Abstract
Background
Toll-like receptors (TLRs) have achieved an extraordinary amount of interest in inflammatory diseases due to their role in the inflammatory activation. By activating the production of several biological factors, TLRs induce type I interferons and other cytokines, which drive the inflammatory response and activate the adaptive immune system.
Aims
The aim of this study was to investigate and compare the expression and clinical relevance of TLRs and interleukins in pediatric and adult celiac disease (CD), defined as intolerance to dietary proteins found in wheat, barley, and rye.
Methods
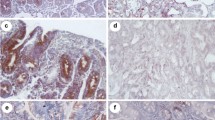
The expression levels of TLR3, TLR4, and TLR7, interleukins, and different transcription factors were analyzed on duodenal biopsies from ten children and 31 adults with CD, and 21 duodenal controls biopsies without CD (ten children and 11 adults). The analyses were performed by immunohistochemistry and real-time PCR.
Results
There were no significant differences in the studied parameters between adults and children. TLR4 expression level was increased twofold in CD specimens compared to controls. CD patients with high levels of TLR4 also showed high levels of interleukins (IL1, IL6, IL8, and IL17) as well as transcription factors (IRAK4, MyD88, and NF-κB).
Conclusions
TLR4 expression is associated with CD independently of age at diagnosis. Pediatric patients and adult patients have a similar inflammatory profile, making it possible to treat both with the same immunological therapy in the future.




Similar content being viewed by others
References
Monteleone I, Sarra M, Del Vecchio Blanco G, et al. Characterization of IL-17A-producing cells in celiac disease mucosa. J Immunol. 2010;184:2211–2218.
Sollid LM, Thorsby E. HLA susceptibility genes in celiac disease: genetic mapping and role in pathogenesis. Gastroenterology. 1993;105:910–922.
Setty M, Hormaza L, Guandalini S. Celiac disease: risk assessment, diagnosis, and monitoring. Mol Diagn Ther. 2008;12:289–298.
Sollid LM. Molecular basis of celiac disease. Annu Rev Immunol. 2000;18:53–81.
Kilmartin C, Lynch S, Abuzakouk M, et al. Avenin fails to induce a Th1 response in coeliac tissue following in vitro culture. Gut. 2003;52:47–52.
Forsberg G, Hernell O, Melgar S, et al. Paradoxical coexpression of proinflammatory and down-regulatory cytokines in intestinal T cells in childhood celiac disease. Gastroenterology. 2002;123:667–678.
Szebeni B, Veres G, Dezsofi A, et al. Increased mucosal expression of Toll-like receptor (TLR)2 and TLR4 in coeliac disease. J Pediatr Gastroenterol Nutr. 2007;45:187–193.
Szebeni B, Veres G, Dezsofi A, et al. Increased expression of Toll-like receptor (TLR) 2 and TLR4 in the colonic mucosa of children with inflammatory bowel disease. Clin Exp Immunol. 2008;151:34–41.
Akira S. Toll-like receptors and innate immunity. Adv Immunol. 2001;78:1–56.
Marshak-Rothstein A. Toll-like receptors in systemic autoimmune disease. Nat Rev Immunol. 2006;6:823–835.
Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4:499–511.
Shang L, Fukata M, Thirunarayanan N, et al. Toll-like receptor signaling in small intestinal epithelium promotes B-cell recruitment and IgA production in lamina propria. Gastroenterology. 2008;135:529–538.
Pasare C, Medzhitov R. Toll-dependent control mechanisms of CD4 T cell activation. Immunity. 2004;21:733–741.
Takeuchi O, Hoshino K, Kawai T, et al. Differential roles of TLR2 and TLR4 in recognition of Gram-negative and Gram-positive bacterial cell wall components. Immunity. 1999;11:443–451.
Kurt-Jones EA, Popova L, Kwinn L, et al. Pattern recognition receptors TLR4 and CD14 mediate response to respiratory syncytial virus. Nat Immunol. 2000;1:398–401.
Gonzalez-Reyes S, Marin L, Gonzalez L, et al. Study of TLR3, TLR4 and TLR9 in breast carcinomas and their association with metastasis. BMC Cancer. 2010;10:665.
Maiuri L, Ciacci C, Ricciardelli I, et al. Association between innate response to gliadin and activation of pathogenic T cells in coeliac disease. Lancet. 2003;362:30–37.
Schuppan D, Esslinger B, Dieterich W. Innate immunity and coeliac disease. Lancet. 2003;362:3–4.
Santin I, Castellanos-Rubio A, Hualde I, et al. Toll-like receptor 4 (TLR4) gene polymorphisms in celiac disease. Tissue Antigens. 2007;70:495–498.
Fernandez-Jimenez N, Castellanos-Rubio A, Plaza-Izurieta L, et al. Analysis of beta-defensin and Toll-like receptor gene copy number variation in celiac disease. Hum Immunol. 2010;71:833–836.
Medzhitov R, Janeway CA Jr. Innate immunity: the virtues of a nonclonal system of recognition. Cell. 1997;91:295–298.
Maiuri L, Picarelli A, Boirivant M, et al. Definition of the initial immunologic modifications upon in vitro gliadin challenge in the small intestine of celiac patients. Gastroenterology. 1996;110:1368–1378.
Sartor RB. Cytokines in intestinal inflammation: pathophysiological and clinical considerations. Gastroenterology. 1994;106:533–539.
Bettelli E, Oukka M, Kuchroo VK. T(H)-17 cells in the circle of immunity and autoimmunity. Nat Immunol. 2007;8:345–350.
Harris KM, Fasano A, Mann DL. Cutting edge: IL-1 controls the IL-23 response induced by gliadin, the etiologic agent in celiac disease. J Immunol. 2008;181:4457–4460.
Cinova J, Palova-Jelinkova L, Smythies LE, et al. Gliadin peptides activate blood monocytes from patients with celiac disease. J Clin Immunol. 2007;27:201–209.
Jelinkova L, Tuckova L, Cinova J, et al. Gliadin stimulates human monocytes to production of IL-8 and TNF-alpha through a mechanism involving NF-kappaB. FEBS Lett. 2004;571:81–85.
Thomas KE, Sapone A, Fasano A, et al. Gliadin stimulation of murine macrophage inflammatory gene expression and intestinal permeability are MyD88-dependent: role of the innate immune response in Celiac disease. J Immunol. 2006;176:2512–2521.
Sacre SM, Andreakos E, Kiriakidis S, et al. The Toll-like receptor adaptor proteins MyD88 and Mal/TIRAP contribute to the inflammatory and destructive processes in a human model of rheumatoid arthritis. Am J Pathol. 2007;170:518–525.
Ciccocioppo R, Di Sabatino A, Bauer M, et al. Matrix metalloproteinase pattern in celiac duodenal mucosa. Lab Invest. 2005;85:397–407.
Escaff S, Fernandez JM, Gonzalez LO, et al. Study of matrix metalloproteinases and their inhibitors in prostate cancer. Br J Cancer. 2010;102:922–929.
Del Casar JM, Gonzalez-Reyes S, Gonzalez LO, et al. Expression of metalloproteases and their inhibitors in different histological types of breast cancer. J Cancer Res Clin Oncol. 2010;136:811–819.
Altadill A, Rodriguez M, Gonzalez LO, et al. Liver expression of matrix metalloproteases and their inhibitors in hepatocellular carcinoma. Dig Liver Dis. 2009;41:740–748.
Gonzalez LO, Corte MD, Vazquez J, et al. Study of matrix metalloproteinases and their tissue inhibitors in ductal in situ carcinomas of the breast. Histopathology. 2008;53:403–415.
Freeman HJ. Adult celiac disease and its malignant complications. Gut Liver. 2009;3:237–246.
Acknowledgments
This work was supported by grants from FICEMU.
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
The authors Noemí Eiró and Salomé González-Reyes contributed equally to this work.
Rights and permissions
About this article
Cite this article
Eiró, N., González-Reyes, S., González, L. et al. Duodenal Expression of Toll-Like Receptors and Interleukins Are Increased in Both Children and Adult Celiac Patients. Dig Dis Sci 57, 2278–2285 (2012). https://doi.org/10.1007/s10620-012-2184-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-012-2184-6