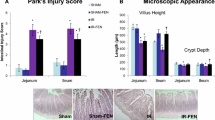
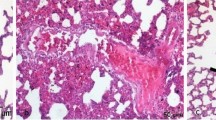
Abstract
Background
Xanthine oxidase has been implicated in the pathogenesis of a wide spectrum of diseases, and is thought to be the most important source of oxygen-free radicals and cell damage during re-oxygenation of hypoxic tissues.
Aims
The present study was undertaken to demonstrate whether febuxostat is superior to allopurinol in prevention of the local and remote harmful effects of small intestinal ischemia/reperfusion injury in rats.
Methods
Intestinal ischemia was induced by superior mesenteric artery ligation. The rats were assigned to five groups: the sham control; the intestinal ischemia/reperfusion; the allopurinol; and the febuxostat 5 and 10 mg/kg pretreated ischemia/reperfusion groups. Treatment was administered from 7 days before ischemia induction. After the reperfusion, the serum and tissues were obtained for biochemical, pharmacological, and histological studies.
Results
Intestinal reperfusion led to an elevation in the serum levels of alanine aminotransferase, aspartate aminotransferase, tumor necrosis factor-α, malondialdehyde, and xanthine oxidase as well as intestinal myeloperoxidase, malonadialdehyde, and xanthine oxidase/xanthine dehydrogenase activity. Furthermore, the ischemia/reperfusion induced a reduction in the contractile responsiveness to acetylcholine. These changes were significantly regulated by the pretreatment with febuxostat compared to allopurinol. The degree of pathological impairment in the intestinal mucosa, liver, and lung tissues were lighter in the pretreated groups.
Conclusions
Febuxostat may offer advantages over allopurinol in lessening local intestinal injury as well as remote hepatic and lung injuries induced by small intestinal ischemia/reperfusion.










Similar content being viewed by others
References
Tendler DA. Acute intestinal ischemia and infarction. Semin Gastrointest Dis. 2003;14:66–76.
Guneli E, Cavdar Z, Islekel H, et al. Erythropoietin protects the intestine against ischemia/reperfusion injury in rats. Mol Med. 2007;13:509–517.
Ismail HM, Wenxuan Y, Marc CW, et al. Ischemia-reperfusion injury of the intestine and protective strategies against injury. Dig Dis Sci. 2004;49:1359–1377.
Ceppa EP, Fuh KC, Bulkley GB. Mesenteric hemodynamic response to circulatory shock. Curr Opin Crit Care. 2003;9:127–132.
Ballabeni V, Barocelli E, Bertoni S, et al. Alterations of intestinal motor responsiveness in a model of mild mesenteric ischemia/reperfusion in rats. Life Sci. 2002;71:2025–2035.
Aldemir M, Gürel A, Büyükbayram H, et al. The effects of glucose-insulin-potassium solution and BN 52021 in intestinal ischaemia-reperfusion injury. Vasc Endovascular Surg. 2003;37:345–351.
Harrison R. Structure and function of xanthine oxidoreductase: where are we now? Free Radic Biol Med. 2002;33:774–797.
Julee PS, Deepa RP, Dhrubo JS. Febuxostat: the new generation novel xanthine oxidase Inhibitors. Int Pharm Sci. 2011;1:107–115. http://www.ipharmsciencia.com./Jan-March%202011.html.
Strazzullo P, Puig JG. Uric acid and oxidative stress: relative impact on cardiovascular risk. Nutr Metab Cardiovasc Dis. 2007;17:409–414.
Bruce SP. Febuxostat: a selective xanthine oxidase inhibitor for the treatment of hyperuricemia and gout. Ann Pharmacother. 2006;40:2187–2194.
Stojadinovic A, Smallridge R, Nath J, et al. Anti-inflammatory effects of U74389F in a rat model of intestinal ischemia/reperfusion injury. Crit Care Med. 1999;27:764–770.
Kulah B, Besler HT, Akdag M, et al. The effects of verapamil vs. allopurinol on intestinal ischemia/reperfusion injury in rats. “An experimental study”. Hepatogastroenterology. 2004;51:401–407.
Terzi C, Kuzu MA, Aşlar AK, et al. Prevention of deleterious effects of reperfusion injury using one-week high-dose allopurinol. Dig Dis Sci. 2001;46:430–437.
Margaritis EV, Yanni AE, Agrogiannis G, et al. Effects of oral administration of (l)-arginine, (l)-NAME and allopurinol on intestinal ischemia/reperfusion injury in rats. Life Sci. 2011;6:1070–1076.
Sánchez-Lozada LG, Tapia E, Bautista-García P, et al. Effects of febuxostat on metabolic and renal alterations in rats with fructose-induced metabolic syndrome. Am J Physiol Renal Physiol. 2008;294:710–718. http://ajprenal.physiology.org/content/294/4/F710.full.
Danielle GS, Vanessa P, Adriana CS, et al. Role of PAF receptors during intestinal ischemia and reperfusion injury. A comparative study between PAF receptor-deficient mice and PAF receptor antagonist treatment. Br J Pharmacol. 2003;139:733–740.
Wasowicz W, Neve J, Peretz A. Optimized steps in fluorometric determination of thiobarbituric acid-reactive substances in serum: importance of extraction pH and influence of sample preservation and storage. Clin Chem. 1993;39:2522–2526.
Beckman JS, Parks DA, Pearson JD, et al. A sensitive fluorometric assay for measuring xanthine dehydrogenase and oxidase in tissues. Free Radic Biol Med. 1989;6:607–615.
Hillegass LM, Griswold DE, Brickson B, et al. Assessment of myeloperoxidase activity in whole rat kidney. J Pharmacol Methods. 1990;24:285–295.
Lowry OH, Rosebrough NJ, Farr AL, et al. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951;193:265–275.
Draper HH, Hadley M. Malondialdehyde determination as index of lipid peroxidation. Methods Enzymol. 1990;186:421–431.
Chiu CJ, McArdle AH, Brown R, Brown R, Scott HJ, Gurd FN. Intestinal mucosal lesion in low-flow states. I. A morphological, hemodynamic, and metabolic reappraisal. Arch Surg. 1970;101:478–483.
Omar R, Nomikos I, Piccorelli G, Savino J, Agarwal N. Prevention of postischaemic lipid peroxidation and liver cell injury by iron chelation. Gut. 1989;30:510–514.
Murakami K, Bjertnaes LJ, Schmalstieg FC, et al. A novel animal model of sepsis after acute lung injury in sheep. Crit Care Med. 2002;30:2083–2090.
Köksoy C, Kuzu MA, Ergŭn H, et al. Intestinal ischemia–reperfusion impairs vasomotor functions of pulmonary vascular bed. Ann Surg. 2000;231:105–111.
Collard CD, Gelman S. Pathophysiology, clinical manifestations, and prevention of ischemia-reperfusion injury. Anesthesiology. 2001;94:1133–1138.
Kaplan N, Yagmurdur H, Kilinc K, et al. The protective effects of intravenous anesthetics and verapamil in gut ischemia/reperfusion-induced liver injury. Anesth Analg. 2007;105:1371–1378.
Hammerman C, Goldschmidt D, Caplan MS, et al. Amelioration of ischemia-reperfusion injury in rat intestine by pentoxifylline-mediated inhibition of xanthine oxidase. J Pediatr Gastroenterol Nutr. 1999;29:69–74.
Cetinkaya A, Bulbuloglu E, Kurutas EB, et al. N-acetylcysteine ameliorates methotrexate-induced oxidative liver damage in rats. Med Sci Monit. 2006;12:274–278. http://www.ncbi.nlm.nih.gov/pubmed/16865059.
Granger DN, Stokes KY, Shigematsu T, et al. Splanchnic ischaemia-reperfusion injury: mechanistic insights provided by mutant mice. Acta Physiol Scand. 2001;173:83–91.
Lee J, Koo N, Min DB. Reactive oxygen species, aging, and antioxidative nutraceuticals. Compr Rev Food Sci Food Saf. 2004;3:21–33.
Okamoto K, Takeshi N. Crystal structures of mammalian xanthine oxidoreductase bound with various inhibitors: allopurinol, febuxostat, and FYX-051. J Nippon Med Sch. 2008;75:2–3. http://www.biomedexperts.com/Abstract.bme/18360072/Crystal_structures_of_mammalian_xanthine_oxidoreductase_bound_with_various_inhibitors_allopurinol_febuxostat_and_FYX.
Anjana P, Mridul C, Himanshu P, et al. Febuxostat—a new treatment for hyperuricaemia in gout—a review article. Natl J Physiol Pharm Pharmacol. 2012;2:23–28. http://njppp.com/.
Takano Y, Hase-Aoki K, Horiuchi H, et al. Selectivity of febuxostat, a novel non-purine inhibitor of xanthine oxidase/xanthine dehydrogenase. Life Sci. 2005;76:1835–1847.
Ahmadinejad M, Rex M, Sutton RH, et al. The effects of allopurinol on the ultrastructure of ischaemic and reperfused large intestine of sheep. Aust Vet J. 1996;74:135–139.
Devecí E. Role of allopurinol, verapamil, dexamethasone and trifluoperazine as prophylactic agents in intestinal ischemia-reperfusion. Anal Quant Cytol Histol. 2008;30:99–104.
Stephanie P, Alexander HT, Roberto A, et al. Allopurinol and xanthine oxidase inhibition in liver ischemia reperfusion. J Hepatobiliary Pancreat Sci. 2011;18:137–146.
Guan W, Osanai T, Kamada T, et al. Effect of allopurinol pretreatment on free radical generation after primary coronary angioplasty for acute myocardial infarction. J Cardiovasc Pharmacol. 2003;41:699–705.
Rawan T, James EG. Potential future neuroprotective therapies for neurodegenerative disorders and stroke. Clin Geriatr Med. 2010;26:125–147.
Liu PG, He SQ, Zhang YH, et al. Protective effects of apocynin and allopurinol on ischemia/reperfusion-induced liver injury in mice. World J Gastroenterol. 2008;14:2832–2837.
Taha MO, Simoẽs MJ, Noguerol EC, et al. Effects of allopurinol on ischemia and reperfusion in rabbit livers. Transpl Proc. 2009;41:820–823.
Hopson SB, Lust RM, Sun YS, et al. Allopurinol improves myocardial reperfusion injury in a xanthine oxidasefree model. J Natl Med Assoc. 1995;87:480–484.
Riaz AA, Wan MX, Schafer T, et al. Allopurinol and superoxide dismutase protect against leucocyte endothelium interactions in a novel model of colonic ischemia-reperfusion. Br J Surg. 2002;89:1572–1580.
Vaughan WG, Horton JW, Walker PB. Allopurinol prevents intestinal permeability changes after ischemia-reperfusion injury. J Pediatr Surg. 1992;27:968–972.
Riaz AA, Schramm R, Sato T, et al. Oxygen radical-dependent expression of CXC chemokines regulates ischemia/reperfusion-induced leukocyte adhesion in the mouse colon. Free Radic Biol Med.. 2003;35:782–789.
Hakguder G, Akgur FM, Ates O, et al. Short-term intestinal ischemia-reperfusion alters intestinal motility that can be preserved by xanthine oxidase inhibition. Dig Dis Sci. 2002;47:1279–1283.
Malkiel S, Har-el R, Schwalb H, et al. Interaction between allopurinol and copper: possible role in myocardial protection. Free Radic Res Commun. 1993;18:7–15.
Kinugasa Y, Ogino K, Furuse Y, et al. Allopurinol improves cardiac dysfunction after ischemia-reperfusion via reduction of oxidative stress in isolated perfused rat hearts. Circ J. 2003;67:781–787.
Nishizawa J, Nakai A, Matsuda K, et al. Reactive oxygen species play an important role in the activation of heat shock factor 1 in ischemic-reperfused heart. Circulation. 1999;99:934–941.
Perez NG, Gao WD, Marban E. Novel myofilament Ca + 2-sensitizing property of xanthine oxidase inhibitors. Circ Res. 1998;83:423–430.
Yang CS, Tsai PJ, Chou ST, et al. The roles of reactive oxygen species and endogenous opioid peptides in ischemia-induced arrhythmia of isolated rat hearts. Free Radic Biol Med.. 1995;18:593–598.
Bielefeldt K, Conklin JL. Intestinal motility during hypoxia and reoxygenation in vitro. Dig Dis Sci. 1997;42:878–884.
Udassin R, Eimerl D, Schiffman J, et al. Postischemic intestinal motility in rat is inversely correlated to length of ischemia. An in vivo animal model. Dig Dis Sci. 1995;40:1035–1038.
Menger MD, Rücker M, Vollmar B. Capillary dysfunction in striated muscle ischemia/reperfusion: on the mechanisms of capillary “no-reflow”. Shock. 1997;8:2–7.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shafik, A.N. Febuxostat Improves the Local and Remote Organ Changes Induced by Intestinal Ischemia/Reperfusion in Rats. Dig Dis Sci 58, 650–659 (2013). https://doi.org/10.1007/s10620-012-2391-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2391-1