Abstract
The Cyto-3D-print is an adapter that adds cytospin capability to a standard centrifuge. Like standard cytospinning, Cyto-3D-print increases the surface attachment of mitotic cells while giving a higher degree of adaptability to other slide chambers than available commercial devices. The use of Cyto-3D-print is cost effective, safe, and applicable to many slide designs. It is durable enough for repeated use and made of biodegradable materials for environment-friendly disposal.
Similar content being viewed by others
Introduction
Cytospin is a cytology method that is specifically designed to concentrate and attach cells for eventual immunochemistry evaluation (Brown et al. 1996; Campaner et al. 2005; Choi and Anderson 1979; Martinazzi 1971; Shanholtzer et al. 1982; Watson 1966). Cytofunnel devices and specialized cytocentrifuges are sometimes used to concentrate cell suspension onto slide surfaces (Nassar et al. 2003; Piaton et al. 2004). In our studies on mitotic cells, we encountered difficulty in cell attachment even with adherent HeLa cells. During mitosis, cells gain a defined geometry and sufficient space for a mitotic spindle with proper orientation and correct chromosome segregation (Brown et al. 1996; Fischer-Friedrich et al. 2014). Adherent cells round up, forming spheres, this is a sign of healthy dividing cells, but the change in attached surface area leads to frequent loss of cells during experiments (Fischer-Friedrich et al. 2014). Cytospinning is employed and increases the number of mitotic cells that remain on the slide surface. Centrifuge adapters are available for standard six-well plates but not for microslide chambers (25 × 75 mm) or 35-mm slide plates. So we developed Cyto-3D-print inserts made of polylactic acid (PLA) plastic, that fit into existing centrifuge buckets for cytospinning these smaller slide chambers. Our aim in this study is to prove that Cyto-3D-print provides a rapid, safe and cost effective way to attach mitotic cells and suspension cell lines to slide surfaces.
Materials and methods
Mammalian cell culture
HeLa cells (ATCC, Manassas, VA, CCL-2) were grown in DMEM (Corning, Corning, NY, USA, 10-017-CV) with 10 % Fetal Bovine Serum (GE Healthcare Life Sciences, HyClone Laboratorie, Logan, Utah, USA, SH30071.03), 1 % Penicillin and Streptomycin (Life Technologies, Grand Island NY, USA, 15240-062). These monolayer cells were sub-cultured every 3–4 days.
Reagents
Thymidine and nocodazole were purchased from Millipore (Billerica, MA, USA; Calbiochem #6060 and #487928) For immunofluorescence preparation, we utilized poly-l-lysine (Sigma, St. Louis, MO, USA; P4832), 10 % formaldehyde (Polysciences, Warrington, PA, USA; 04018) and BSA (Sigma, A3059) in Lab-Tek microslide chambers (Thermo Scientific, USA, Nunc 155361).
Antibodies
MAb414 (Covance, Princeton, NJ, USA; MMS-120P) and anti-lamin B1 (Santa Cruz Biotechnology polyclonal gAb, Santa Cruz, CA, USA; 6216) antibodies were used for immunofluorescence with DAPI (Sigma, 471224) to visualize DNA.
Synchronizing HeLa cells
Cells were synchronized by double thymidine and nocodazole block (Glavy et al. 2007; Kaur et al. 2010). A 2 mM concentration of thymidine was added to the medium for 18 h in which HeLa cells were grown in culture. Introducing thymidine prohibits synthesis of DNA by a negative feedback mechanism. Cells in this population go through the cell cycle and will be halted at interphase. Cells were washed 3X with phosphate buffered saline pH 7.4 (PBS) and released into the medium without thymidine for the next 8 h. Cells were then treated again with 2 mM thymidine for 18 h and released into the medium with 0.02 μg/mL nocodazole for 12 h. Cells were collected by mitotic-shake off.
Cyto-3D-print
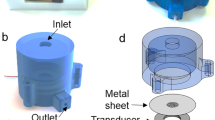
3D print material used was polylactic acid (PLA) 0.22 kg Small Spool 1.75 mm PLA Filament F with Replicator Mini 3D Printer (True Orange Item: IM1UX7688 (Staples, FL, USA, $18.00) Makerbot, Brooklyn, NY, USA; Model: MP05787). We designed a prototype file in Solidworks (Dassault Systèmes, Vélizy, France) with dimensions to fit into a centrifuge bucket from an Eppendorf 5810R Centrifuge (Fig. 1a). This prototype file can be modified to fit within any centrifuge bucket as well as adjusted to fit other slide chambers inside its holder chamber. The file was designed to fit a parafilm wrapped microslide chamber across and leave enough finger-friendly space for easy placement and removal as shown in Fig. 1. A MakerBot Replicator 2x printer was used to print a plastic 3D version of our file (estimated cost under $5 per unit). Files can be printed on any 3D printer or 3D print service at very low cost ($20 for two units).
Cyto-3D-print development. Panel A is the solidworks 3-D print prototype designed to hold a parafilm wrapped microslide chamber within its slide chamber holder and fit inside an Eppendorf 5810R centrifuge bucket. Panel B is a PLA Cyto-3D-print next to a wrapped microslide chamber. Panel C is the Cyto-3D-print containing parafilm wrapped microslide chamber in Eppendorf 5810R centrifuge bucket. Panel D shows balanced device in the centrifuge rotor
Evaluation of cell attachment using Cytospin with the Cyto-3D-print
Cells were placed in microslide chambers, centrifuged and measured for cell attachment with and without poly-l-lysine coated slides (Glavy et al. 2007; Kaur et al. 2010).
An 8-well microslide chamber was used and set up as follows:
Both poly-l-lysine and cytospinning | Poly-l-lysine only | Cytospinning only | Neither |
Both poly-l-lysine and cytospinning | Poly-l-lysine only | Cytospinning only | Neither |
The first and second columns were coated with poly-l-lysine a few hours before the experiment. Mitotic HeLa cells or suspension cells were first placed on the first and third columns. 10 μL of the cell suspension were placed in a hemocytometer to count how many cells were originally in each well. To ultimately test the function of the Cyto-3D-print, equal numbers of isolated mitotic cells were seeded in each well (approximately 5 × 104 HeLa cells). The cells were then spun in an Eppendorf 5810R Centrifuge for 3 min at 800 rpm (130 g) (Fig. 1). The microslide chamber fits inside the Cyto-3D-print, which fits inside the centrifuge bucket (Fig. 1). The centrifuge was balanced. After, mitotic HeLa cells or suspension cells were then placed in the second and fourth columns. After a 10-min incubation period at 37 °C, 5 % CO2, the cells were washed twice with 250 μL of PBS. Cells were counted and photographed under a light compound microscope. The percentage of cells that remained attached to the surface chamber over the original cell number for each well was calculated.
Immunofluorescence
Mitotic cell suspension was seeded and cyto-spun on pre-treated microslide chambers as described above. Cells were then fixed with 2 % formaldehyde in PBS for 30 min. Chambers were washed three times for 7 min with PBS and permeabilized with 0.2 % Triton X-100 for 5 min. After two additional PBS washings, cells were blocked with 5 % BSA in PBS for 1 h. Cells were washed with PBS three times (7 min each time), and incubated with designated dilutions of primary antibodies in 2 % BSA for 1 h. After PBS washing, cells were incubated with Alexa Fluor 488 or Alexa Fluor 555-conjugated secondary antibodies for 40 min (anti-mouse A21422, anti-goat A21467, Invitrogen, Carlsbad, CA, USA), which were previously diluted 1:100 in 2 % BSA-PBS. Based on the need of specific purposes, the incubation was repeated with a different pair of primary and corresponding secondary antibody. The nuclei in the cells were stained with DAPI. The prepared samples were visualized using a Zeiss LSM 5 Pascal confocal microscope.
Results and discussion
We have employed a simple application of plastic 3D prints for our experiments (Del Junco et al. 2015; Wong and Pfahnl 2014). Our aim is to overcome the problem of mitotic cell adherence to glass or plastic surfaces without expensive adaptors. Our results are clear and have been repeated several times with microslide chambers (Fig. 1). The plastic used was PLA from MakerBot (Nakagaito et al. 2009), which is a more biodegradable polymer compared to petroleum-based plastics. It holds its shape and is very sturdy compared to other plastics (Fig. 1b with microslide chamber). As envisioned, our Cyto-3D-print fits snuggly inside the centrifuge bucket without any gaps (Fig. 1c). In our procedure, we applied equal numbers of isolated mitotic cells in each well of the microslide chamber (Fig. 1d) and centrifuged. After, wells were washed twice with PBS then observed under a light microscope (Fig. 2a–d) and attached cells were counted for each condition (Fig. 2, bar chart of cell attachment). We found the combination of our Cyto-3D-print with poly-l-lysine coated slides had nearly 90 % attachment of the total cells added to each well, twice as good as coating alone (Fig. 2d). We observed no cracking or leakage in the parafilm wrapped microslide chambers. Our inserts are reusable and are more environmentally safe to discard than other metal based materials. Using conditions of Fig. 2d, Cyto-3D-print with poly-l-lysine coating, immunofluorescence shows the full range of mitotic cell attachment (Fig. 3). Condensed mitotic DNA is seen through the field of cells ranging from anaphase to telophase (Fig. 3a). Lamin B1 and MAb 414 mark only one interphase cell with full NE rim-staining (*, bottom left corner of panels B–D). Mitotic cells show different levels of NE rim-staining from partial to none signifying the breakdown of NE during mitosis.
Cyto-3D-print difference: Mitotic cells were placed in a microslide chamber and measured for cell attachment with and without poly-l-lysine coating after centrifugation. In Panel A, just cells alone; Panel B, cells cytospun in Cyto-3D-print; Panel C, cells with only poly-l-lysine coating; Panel D, cells with poly-l-lysine coating and then cytospun in Cyto-3D-print. Bar chart shows the percentage of cells that attached to chamber surfaces with * marking the highest value
Immunofluorescence microscopy analysis of mitotic HeLa cells with nuclear envelope rim-staining antibodies against Lamin B1 and MAb414. Panel A: DAPI stained DNA of cells at different phases of mitosis from anaphase to telophase have all attached to the surface. Panel B and C: nuclear envelope (NE) staining by αLamin B1 antibodies (green) and MAb414 staining (red) of the nuclear pore complexes. Mitotic cells show different levels of NE staining from partial to none. Panel D: merged signals showing the range of mitotic cells and one interphase (asterik) in the left corner with full NE rim staining and non-condensed DNA
In summary, our Cyto-3D-print offers an inexpensive flexible alternative to expensive metal plate adaptors with additional inserts. The use of microslide chambers has increased in recent years. Their reduced surface area saves on antibodies and reagents compared to larger six-well plates. We have developed Cyto-3D-print to bridge the gap between cytospinning and new slide technology. We hope this work stimulates more ideas to apply 3D printing to lab use.
References
Brown KD, Wood KW, Cleveland DW (1996) The kinesin-like protein CENP-E is kinetochore-associated throughout poleward chromosome segregation during anaphase-A. J Cell Sci 109(Pt 5):961–969
Campaner S, Kaldis P, Izraeli S, Kirsch IR (2005) Sil phosphorylation in a Pin1 binding domain affects the duration of the spindle checkpoint. Mol Cell Biol 25:6660–6672. doi:10.1128/MCB.25.15.6660-6672.2005
Choi HS, Anderson PJ (1979) Diagnostic cytology of cerebrospinal fluid by the cytocentrifuge method. Am J Clin Pathol 72:931–943
Del Junco M, Okhunov Z, Yoon R, Khanipour R, Juncal S, Abedi G, Lusch A, Landman J (2015) Development and initial porcine and cadaver experience with three-dimensional printing of endoscopic and laparoscopic equipment. J Endourol 29:58–62. doi:10.1089/end.2014.0280
Fischer-Friedrich E, Hyman AA, Julicher F, Muller DJ, Helenius J (2014) Quantification of surface tension and internal pressure generated by single mitotic cells. Sci Rep 4:6213. doi:10.1038/srep06213
Glavy JS, Krutchinsky AN, Cristea IM, Berke IC, Boehmer T, Blobel G, Chait BT (2007) Cell-cycle-dependent phosphorylation of the nuclear pore Nup107–160 subcomplex. Proc Natl Acad Sci USA 104:3811–3816. doi:10.1073/pnas.0700058104
Kaur S, White TE, DiGuilio AL, Glavy JS (2010) The discovery of a Werner helicase interacting protein (WHIP) association with the nuclear pore complex. Cell Cycle 9:3106–3111
Martinazzi M (1971) A slide centrifugation technic for concentrating blood leukocytes and nucleated cells from bone marrow blood. Am J Clin Pathol 56:719–722
Nakagaito AN, Fujimura A, Sakai T, Hama Y, Yano H (2009) Production of microfibrillated cellulose (MFC)-reinforced polylactic acid (PLA) nanocomposites from sheets obtained by a papermaking-like process. Compos Sci Technol 69:1293–1297. doi:10.1016/J.Compscitech.03.004
Nassar H, Ali-Fehmi R, Madan S (2003) Use of ThinPrep monolayer technique and cytospin preparation in urine cytology: a comparative analysis. Diagn Cytopathol 28:115–118. doi:10.1002/dc.10245
Piaton E, Hutin K, Faynel J, Ranchin MC, Cottier M (2004) Cost efficiency analysis of modern cytocentrifugation methods versus liquid based (Cytyc Thinprep) processing of urinary samples. J Clin Pathol 57:1208–1212. doi:10.1136/jcp.2004.018648
Shanholtzer CJ, Schaper PJ, Peterson LR (1982) Concentrated gram stain smears prepared with a cytospin centrifuge. J Clin Microbiol 16:1052–1056
Watson P (1966) A slide centrifuge: an apparatus for concentrating cells in suspension onto a microscope slide. J Lab Clin Med 68:494–501
Wong JY, Pfahnl AC (2014) 3D printing of surgical instruments for long-duration space missions. Aviat Space Environ Med 85:758–763
Acknowledgments
We thank David Peacock, Sandra Furnbach and Stevens’ office of Innovation and Entrepreneurship for support of this project. We thank Arthur B. Ritter and members of the Warren G. Wells BioRobotics Laboratory. A Shulman Scholarship and Innovation and Entrepreneurship Summer Scholarship supported MRC. National Institute on Aging (NIA) grant 1R21AG047433-01 and Stevens’ Ignition Grant Initiative supported ZL, YZ and JSG. We thank Thomas Cattabiani for critical reading of the manuscript.
Authors contribution
MRC, ZL, YZ and JSG designed the experiments. MRC, ZL, and YZ executed the experiments and finalized the figures. MGM printed the 3D insert. JSG wrote the initial draft and JSG, ZL, YZ and MRC revised the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
U.S. Provisional Patent Application No. 62/063,595.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary material 1 (MOV 166666 kb)
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Castroagudin, M.R., Zhai, Y., Li, Z. et al. Cyto-3D-print to attach mitotic cells. Cytotechnology 68, 1641–1645 (2016). https://doi.org/10.1007/s10616-015-9917-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10616-015-9917-2