Abstract
Background
Although several types of diet have been used in experimental steatohepatitis models, comparison of gut microbiota and immunological alterations in the gut among diets has not yet been performed.
Aim
We attempted to clarify the difference in the gut environment between mice administrated several experimental diets.
Methods
Male wild-type mice were fed a high-fat (HF) diet, a choline-deficient amino acid-defined (CDAA) diet, and a methionine-choline-deficient (MCD) diet for 8 weeks. We compared the severity of steatohepatitis, the composition of gut microbiota, and the intestinal expression of interleukin (IL)-17, an immune modulator.
Results
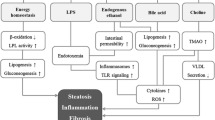
Steatohepatitis was most severe in the mice fed the CDAA diet, followed by the MCD diet, and the HF diet. Analysis of gut microbiota showed that the composition of the Firmicutes phylum differed markedly at order level between the mice fed the CDAA and HF diet. The CDAA diet increased the abundance of Clostridiales, while the HF diet increased that of lactate-producing bacteria. In addition, the CDAA diet decreased the abundance of lactate-producing bacteria and antiinflammatory bacterium Parabacteroides goldsteinii in the phylum Bacteroidetes. In CDAA-fed mice, IL-17 levels were increased in ileum as well as portal vein. In addition, the CDAA diet also elevated hepatic expression of chemokines, downstream targets of IL-17.
Conclusions
The composition of gut microbiota and IL-17 expression varied considerably between mice administrated different experimental diets to induce steatohepatitis.




Similar content being viewed by others
Abbreviations
- ALT:
-
Alanine transaminase
- CDAA:
-
Choline-deficient amino acid-defined
- F/B :
-
Firmicutes/Bacteroidetes
- HF:
-
High-fat
- H&E:
-
Hematoxylin and eosin
- IL:
-
Interleukin
- MCD:
-
Methionine-choline-deficient
- NAFLD:
-
Nonalcoholic fatty liver disease
- NASH:
-
Nonalcoholic steatohepatitis
- NC:
-
Normal chow
- SCFAs:
-
Short-chain fatty acids
- TLR:
-
Toll-like receptor
References
Okanoue T, Umemura A, Yasui K, et al. Nonalcoholic fatty liver disease and nonalcoholic steatohepatitis in Japan. J Gastroenterol Hepatol. 2011;1:153–162.
Mouzaki M, Comelli EM, Arendt BM, et al. Intestinal microbiota in patients with nonalcoholic fatty liver disease. Hepatology. 2013;58:120–127.
Miura K, Ohnishi H. Role of gut microbiota and toll-like receptors in nonalcoholic fatty liver disease. World J Gastroenterol. 2014;20:7381–7391.
Bäckhed F, Ley RE, Sonnenburg JL, et al. Host-bacterial mutualism in the human intestine. Science. 2005;307:1915–1920.
Ley RE, Turnbaugh PJ, Klein S, et al. Microbial ecology: human gut microbes associated with obesity. Nature. 2006;444:1022–1023.
Mathur R, Barlow GM. Obesity and the microbiome. Expert Rev Gastroenterol Hepatol. 2015;9:1087–1099.
Damms-Machado A, Mitra S, Schollenberger AE, et al. Effects of surgical and dietary weight loss therapy for obesity on gut microbiota composition and nutrient absorption. Biomed Res Int. 2015;2015:806248.
Huang Y, Fan XG, Wang ZM, et al. Identification of helicobacter species in human liver samples from patients with primary hepatocellular carcinoma. J Clin Pathol. 2004;57:1273–1277.
Ferolla SM, Armiliato GN, Couto CA, et al. The role of intestinal bacteria overgrowth in obesity-related nonalcoholic fatty liver disease. Nutrients. 2014;6:5583–5599.
Zhu L, Baker SS, Gill C, et al. Characterization of gut microbiomes in nonalcoholic steatohepatitis (NASH) patients: a connection between endogenous alcohol and NASH. Hepatology. 2013;57:601–609.
Okubo H, Sakoda H, Kushiyama A, et al. Lactobacillus casei strain Shirota protects against nonalcoholic steatohepatitis development in a rodent model. Am J Physiol Gastrointest Liver Physiol. 2013;305:G911–G918.
Naito E, Yoshida Y, Makino K, et al. Beneficial effect of oral administration of Lactobacillus casei strain Shirota on insulin resistance in diet-induced obesity mice. J Appl Microbiol. 2011;110:650–657.
Ivanov II, Frutos Rde L, Manel N, et al. Specific microbiota direct the differentiation of IL-17-producing T-helper cells in the mucosa of the small intestine. Cell Host Microbe. 2008;4:337–349.
Tang Y, Bian Z, Zhao L, et al. Interleukin-17 exacerbates hepatic steatosis and inflammation in nonalcoholic fatty liver disease. Clin Exp Immunol. 2011;166:281–290.
Ibrahim SH, Hirsova P, Malhi H, et al. Animal models of nonalcoholic steatohepatitis: Eat, delete, and inflame. Dig Dis Sci. 2016;61:1325–1336.
Yamaguchi K, Yang L, McCall S, et al. Inhibiting triglyceride synthesis improves hepatic steatosis but exacerbates liver damage and fibrosis in obese mice with nonalcoholic steatohepatitis. Hepatology. 2007;45:1366–1374.
Hebbard L, George J. Animal models of nonalcoholic fatty liver disease. Nat Rev Gastroenterol Hepatol. 2011;8:35–44.
Ito M, Suzuki J, Tsujioka S, et al. Longitudinal analysis of murine steatohepatitis model induced by chronic exposure to high-fat diet. Hepatol Res. 2007;37:50–57.
Miura K, Kodama Y, Inokuchi S, et al. Toll-like receptor 9 promotes steatohepatitis by induction of interleukin-1beta in mice. Gastroenterology. 2010;139:e7.
Miura K, Ishioka M, Minami S, et al. Toll-like receptor 4 on macrophage promotes the development of steatohepatitis-related hepatocellular carcinoma in mice. J Biol Chem. 2016;27:11504–11517.
Brunt EM, Kleiner DE, Wilson LA, et al. NASH clinical research network (CRN). Nonalcoholic fatty liver disease (NAFLD) activity score and the histopathologic diagnosis in NAFLD: distinct clinicopathologic meanings. Hepatology. 2011;53:810–820.
Loman NJ, Misra RV, Dallman TJ, et al. Performance comparison of benchtop high-throughput sequencing platforms. Nat Biotechnol. 2012;30:434–439.
Wright ES, Yilmaz LS, Noguera DR. DECIPHER, a search-based approach to chimera identification for 16S rRNA sequences. Appl Environ Microbiol. 2012;78:717–725.
Kitani H, Takenouchi T, Sato M, Yoshioka M, Yamanaka N. A simple and efficient method to isolate macrophages from mixed primary cultures of adult liver cells. J Vis Exp JoVE. 2011;e0000.
Petit E, LaTouf WG, Coppi MV, et al. Involvement of a bacterial microcompartment in the metabolism of fucose and rhamnose by Clostridium phytofermentans. PLoS ONE. 2013;8:e54337.
Collins MD, Lawson PA, Willems A, et al. The phylogeny of the genus Clostridium: proposal of five new genera and eleven new species combinations. Int J Syst Bacteriol. 1994;44:812–826.
Tuck LR, Altenbach K, Ang TF, et al. Insight into Coenzyme A cofactor binding and the mechanism of acyl-transfer in an acylating aldehyde dehydrogenase from Clostridium phytofermentans. Sci Rep. 2016;6:22108.
Chang CJ, Lin CS, Lu CC, et al. Ganoderma lucidum reduces obesity in mice by modulating the composition of the gut microbiota. Nat Commun. 2015;6:7489.
Neyrinck AM, Etxeberria U, Taminiau B, et al. Rhubarb extract prevents hepatic inflammation induced by acute alcohol intake, an effect related to the modulation of the gut microbiota. Mol Nutr Food Res. 2016. doi:10.1002/mnfr.201500899.
Chen Y, Yang F, Lu H, et al. Characterization of fecal microbial communities in patients with liver cirrhosis. Hepatology. 2011;54:562–572.
Chinen T, Rudensky AY. The effects of commensal microbiota on immune cell subsets and inflammatory responses. Immunol Rev. 2012;245:45–55.
Lemmers A, Moreno C, Gustot T, et al. The interleukin-17 pathway is involved in human alcoholic liver disease. Hepatology. 2009;49:646–657.
Zhang Y, Chen L, Gao W, et al. IL-17 neutralization significantly ameliorates hepatic granulomatous inflammation and liver damage in Schistosoma japonicum infected mice. Eur J Immunol. 2012;42:1523–1535.
Delzenne NM, Cani PD. Interaction between obesity and the gut microbiota: relevance in nutrition. Annu Rev Nutr. 2011;31:15–31.
Sakamoto M. Reclassification of Bacteroides distasonis, Bacteroides goldsteinii and Bacteroides merdae as Parabacteroides distasonis gen. nov., comb. nov., Parabacteroides goldsteinii comb. nov. and Parabacteroides merdae comb. nov. Int J Syst Evol Microbiol. 2006;56:1599–1605.
Pfalzer AC, Nesbeth PD, Parnell LD, et al. Diet- and genetically-induced obesity differentially affect the fecal microbiome and metabolome in Apc1638N mice. PLoS ONE. 2015;10:e0135758.
Kverka M, Zakostelska Z, Klimesova K, et al. Oral administration of Parabacteroides distasonis antigens attenuates experimental murine colitis through modulation of immunity and microbiota composition. Clin Exp Immunol. 2011;163:250–259.
Murphy EF, Cotter PD, Healy S, et al. Composition and energy harvesting capacity of the gut microbiota: relationship to diet, obesity and time in mouse models. Gut. 2010;59:1635–1642.
Segura-López FK, Güitrón-Cantú A, Torres J. Association between Helicobacter spp. infections and hepatobiliary malignancies: a review. World J Gastroenterol. 2015;21:1414–1423.
Goldstein EJ, Citron DM, Peraino VA, et al. Desulfovibrio desulfuricans bacteremia and review of human Desulfovibrio infections. J Clin Microbiol. 2003;41:2752–2754.
Imajo K, Fujita K, Yoneda M, et al. Hyperresponsivity to low-dose endotoxin during progression to nonalcoholic steatohepatitis is regulated by leptin-mediated signaling. Cell Metab. 2012;16:44–54.
D’Argenio V, Casaburi G, Precone V, Salvatore F. Comparative metagenomic analysis of human gut microbiome composition using two different bioinformatic pipelines. BioMed Res Int. 2014;2014:1–10.
Rolla S, Alchera E, Imarisio C, et al. The balance between IL-17 and IL-22 produced by liver-infiltrating T-helper cells critically controls NASH development in mice. Clin Sci (Lond). 2016;130:193–203.
Takahashi N, Vanlaere I, de Rycke R, et al. IL-17 produced by Paneth cells drives TNF-induced shock. J Exp Med. 2008;205:1755–1761.
Park SW, Kim M, Brown KM, et al. Paneth cell-derived interleukin-17A causes multiorgan dysfunction after hepatic ischemia and reperfusion injury. Hepatology. 2011;53:1662–1675.
Jin W, Dong C. IL-17 cytokines in immunity and inflammation. Emerg Microbes Infect. 2013;2:e60.
Miura K, Yang L, van Rooijen N, et al. Hepatic recruitment of macrophages promotes nonalcoholic steatohepatitis through CCR2. Am J Physiol Gastrointest Liver Physiol. 2012;302:G1310–G1321.
Gomes AC, Bueno AA, de Souza RG, et al. Gut microbiota, probiotics and diabetes. Nutr J. 2014;13:60.
Haileselassie Y, Johansson MA, Zimmer CL, et al. Lactobacilli regulate Staphylococcus aureus 161:2-induced pro-inflammatory T-cell responses in vitro. PLoS ONE. 2013;8:e77893.
Geem D, Medina-Contreras O, McBride M, et al. Specific microbiota-induced intestinal Th17 differentiation requires MHC class II but not GALT and mesenteric lymph nodes. J Immunol. 2014;193:431–438.
Harley IT, Stankiewicz TE, Giles DA, et al. IL-17 signaling accelerates the progression of nonalcoholic fatty liver disease in mice. Hepatology. 2014;59:1830–1839.
Acknowledgments
We thank Daichi Nakagawa for excellent technical assistance and the Biotechnology Center, Faculty of Bioresource Sciences, Akita Prefectural University, for assistance with the GS Junior instrument for pyrosequencing analyses.
Funding
This study was supported by a Grant-in-Aid for Scientific Research from the Japan Society for the Promotion of Science (K.M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors have no conflicts to disclose.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplemental Figure 1
Composition of gut microbiota examined by the GS Junior system. The relative abundance of gut microbiota is shown for HF, CDAA, and MCD diets, compared with the NC group. First, the percentage of the gut microbiota was calculated in the total count reads (NC: 10,829, HF: 13,374, CDAA: 26,009, MCD: 22,601 reads). Red indicates increased abundance, while green indicates decreased abundance, compared with the NC group. The composition of the gut microbiota is shown at phylum, order, genus, and species level. (A) Composition of gut microbiota in phylum Firmicutes. “C” indicates Clostridium. (B) Composition of gut microbiota in phylum Bacteroidetes. “B” and “P” indicate Bacteroides and Parabacteroides, respectively. (C) Composition of gut microbiota in phylum Proteobacteria. “D” and “H” indicate Desulfovibrio and Helicobacter, respectively. (TIFF 2927 kb)
Rights and permissions
About this article
Cite this article
Ishioka, M., Miura, K., Minami, S. et al. Altered Gut Microbiota Composition and Immune Response in Experimental Steatohepatitis Mouse Models. Dig Dis Sci 62, 396–406 (2017). https://doi.org/10.1007/s10620-016-4393-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-016-4393-x