Abstract
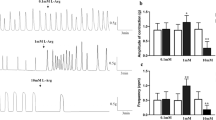
The effect of hyperhomocysteinemia induced by pretreatment with methionine 12 weeks prior to the study on the responses induced by γ-aminobutyric acid (GABA), electrical field stimulation (EFS), and ATP have been evaluated in isolated rat duodenum. In the presence of adrenergic and cholinergic blockade, EFS (60 V, 1 ms, 1–3 Hz) induced frequency-dependent relaxations of the preparation. GABA and ATP also caused submaximal relaxation of the rat duodenum. The relaxations induced by GABA, EFS, and ATP were not significantly changed in duodenal tissues from hyperhomocysteinemic rats compared with control rats. GABA- and EFS-induced relaxations were inhibited by N-nitro-l-arginine methyl ester (L-NAME; 3 × 10−4 M) in both hyperhomocysteinemic and control rats. On the other hand, L-NAME incubation did not affect ATP-induced relaxation. These results suggest that hyperhomocysteinemia does not cause an important impairment on non-adrenergic non-cholinergic innervation of the rat duodenum.




Similar content being viewed by others
References
de Bree A, Verschuren M, Kromhout D, Kluıjtmans AJ, Blom HJ (2002) Homocysteine determinants and the evidence to what extent homocysteine determines the risk of coronary heart disease. Pharmacol Rev 54:599–618
Kang SS, Wong PWK, Malinow MR (1992) Hyperhomocyst(e)inemia as a risk factor for occlusive vascular disease. Ann Rev Nutr 12:279–298
Perna AF, Castaldo P, Ingrosso D, De Santo NG (1999) Homocysteine, a new cardiovascular risk factor, is also a powerful uremic toxin. J Nephrol 12:230–240
Verklej-Hagoort A, Verlinde M, Ursem N (2006) Maternal hyperhomocysteine is a risk factor for congenital heart disease. BJOG 113:1412–1418
Parsons RB, Waring RH, Ramsden DB, Williams AC (1998) In vitro effect of the cysteine metabolites homocysteic acid, homocysteine and cysteic acid upon human neuronal cell lines. Neurotoxicology 19(4–5):599–603
Seshadri S, Beiser A, Selhub J, Jacques PF, Rosenberg IH, D’Agostıno RB, Wılson PW, Wolf PA (2002) Plasma homocysteine as a risk factor for dementia and Alzheimer’s disease. N Engl J Med 346:476–483
Lentz SR, Sobey CG, Piegors DJ, Bhopatkar MY, Faraci FM, Malinow MR (1996) Vascular dysfunction in monkeys with diet-induced hyperhomocyst(e)inemia. J Clin Invest 98:24–29
Ungvari Z, Pacher P, Rischak K, Szollar L, Koller A (1999) Dysfunction of nitric oxide mediation in isolated rat arterioles with methionine diet-induced hyperhomocysteinemia. Arterioscler Thromb Vasc Biol 19(8):1899–1904
Jones RWA, Jeremy JY, Koupparis A, Persad R, Shukla N (2005) Cavernosal dysfunction in a rabbit model of hyperhomocysteinemia. Br J Urol 95:125–130
Tasatargıl A, Sadan G, Golbası I, Karasu E, Turkay C (2004) Effects of short-term exposure to homocysteine on vascular responsiveness of human internal mammary artery. J Cardiovasc Pharmacol 43:692–697
Tasatargıl A, Sadan G, Karasu E (2007) Homocysteine-induced changes in vascular reactivity of guinea-pig pulmonary arteries: role of the oxidative stress and poly (ADP-ribose) polymerase activation. Pulm Pharmacol Ther 20(3):265–272
Tasatargıl A, Sadan G, Karasu E, Ozdem S (2006) Changes in atrium and thoracic aorta reactivity to adenosinergic and adrenergic agonists in experimental hyperhomocysteinemia. J Cardiovasc Pharmacol 47:673–679
Glaskow I, Mattar K, Krantis A (1998) Rat gastroduodenal motility in vivo: involvement of NO and ATP in spontaneous motor activity. Gastrointest Liver Physiol 38:G889–G896
Manzini S, Maggi CA, Meli A (1986) Pharmacological evidence that at least two different non-adrenergic non-cholinergic inhibitory systems are present in the rat small intestine. Eur J Pharmacol 123:229–236
Matharu MS, Hollingsworth M (1992) Purinoreceptors mediating relaxation and spasm in the rat gastric fundus. Br J Pharmacol 106:395–403
Matusak A, Bauer V (1986) Effect of desensitization induced by adenosine 5′-triphosphate, substance P, bradykinin, serotonin, GABA and endogenous noncholinergic–nonadrenergic transmitter in the guinea-pig ileum. Eur J Pharmacol 126:199–209
Mercier-Parot L, Tuchmann-Duplessis H (1973) Abortifactent and teratogenic effect of suramin, a trypanocide. C R Seances Soc Biol Ses Filiales 167:1518–1522
Windscheif U, Pfaff O, Ziganshin AU, Hoyle CHV, Baumert HG, Mutschler E, Burnstock G, Lambrecht G (1995) Inhibitory action of PPADS on relaxant responses to adenine nucleotides or electrical field stimulation in guinea-pig taenia coli and rat doudenum. Br J Pharmacol 115:1509–1517
Duarte IDG, Lorezetti BB, Ferreira SH (1990) Acetylcholine induces peripheral analgesia by the release of nitric oxide. In: Moncada S, Higgs EA (eds) The nitric oxide from l-arginine: a biorgulatory system. Elsevier, Amsterdam, p 165
Rand JM (1992) Nitric oxide as a mediator of non-adrenergic, non-cholinergic neuro-effector transmission. Clin Exp Pharmacol Physiol 19:147–169
Bredt DS, Hwang PM, Synder SH (1990) Localization of nitric oxide synthase indicating a neuronal role for nitric oxide. Nature 347:768–770
Belai A, Schmidt HHHW, Hoyle CHV (1992) Colocalization of nitric oxide synthase and NADPH-diaphorase in the myenteric plexus of the rat gut. Neurosci Lett 143:60–64
Martins SR, Bicudo R, Oliveira RB, Ballejo G (1993) Evidence for the partipicipation of the l-arginine–nitric oxide pathway in neurally induced relaxation of the isolated rat duodenum. Braz J Med Biol Res 26:1325–1335
Nichols K, Krantis A, Staines W (1992) Histochemical localization of nitric oxide-synthesizing neurons and vascular sides in the guinea-pig intestine. Neuroscience 51:791–799
Nichols K, Staines W, Krantis A (1993) Nitric oxide synthase distribution in rat intestine: a histochemical analysis. Gastroenterology 105:1651–1661
Nichols K, Staines W, Wu-Y JY, Krantis A (1995) Immonupositive GABAergic neural sides display nitric oxide synthase-releated NADPH diaphorase activitiy in the human colon. J Auton Nerv Syst 50:253–262
Jessen KR, Mirsky R, Densson ME, Burnstock G (1979) GABA may be a neurotransmitter in the vertebrate peripheral nervous system. Nature 281:71–74
Maggi CA, Manzini S, Meli A (1984) Evidence that GABAA receptors mediate relaxation of rat duodenum by activating intramural nonadrenergic–noncholinergic neurons. J Auton Pharmacol 4:77–85
Krantis A, Costa M, Furness JB, Orbach J (1980) γ-Aminobutyric acid stimulates intrinsic inhibitory and excitatory nerves in the guinea-pig intestine. Eur J Pharmacol 141:461–468
Tonini M, Crema A, Frigo GM, Rizzi CA, Manzo L, Candura SM, Onori L (1989) An in vitro study of the relation between GABA receptor function and propulsive motility in the distal colon of the rabbit. Br J Pharmacol 98:1109–1118
Boeckxstaens GE, Pelckmans PA, Rampart M, Ruytjens IF, Verbeuren TJ, Herman AG, Van Maercke YM (1990) GABAA receptor mediated stimulation of non-adrenergic non-cholinergic neurons in the dog ileocolonic junction. Br J Pharmacol 101:460–464
Krantis A, Mattar K, Glasgow I (1998) Rat gastroduodenal motility in vivo: interaction of GABA and VIP in control of spontaneouys relaxations. Am J Physiol Gastrointest Liver Physiol 275:897–903
Gustafsson B, Delbro D (1993) Tonic inhibition of small intestinal motility by nitric oxide. J Auton Nerv Syst 44:179–187
Kaputlu İ, Özdem S, Şadan G, Gökalp O (1999) Effects of diabetes on non-adrenergic, non-cholinergic relaxation induced by GABA and electrical stimulation in the rat isolated duodenum. Clin Exp Pharm Physiol 26:724–728
Stampfer MJ, Malinow MR (1995) Can lowering homocysteine levels reduce cardiovascular risk? N Engl J Med 332:328–329
Verhoef P, Kok FJ, Kruyssen DACM, Schouten EG, Witteman JCM, Grobbee DE, Ueland PM, Refsum H (1997) Plasma total homocysteine, B vitamins, of coronary atherosclerosis. Arterioscler Thromb Vasc Biol 17:989–995
De Bree A, Verschuren WM, Kromhout D, Kluijtmans LA, Blom HJ (2002) Homocysteine determinants and the evidence to what extent homocysteine determines the risk of coronary heart disease. Pharmacol Rev 54:599–618
Boysen G, Brander T, Christensen H, Gideon R, Truelsen T (2003) Homocysteine and risk of recurrent stroke. Stroke 34:1258–1261
Vollset SE, Refsum H, Tverdal A, Nygard O, Nordrehaug J, Tell GS, Ueland PM (2001) Plasma total homocysteine and cardiovascular and non-cardiovascular mortality: the Hordalans homocysteine study. Am J Clin Nutr 74:130–136
Malinow MR, Kang SS, Taylor LM, Wong PWK, Coull B, Inahara T, Mukerjee D, Sexton G, Upson B (1989) Prevalence of hyperhomocyteinemia in patients with peripheral arterial occlusive disease. Circulation 79:1180–1188
Clarke R, Daly L, Robinson K, Naughten E, Cahalane S, Fowler B, Graham I (1991) Hyperhomocysteinemia: an independent risk factor for vascular disease. N Engl J Med 313:709–715
Burnstock G, Campbell G, Rand MJ (1966) The inhibitory innervation of the taenia of the guinea-pig caecum. J Physiol Lond 182:504–526
Burnstock G, Campbell G, Bennett M, Holman ME (1963) Inhibition of the smooth muscle of the taenia coli. Nature 200:581–582
Sanders KM, Ward SM (1992) Nitric oxide as a mediator of nonadrenergic noncholinergic neurotransmission. Am J Physiol 262:G379–G392
Takahashı T (2003) Pathophysiological significance of neuronal nitric oxide synthase in the gastrointestinal tract. J Gastroenterol 38:421–430
Allescher HD, Lu S, Daniel EE, Classen M (1993) Nitric oxide as a putative nonadrenergic noncholinergic inhibitory neurotransmitter in the opossum sphincter of Oddi. Can J Physiol Pharmacol 71:525–530
Mourelle M, Guarner F, Moncada S, Malagelada JR (1993) The arginine/nitric oıxide pathway modulates sphincter of Oddi motor activity in guinea pigs and rabbits. Gastroenterology 105:1299–1305
Szilvassy Z, Nagy I, Szilvassy J, Jakab I, Csati S, Lonovics J (1996) Impaired nitrergic relaxation of the sphincter of Oddi of hyperlipidemic rabbits. Eur J Pharmacol 301:R17–R18
Cattaneo M, Vecchi M, Zighetti ML, Saibeni S, Martinelli I, Omodei P, Mannucci PM, de Francis R (1998) High prevalence of hyperhomocysteinemia in patients with inflammatory bowel disease: a pathogenic link thromboembolic complications? Thromb Haemost 80:542–545
Oldenburg B, Fijnheer R, van der Griend R, vanBerge-Henegouwen GP, Koningsberger JC (2000) Homocysteine in inflammatory bowel disease: a risk factor for thromboembolic complications? Am J Gastroenterol 95:2825–2830
Romagnoulo J, Fedorak RN, Dias VC, Bamforth F, Teltscher M (2001) Hyperhomocysteinemia and inflammatory bowel disease: prevalence and predictors in cross-sectional study. Am J Gastroenterol 96:2143–2149
Koutroubakis LE, Dilaveraki E, Vlachonikolis IG, Vardas E, Vrentzos G, Ganotakis E (2000) Hyperhomocystenemia in Greek patients with inflammatory bowel disease. Dig Dis Sci 45:2347–2351
Papa A, De-Stefano V, Danese S, Chiusolo P, Persichilli S, Casorelli I, Zappacosta B, Gıardina B, Gasbarrini A, Leone G, Gasbarrini G (2001) Hyperhomocysteinemia and prevalence of polymorphism of homocysteine metabolism-related enzymes in patients with inflammatory bowel disease. Am J Gastroenterol 96:2677–2682
Kaputlu I, Sadan G (1996) Evidence that nitric oxide mediates non-adrenergic non-cholinergic relaxation induced by GABA and electrical stimulation in the rat isolated duodenum. J Auton Pharmacol 16:177–182
Maggi CA, Manzini S, Mell A (1984) Evidence that GABAA receptors mediate relaxation of rat deoudenum by activating intramural nonadrenergic- noncholinergic neurons. J Auton Pharmacol 4:77–85
Manzini S, Maggi CA, Mell A (1985) Further evidence for involvement of adenosine-5′-triphosphate in non-adrenergic non-cholinergic relaxation of the isolated rat duodenum. Eur J Pharmacol 86:9–17
Ozdem SS, Sadan G (1999) Impairment of GABA-mediated contractions of rat isolated ileum by experimental diabetes. Pharmacology 59:165–170
Park KJ, Baker SA, Cho SY, Sanders KM, Koh DS (2005) Sulfur-containing amino acids block stretch-dependent K+ channels and nitrergic responses in the murine colon. Br J Pharmacol 144:1126–1137
Acknowledgment
The authors thank the support provided by the Akdeniz University Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Karasu, E., Şadan, G. & Tasatargil, A. Effects of Hyperhomocysteinemia on Non-Adrenergic Non-Cholinergic Relaxation in Isolated Rat Duodenum. Dig Dis Sci 53, 2106–2112 (2008). https://doi.org/10.1007/s10620-008-0318-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10620-008-0318-7