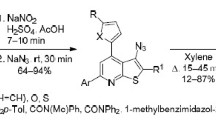
The reaction of aryl(hetaryl)-substituted 1,3-diketones with chloroacetamide gave the respective N-(3-oxoalkenyl)chloroacetamides, which were converted to 3-pyridinium-substituted pyridin-2(1Н)-ones upon heating with excess of pyridine in n-butanol. The decomposition of pyridinium salts with hydrazine hydrate resulted in new 4-aryl(hetaryl)-substituted 3-aminopyridin-2(1Н)-ones, which showed strong antiradical activity with respect to ABTS and DPPH radicals.
Similar content being viewed by others
References
Kusakabe, K.; Tada, Y.; Iso, Y.; Sakagami, M.; Morioka, Y.; Chomei, N.; Shinonome, S.; Kawamoto, K.; Takenaka, H.; Yasui, K.; Hamana, H.; Hanasaki, K. Bioorg. Med. Chem. 2013, 21, 2045.
Zhang, Y.-M.; Fan, X.; Chakaravarty, D.; Xiang, B.; Scannevin, R.H.; Huang, Z.; Ma, J.; Burke, S.L.; Karnachi, P.; Rhodesa, K.J.; Jackson, P.F. Bioorg. Med. Chem. Lett. 2008, 18, 409.
Ward, A.; Brogden, R.N.; Heel, R. C.; Speight, T.M.; Avery, G. S. Drugs, 1983, 26(6), 468.
Hoffman, J. M.; Wai, J. S.; Thomas, C. M.; Levin, R. B.; O'Brien, J. A.; Goldman, M. E. J. Med. Chem. 1992, 35 (21), 3784.
Saari, W. S.; Wai, J. S.; Fisher, T. E.; Thomas, C. M.; Hoffman, J. M.; Rooney, C. S.; Smith, A. M.; Jones, J. H.; Bamberger, D. L. J. Med. Chem. 1992, 35 (21), 3792.
Sanderson, P. E.J.; Cutrona, K. J.; Savage, K. L.; Naylor-Olsen, A. M.; Bickel, D. J.; Bohn, D. L.; Clayton, F. C.; Krueger, J. A.; Lewis, S.D.; Lucas, B. J.; Lyle, E. A.; Wallace, A. A.; Welsh, D. C.; Yan, Y. Bioorg. Med. Chem. Lett. 2003, 13, 1441.
Dragovich, P. S.; Prins, T. J.; Zhou, R.; Brown, E. L.; Maldonado, F. C.; Fuhrman, S. A.; Zalman, L. S.; Tuntland, T.; Lee, C. A.; Patick, A. K.; Matthews, D. A.; Hendrickson, T. F.; Kosa, M. B.; Liu, B.; Batugo, M. R.; Gleeson, J.-P. R.; Sakata, S. K.; Chen, L.; Guzman, M. C.; Meador, J. W.; Ferre, R. A.; Worland, S. T. J. Med. Chem. 2002, 45, 1607.
Verissimo, E.; Berry, N.; Gibbons, P.; Lurdes, M.; Cristiano, S.; Rosenthal, P. J.; Gut, J.; Ward, S. A., O'Neill, P. M. Bioorg. Med. Chem. Lett. 2008, 18, 4210.
Zhu, S.; Hudson, T. H.; Kyle, D. E.; Lin, A. J. J. Med. Chem., 2002, 45, 3491.
Li, Y. H.; Tseng, P.-S.; Evans, K. A.; Jaworski, J.-P.; Morrow, D. M.; Fries, H. E.; Wu, C. W.; Edwards, R. M.; Jin, J. Bioorg. Med. Chem. Lett. 2010, 20 (22), 6744.
Zhang, X.; Schmitt, A. C.; Decicco, C. P. Tetrahedron Letters. 2002, 43 (52), 9663.
Seger, H.; Geyer, A. Synthesis. 2006, 19, 3224.
Singh, B.; Lesher, G. Y. J. Heterocyclic Chem. 1990, 27 (7), 2085.
Damewood, J. R.; Edwards, P.D.; Feeney, S.; Gomes, B. C.; Steelman, G. B.; Tuthill, P. A.; Williams, J. C.; Warner, P.; Woolson, S. A.; Wolanin, D. J; Veale, C. A. J. Med. Chem. 1994, 37 (20), 3303.
Elzanate, A. M. Heterocycl. Comm. 2002, 8 (2), 145.
Elgemeie, G. E. H.; Hussain, B. A. W.; Elgemeie, G. H.; El-Ezbawy, S. R.; Ramiz, M. M.; Mansour, O. A. Org. Prep. and Proc. Int. 1991, 23 (5), 645.
Elgemeie, G. E. H.; El-Ezbawy, S. R.; Ali, H. A.; Mansour, A.-K. Org. Prep. and Proc. Int. 1994, 26 (4), 465.
Biediger, R. J.; Chen, Q.; Decker, E. R.; Holland, G. W.; Kassir, J. M.; Li, W.; Market, R. V.; Scott, I. L.; Wu, C.; Li, J. US Patent 2004/63955.
Muraoka, M.; Morishita, K.; Aida, N.; Tanaka, M.; Yuri, M.; Ohashi, N. US Patent 6300500.
Dominguez, C.; Harvey, T. S.; Liu, L.; Siegmund, A. US Patent 2005020592.
Hanfeld, V.; Leistner, S.; Wagner, G.; Lohmann, D.; Poppe, H.; Heer, S. Pharmazie. 1988, 43 (10), 677.
Fisyuk, A. S.; Bogza, Y. P.; Poendaev, N. V.; Goncharov, D. S. Chem. Heterocycl. Compd. 2010, 46, 844. [Khim. Geterotsikl. Soedin. 2010, 1044.]
Goncharov, D. S.; Kostuchenko, A. S.; Fisyuk, A. S. Chem. Heterocycl. Compd. 2009, 45, 793. [Khim. Geterotsikl. Soedin. 2009, 1005.]
Fisyuk, A. S.; Kulakov, I. V.; Goncharov, D. S.; Nikitina, O. S.; Bogza, Y. P.; Shatsauskas, A. L. Chem. Heterocycl. Compd., 2014, 50, 217. [Khim. Geterotsikl. Soedin. 2014, 241.]
Goncharov, D. S.; Garkushenko,·A. K.; Savelieva, A. P.; Fisyuk, A. S. ARKIVOC 2015, (v), 176.
Kulakov, I. V.; Nikitina, O. S.; Fisyuk, A. S.; Goncharov, D. S.; Shul'gau, Z. T., Gulyaev, A. E. Chem. Heterocycl. Compd., 2014, 50, 670. [Khim. Geterotsikl. Soedin. 2014, 729.]
Practicum on Organic Chemistry [in Russian]; V. I. Terenin; Ed.; BINOM. Laboratoriya Znanii: Moscow, 2010, p. 323.
Sneed, J. K.; Levine, R. J. Am. Chem. Soc. 1950, 72 (11), 5219.
Davis, B. R.; Hinds, M.G.; Ting, P. P. C. Austr. J. Chem., 1992, 45, 865.
Berti, F.; Bincoletto, S.; Donati, I.; Fontanive, G.; Fregonese, M.; Benedetti, F. Org. and Biomol. Chem., 2011, 9 (6), 1987.
Miller, N. J.; Rice-Evans, C.; Davies, M. J.; Gopinathan, V.; Milner, A. Clin. Sci. 1993, 84, 407.
Brand-Williams, W.; Cuvelier, M. E.; Berset, C. Lebensm.-Wiss. Technol. 1995, 28, 25.
Biometriya [in Russian]; Lakin, G. F., Ed.; Vysshaya shkola: Moscow, 1980, p. 293.
Plattner, S.; Erb, R.; Chervet, J.-P. and Oberacher, H. Anal. Bioanal. Chem. 2014, 406 (1), 213.
Kamal, Z.; Ullah, F.; Ayaz, M.; Sadiq, A; Ahmad, S.; Zeb, A.; Hussain, A.; Imran, M. Biol. Res. 2015; 48, 21. DOI: 10.1186/s40659-015-0011-1.
This study was performed with financial support from the Russian Foundation for Basic Research (project 15-53-45084 IND_a).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, 2015, 51(11/12), 991–996
Rights and permissions
About this article
Cite this article
Kulakov, I.V., Matsukevich, M.V., Shulgau, Z.T. et al. Synthesis and antiradical activity of 4-aryl(hetaryl)-substituted 3-aminopyridin-2(1Н)-ones. Chem Heterocycl Comp 51, 991–996 (2015). https://doi.org/10.1007/s10593-016-1809-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-016-1809-7