Abstract
Background and Aims
Multiple clinical trials have demonstrated the efficacy and safety of tenofovir disoproxil fumarate (TDF) in chronic hepatitis B (CHB). However, long-term efficacy and safety data for TDF in real-life clinical practice are limited.
Methods
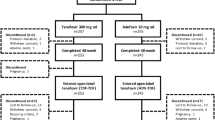
Prospective German field practice study in CHB-mono-infected patients. Patients were TDF-naïve but could have been treated previously with other HBV antivirals.
Results
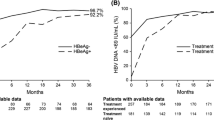
Efficacy analysis included 400 patients; 301 (75 %) completed 36 months of TDF treatment. Both treatment-naïve and treatment-experienced patients showed a rapid decline in HBV DNA within 3 months of TDF initiation. After 36 months, HBV DNA < 69 IU/mL was achieved by 91 % of treatment-naïve patients (90 and 92 % in hepatitis B “e” antigen [HBeAg]-positive and [HBeAg]-negative, respectively) and 96 % of treatment-experienced patients (93 and 97 %, respectively). Three patients experienced virologic breakthrough, all with reported non-compliance. Overall, 5.7 % HBeAg-positive and 2.2 % HBeAg-negative patients lost hepatitis B surface antigen. Safety data were consistent with the known TDF safety profile; the most commonly reported adverse events possibly related to TDF were fatigue (2.0 %) and headache (2.0 %). Few patients (1.3 %) experienced renal-related adverse reactions. Creatinine clearance remained relatively stable over time; patients responded favorably where TDF was dose adjusted per label for decreased creatinine clearance.
Conclusions
TDF showed a favorable tolerability profile and induced rapid and sustained suppression of HBV DNA in patients with CHB treated for up to 3 years in routine clinical practice, irrespective of treatment history. Efficacy and safety in this heterogeneous patient population were consistent with data from clinical trials.




Similar content being viewed by others
References
Marcellin P, Heathcote EJ, Buti M, et al. Tenofovir disoproxil fumarate versus adefovir dipivoxil for chronic hepatitis B. N Engl J Med. 2008;359:2442–2455.
Marcellin P, Gane E, Buti M, et al. Regression of cirrhosis during treatment with tenofovir disoproxil fumarate for chronic hepatitis B: a 5-year open-label follow-up study. Lancet. 2013;381:468–475.
Kitrinos KM, Corsa A, Liu Y, et al. No detectable resistance to tenofovir disoproxil fumarate after 6 years of therapy in patients with chronic hepatitis B. Hepatology. 2014;59:434–442.
Fung S, Kwan P, Fabri M, et al. Randomized comparison of tenofovir disoproxil fumarate vs emtricitabine and tenofovir disoproxil fumarate in patients with lamivudine-resistant chronic hepatitis B. Gastroenterology. 2014;146:980–988.
Berg T, Marcellin P, Zoulim F, et al. Tenofovir is effective alone or with emtricitabine in adefovir-treated patients with chronic-hepatitis B virus infection. Gastroenterology. 2010;139:1207–1217.
Patterson SJ, George J, Strasser SI, et al. Tenofovir disoproxil fumarate rescue therapy following failure of both lamivudine and adefovir dipivoxil in chronic hepatitis B. Gut. 2011;60:247–254.
Pan CQ, Hu KQ, Yu AS, et al. Response to tenofovir monotherapy in chronic hepatitis B patients with prior suboptimal response to entecavir. J Viral Hepatitis. 2012;19:213–219.
Liaw YF, Sheen IS, Lee CM, et al. Tenofovir disoproxil fumarate (TDF), emtricitabine/TDF, and entecavir in patients with decompensated chronic hepatitis B liver disease. Hepatology. 2011;53:62–72.
Lampertico P, Soffredini R, Yurdaydin C, et al. Four years of TDF for NUC naïve field practice European patients suppress HBV replication in most patients with a favorable renal safety profile but do not prevent HCC in patients with or without cirrhosis. Presented at AASLD, Washington DC, USA, November 1–5, 2013; poster 933.
van Bömmel F, deMan RA, Wedemeyer H, et al. Long-term efficacy of tenofovir monotherapy for hepatitis B virus-monoinfected patients after failure of nucleoside/nucleotide analogues. Hepatology. 2010;51:73–80.
van Bömmel F, de Man RA, Rutter R, et al. A European multicenter analysis of long term tenofovir (TDF) monotherapy for chronic hepatitis B in real life setting: efficacy, safety and HCC incidence. Presented at AASLD, Washington DC, USA, November 1–5, 2013; poster 941.
Levrero M, Cimino L, Lampertico P, et al. OptiB—A multicenter prospective open label study on Tenofovir (TDF) for chronic hepatitis B patients with suboptimal response to adefovir (ADV) or ADV/LAM treatment. Presented at EASL, Berlin, Germany, March 30–April 3, 2011; poster 732.
Lim L, Patterson S, George J, et al. Tenofovir rescue therapy achieves long-term suppression of HBV replication in patients with multi-drug resistant HBV: 5 year follow-up of the TDF109 cohort. Presented at AASLD, Boston MA, USA, November 9–13, 2012; poster 361.
Peterson J, Ratziu V, Buti M, et al. Entecavir plus tenofovir combination as rescue therapy in pre-treated chronic hepatitis B patients: an international multicenter cohort study. J Hepatol. 2012;56:520–526.
Pol S, Lampertico P. First-line treatment of chronic hepatitis B with entecavir or tenofovir in “real-life” settings: from clinical trials to clinical practice. J Viral Hepatitis. 2012;19:377–386.
Cornberg M, Protzer U, Petersen J, et al. Aktualisierung der s3-leitlinie zur prophylaxe, diagnostik und therapie der hepatitis-b-virusinfektion. Z Gastroenterol. 2011;49:871–930.
EASL. EASL clinical practice guidelines: management of chronic hepatitis B virus infection. J Hepatol. 2012;57:167–185.
Iloeje UH, Yang HI, Su J, et al. Predicting cirrhosis risk based on the level of circulating hepatitis B viral load. Gastroenterology. 2006;130:678–686.
Zoutendik R, Rejinders GP, Zoulim F, et al. Virological response to entecavir is associated with a better clinical outcome in chronic hepatitis B patients with cirrhosis. Gut. 2013;62:760–765.
Yuan HJ, Yuen MF, Ka-Ho WD, Sablon E, Lai CL. The relationship between HBV–DNA levels and cirrhosis-related complications in Chinese with chronic hepatitis B. J Viral Hepatol.. 2005;12:373–379.
Chen CJ, Iloeje UH, Yang HI. Serum hepatitis B virus DNA as a predictor of the development of cirrhosis and hepatocellular carcinoma. Curr Hepatol Rep. 2007;6:9–16.
Heathcote EJ, Marcellin P, Buti M, et al. Three year efficacy and safety of tenofovir disoproxil fumarate treatment for chronic hepatitis B. Gastroenterology. 2011;140:132–143.
Lock E. Sensitive and early markers of renal injury: where are we and what is the way forward? Toxicol Sci. 2010;116:1–4.
Tsai PJ, Chang A, Yamada S, Tsai N, Bartholomew ML. Use of tenofovir disoproxil fumarate in highly viremic, hepatitis B mono-infected pregnant women. Dig Dis Sci. 2014;59:2797–2803.
Greenup AJ, Tan PK, Nguyen V, et al. Efficacy and safety of tenofovir disoproxil fumarate in pregnancy to prevent perinatal transmission of hepatitis B virus. J Hepatol. 2014;61:502–507.
Funding
This study was supported by Gilead Sciences GmbH.
Author contributions
All authors were involved in data acquisition and analysis and interpretation of the data, contributed to drafting the manuscript with regards to important intellectual content and approved the manuscript prior to submission. Medical writing assistance was provided by Carol Lovegrove and Liesje Quine, Elements Communications Ltd, Westerham, UK and Jane Anderson, Chapel Hill, North Carolina, USA and funded by Gilead Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
JP is an investigator and speaker/consultant for Bristol-Myers Squibb, Novartis, Roche, AbbVie and a speaker/consultant for Abbott, Boehringer, Gilead, and GlaxoSmithKline. CE is a speaker/consultant for Bristol-Myers Squibb, Gilead, Janssen, Merck Sharp & Dohme and Roche. SM is a speaker/consultant for Gilead, Bristol-Myers Squibb, Roche and Merck Sharp & Dohme. DH is on the Speakers Bureau for Gilead, Janssen, Merck Sharp & Dohme, Roche, and has served on Advisory Boards for Gilead, Janssen and Roche. KB is a speaker/consultant for Bristol-Myers-Squibb, Gilead, Roche, Merck Sharp & Dohme, Novartis, Janssen-Cilag, and Gilead. SR and TW are employees of Gilead. JS, HH, CJ, and CT, have no conflicts of interest to declare.
Research involving human participants and/or animals
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Petersen, J., Heyne, R., Mauss, S. et al. Effectiveness and Safety of Tenofovir Disoproxil Fumarate in Chronic Hepatitis B: A 3-Year Prospective Field Practice Study in Germany. Dig Dis Sci 61, 3061–3071 (2016). https://doi.org/10.1007/s10620-015-3960-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-015-3960-x