoxic effects of potassium dichromate in Pseudokirchneriella subcapitata detected by microscopy and AFLP marker analysis. Aquat Bot 86:229–235
Labra M, De Mattia F, Bernasconi M, Bertacchi D, Grassi F, Bruni I, Citterio S (2010) The combined toxic and genotoxic effects of chromium and volatile organic contaminants to Pseudokirchneriella subcapitata. Water Air Soil Poll 213:57–70
Lerebours A, Cambier S, Hislop L, Adam-Guillermin C, Bourdineaud J-P (2013) Genotoxic effects of exposure to waterborne uranium, dietary methylmercury and hyperoxia in zebrafish assessed by the quantitative RAPD-PCR method. Mutat Res 755:55–60
Lewis PD, Parry JM (2004) In Silico p53 mutation hotspots in lung cancer. Carcinogenesis 25:1099–1107
Liu B, Yu Z, Song X, Yang F (2010) Effects of sodium dodecylbenzene sulfonate and sodium dodecyl sulphate on the Mytilus galloprovincialis biomarker system. Ecotox Environ Safe 73:835–841
Lu G, Wu X, Chen B, Gao G, Xu K (2007) Evaluation of genetic and epigenetic modification in rapeseed (Brassica napus) induced by salt stress. J Integr Plant Biol 49:1599–1607
Miller SA, Dykes DD, Polesky HF (1988) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Notch EG, Miniutti DM, Mayer GD (2007) 17α-Ethinylestradiol decreases expression of multiple hepatic nucleotide excision repair genes in zebrafish (Danio rerio). Aquat Toxicol 84:301–309
O’Brien JM, Williams A, Gingerich J, Douglas GR, Marchetti F, Yauk CL (2013) No evidence for transgenerational genomic instability in the F1 or F2 descendants of Muta ™Mouse males exposed to N-ethyl-N-nitrosourea. Mutat Res 741–742:11–17
Oliveira R, Domingues I, Grisolia CK, Soares AMVM (2009) Effects of triclosan on zebrafish early-life stages and adults. Environ Sci Pollut Res 16:679–688
Orieux N, Cambier S, Gonzalez P, Morin B, Adam C, Garnier-Laplace J, Bourdineaud J-P (2011) Genotoxic damages in zebrafish submitted to a polymetallic gradient displayed by the Lot River (France). Ecotox Environ Safe 74:974–983
Osterauer R, Faßbender C, Braunbeck T, Köhler H-R (2011) Genotoxicity of platinum in zebrafish (Danio rerio) and ramshorn snail (Marisa cornuarietis). Sci Total Environ 409:2114–2119
Pietrasanta LI, Smith BL, MacLeod MC (2000) A novel approach for analyzing the structure of DNA modified by benzo[a]pyrene diol epoxide at single-molecule resolution. Chem Res Toxicol 13:351–355
Piraino F, Aina R, Palin L, Prato N, Sgorbati S, Santagostino A, Citterio S (2006) Air quality biomonitoring: assessment of air pollution genotoxicity in the Province of Novara (North Italy) by using Trifolium repens L. and molecular markers. Sci Total Environ 372:350–359
Powell CL, Swenberg JA, Rusyn I (2005) Expression of base excision DNA repair genes as a biomarker of oxidative DNA damage. Cancer Lett 229:1–11
Qi XM, Li PJ, Liu W, Xie LJ (2006) Multiple biomarkers response in maize (Zea mays L.) during exposure to copper. J Environ Sci-China 18:1182–1188
Ramírez MJ, Surrallés J, Puerto S, Creus A, Marcos R (1999) Low persistence of radiation-induced centromere positive and negative micronuclei in cultured human cells. Mutat Res 440:163–169
Rocco L, Frenzilli G, Fusco D, Peluso C, Stingo V (2010) Evaluation of zebrafish DNA integrity after exposure to pharmacological agents present in aquatic environment. Ecotox Environ Safe 73:1530–1536
Rocco L, Frenzilli G, Zito G, Archimandritis A, Peluso C, Stingo V (2012) Genotoxic effects in fish induced by pharmacological agents present in the sewage of some Italian water-treatment plants. Environ Toxicol 27:18–25
Segner H (2011) Reproductive and developmental toxicity in fishes. In: Gupta RC (ed) Reproductive and developmental toxicology, 1st edn. Elsevier, USA, pp 1145–1166
Silva IAL, Cancela ML, Conceição N (2012) Molecular cloning and expression analysis of xpd from zebrafish (Danio rerio). Mol Biol Rep 39:5339–5348
Singh NP, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of damage in individual cells. Exp Cell Res 175:184–191
Smith LE, Denissenko MF, Bennett WP, Li H, Amin S, Tang M-S, Pfeifer GP (2000) Targeting of lung cancer mutational hotspots by polycyclic aromatic hydrocarbons. J Natl Cancer I 92:803–811
Šrut M, Traven L, Štambuk A, Kralj S, Žaja R, Mićović Klobučar GIV (2011) Genotoxicity of marine sediments in the fish hepatoma cell line PLHC-1 as assessed by the Comet assay. Toxicol In Vitro 25:308–314
Šrut M, Štambuk A, Klobučar GIV (2013) What is Comet assay not telling us: AFLP reveals wider aspects of genotoxicity. Toxicol In Vitro 27:1226–1232
Šrut M, Bourdineaud J-P, Štambuk A, Klobučar GIV (2015) Genomic and gene expression responses to genotoxic stress in PAC2 zebrafish embryonic cell line. J Appl Toxicol. doi:10.1002/jat.3113
Štambuk A, Pavlica M, Malović L, Klobučar GIV (2008) Persistence of DNA damage in the freshwater mussel Unio pictorum upon exposure to ethyl methanesulphonate and hydrogen peroxide. Environ Mol Mutagen 49:217–225
Sussman R (2007) DNA repair capacity of zebrafish. Proc Natl Acad Sci USA 104:13379–13383
Tarantini A, Maitre A, Lefebvre E, Marques M, Marie C, Ravanat J-L, Douki T (2009) Relative contribution of DNA strand breaks and DNA adducts to the genotoxicity of benzo[a]pyrene as a pure compound and in complex mixtures. Mutat Res 671:67–75
Thurston LM, Siggins K, Mileham AJ, Watson PF, Holt WV (2002) Identification of amplified restriction fragment length polymorphism markers linked to genes controlling boar sperm viability following cryopreservation. Biol Reprod 66:545–554
Van der Veen LA, Druckova A, Riggins JN, Sorrells JL, Guengerich FP, Marnett LJ (2005) Differential DNA recognition and cleavage by EcoRI dependent on the dynamic equilibrium between the two forms of the malondialdehyde—deoxyguanosine adduct. Biochemistry 44:5024–5033
Vos P, Hogers R, Bleeker M, Reijans M, van de Lee T, Hornes M, Frijters A, Pot J, Peleman J, Kuiper M, Zabeau M (1995) AFLP: a new technique for DNA fingerprinting. Nucleic Acids Res 23:4407–4414
Zeng X, Wen J, Wan Z, Yi B, Shen J, Ma C, Tu J, Fu T (2010) Effects of Bleomycin on microspore embryogenesis in Brassica napus and detection of somaclonal variation using AFLP molecular markers. Plant Cell Tiss Org 101:23–29
Zhiyi R, Haowen Y (2004) A method for genotoxicity detection using random amplified polymorphism DNA with Danio rerio. Ecotox Environ Safe 58:96–103
Acknowledgments
The authors are grateful to the students Antonio Karaga and Aleksandar Lazić for help with the preparation and analysis of comet assay slides and to Adam Maguire for English revision of the manuscript. This study was part of a Ph.D. thesis and was conducted within the framework of Project No. 119-0982934-3110 supported by the Ministry of Science, Education and Sports of the Republic of Croatia and of program Tractifs No. ANR-07-SEST-02301 supported by the French National Research Agency. Maja Šrut was awarded a scholarship by the Croatian Science Foundation to perform a portion of the research at the University of Bordeaux, France.
Ethical standards
Experiments performed in this study comply with the current laws of Croatia.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Šrut, M., Štambuk, A., Bourdineaud, JP. et al. Zebrafish genome instability after exposure to model genotoxicants. Ecotoxicology 24, 887–902 (2015). https://doi.org/10.1007/s10646-015-1432-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10646-015-1432-x