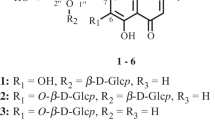
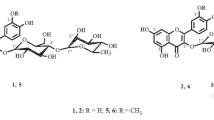
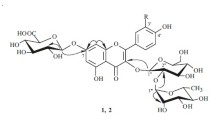
A total of 39 known compounds and two new flavonoid glycosides that were identified as isorhamnetin-3-O-(2″-acetyl)-β-D-glucopyranoside and isorhamnetin-3-O-(2″,6″-diacetyl)- β-D-glucopyranoside were isolated from florets of Calendula officinalis (Asteraceae). The distribution of phenolic compounds in morphological groups of C. officinalis was studied. It was found that peripheral florets had the highest flavonoid content (36.66 mg/g); tubular florets (9.95 mg/g) and leaves (9.65 mg/g), phenylpropanoids. Anthocyanins, among which cyanidin derivatives dominated, were identified for the first time in C. officinalis florets.
Similar content being viewed by others
References
B. P. Muley, S. S. Khadabadi, and N. B. Banarase, Trop. J. Pharm. Res., 8, 455 (2009).
O. A. Konovalova and K. S. Rybalko, Rastit. Resur., 26, 448 (1990).
A. Raal and K. Kirsipuu, Nat. Prod. Res., 25, 658 (2011).
R. Piccaglia, M. Marotti, G. Chiavari, and N. Gandini, Flavour Fragrance J., 12, 85 (1997).
L. F. N. A. Paim, M. Fontana, M. Winckler, A. A. Grando, T. L. Muneron, Jr., and W. A. Roman, Braz. J. Pharmacogn., 20, 974 (2010).
G. G. Zapesochnaya, S. Z. Ivanova, S. A. Medvedeva, and N. A. Tyukavkina, Chem. Nat. Compd., 14, 156 (1978).
L. O. A. Manguro, I. Ugi, R. Hermann, and P. Lemmen, Phytochemistry, 63, 497 (2003).
L. Swiatek and J. Gora, Herba Pol., 24, 187 (1978).
N. F. Komissarenko, V. T. Chernobai, and A. I. Derkach, Chem. Nat. Compd., 24, 675 (1988).
E. Vidal-Ollivier, R. Elias, F. Faure, A. Babadjamian, F. Crespin, G. Balansard, and G. Boudon, Planta Med., 55, 73 (1989).
G. Matysik, M. Wojciak-Kosior, and R. Paduch, J. Pharm. Biomed. Anal., 38, 285 (2005).
A. R. Bilia, M. C. Bergonzi, S. Gallori, G. Mazzi, and F. F. Vincieri, J. Pharm. Biomed. Anal., 30, 613 (2002).
I. Z. Matic, Z. Juranic, K. Savikin, G. Zdunic, N. Nadvinski, and D. Godevac, Phytother. Res., 26, 852 (2013).
L. V. Poludennyi, V. F. Sotnik, and E. E. Khlaptsev, Essential-oil and Medicinal Plants [in Russian], Kolos, Moscow, 1979, 286 pp.
N. K. Chirikova, D. N. Olennikov, and L. M. Tankhaeva, Russ. J. Bioorg. Chem., 36, 909 (2010).
R. E. Wrolstad, T. E. Acree, E. A. Decker, M. H. Penner, D. S. Reid, S. J. Schwartz, C. F. Shoemaker, and D. M. Smith (eds.), Current Protocols in Food Analytical Chemistry, Wiley & Sons, Inc., New York, 2001.
D. N. Olennikov, L. M. Tankhaeva, and A. B. Samuelsen, Chem. Nat. Compd., 42, 265 (2006).
A. Seyoum, K. Asres, and F. K. El-Fiky, Phytochemistry, 67, 2058 (2006).
D. N. Olennikov, S. V. Agafonova, A. V. Stolbikova, and A. V. Rokhin, Appl. Biochem. Microbiol., 47, 298 (2011).
Y. Nakajima, Y. Sato, and T. Konishi, Chem. Pharm. Bull., 55, 1222 (2007).
D. N. Olennikov, L. M. Tankhaeva, and S. V. Agafonova, Appl. Biochem. Microbiol., 47, 419 (2011).
V. A. Kurkin, Chem. Nat. Compd., 39, 123 (2003).
D. N. Olennikov, L. M. Tankhaeva, V. V. Partilkhaev, and A. V. Rokhin, Braz. J. Pharmacogn., 22, 490 (2012).
L. Lin and J. M. Harnly, J. Agric. Food Chem., 56, 10105 (2008).
D. N. Olennikov and V. V. Partilkhaev, J. Planar Chromatogr., 25, 30 (2012).
Y. B. Wang, J. X. Pu, H. Y. Ren, J. F. Zhao, S. X. Mei, Z. Y. Li, H. B. Zhang, and L. Li, Chin. Chem. Lett., 14, 1268 (2003).
A. Sakushima, S. Hisada, Y. Ogihara, and S. Nishibe, Chem. Pharm. Bull., 28, 1219 (1980).
Acknowledgment
The work was supported financially by the RFBR Project 12-03-31547(mol_a).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Prirodnykh Soedinenii, No. 5, September–October, 2013, pp. 717–723.
Rights and permissions
About this article
Cite this article
Olennikov, D.N., Kashchenko, N.I. New Isorhamnetin Glycosides and other Phenolic Compounds from Calendula officinalis . Chem Nat Compd 49, 833–840 (2013). https://doi.org/10.1007/s10600-013-0759-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10600-013-0759-x