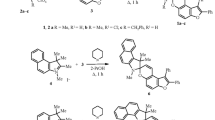
Formylation of 2-(2-hydroxyphenyl)benzoxazole gave 5-benzoxazolyl-substituted 4-hydroxyisophthalic aldehyde, which served as a precursor to the synthesis of novel photochromic spiro[indoline-benzopyrans] containing a benzoxazole group at position 8 of the benzopyran moiety. The obtained compounds possess photochromic properties in solution.

Similar content being viewed by others
References
M. B. Lukyanova, B. S. Lukyanov, I. V. Dorogan, S. O. Bezugliy, and M. S. Korobov, Khim. Geterotsikl. Soedin., 1951 (2013). [Chem. Heterocycl. Compd., 49, 1808 (2014)].
J. C. Crano and R. J. Guglielmetti (editors), Organic Photochromic and Thermochromic Compounds, Vol. I, II, Plenum Press, New York (2002).
B. L. Feringa and W. R. Browne (editors), Molecular Switches, Vol. 1, 2, 2nd ed., Wiley, Weinheim (2011).
V. I. Minkin, Chem. Rev., 104, 2751, (2004).
V. I. Minkin, Izv. Akad. Nauk, Ser. Khim., 673 (2008). [Russ. Chem. Bull., 57, 687 (2008).]
R. C. Bertelson, in: J. C. Crano and R. J. Guglielmetti (editors), Organic Photochromic and Thermochromic Compounds, Vol. 1, Plenum Press, New York (1999), p 11.
G. Bercovic, V. Krongauz, and V. Weiss, Chem. Rev., 100, 1741 (2000).
V. I. Minkin, in: B. L. Feringa and W. R. Browne (editors), Molecular Switches, Vol. 1, Ch. 2, Wiley (2011), p. 37.
S. M. Aldoshin, J. Photochem. Photobiol., A: Chem., 200, 19 (2008).
J. R. Chen, J. B. Wong, P. Y. Kuo, and D. Y. Yang, Org. Lett., 10, 4823 (2008).
B. Seefeldt, R. Kasper, M. Beining, J. Mattay, J. Arden-Jacob, N. Kemnitzer, K. H. Drexhage, M. Heilemann, and M. Sauer, Photochem. Photobiol. Sci., 9, 213 (2010).
S. A. Ahmed, M. Tanaka, H. Ando, K. Tawa, and K. Kimura, Tetrahedron, 60, 6029 (2004).
A. V. Chernishev, I. V. Dorogan, N. A. Voloshin, A. V. Metelitsa, and V. I. Minkin, Izv. Akad. Nauk, Ser. Khim., 447 (2011). [Russ. Chem. Bull., 60, 456 (2011).]
M. Tomasulo, E. Deniz, R. J. Alvarado, and F. M. Raymo, J. Phys. Chem. s, 112, 8038 (2008).
M. Inouye, Coord. Chem. Rev., 148, 265 (1996).
S. V. Paramonov, V. Lokshin, and O. A. Fedorova, J. Photochem. Phobiol., C., 12, 209 (2011).
S. Kume and H. Nishihara, Struct. Bond., 123, 79 (2007).
M. I. Zakharova, C. Coudret, V. Pimienta, J. C. Micheau, S. Delbaere, G. Vermeersch, A. V. Metelitsa, N. Voloshin, and V. I. Minkin, Photochem. Photobiol. Sci., 9, 199 (2010).
V. A. Barachevskii, Obzorn. Zh. Khim., 3, 58 (2013).
T. Deligeorgiev, S. Minkovska, B. Jeliazkova, and S. Rakovsky, Dyes Pigm., 53, 101 (2002).
S. Minkovska, B. Jeliazkova, E. Borisova, L. Avramov, and T. Deligeorgiev, J. Photochem. Photobiol., A, 163, 121 (2004).
B. G. Jeliazkova, S. Minkovska, and T. Deligeorgiev, J. Photochem. Photobiol., A, 171, 153 (2005).
N. Alhashimy, R. Byrne, S. Minkovska, and D. Diamond, Tetrahedron Lett., 50, 2573 (2009).
A. V. Chernyshev, N. A. Voloshin, I. M. Raskita, A. V. Metelitsa, and V. I. Minkin, J. Photochem. Photobiol., A, 184, 289 (2006).
N. A. Voloshin, E. B. Gaeva, A. V. Chernyshev, A. V. Metelitsa, and V. I. Minkin, Izv. Akad. Nauk, Ser. Khim., 156 (2009). [Russ. Chem. Bull., 58, 156 (2009).]
M. I. Zakharova, C. Coudret, V. Pimienta, J. C. Micheau, M. Sliwa, O. Poizat, G. Buntinx, S. Delbaere, G. Vermeersch, A. V. Metelitsa, N. Voloshin, and V. I. Minkin, Dyes Pigm., 89, 324 (2011).
W. H. Chen, Y. Xing, and Y. Pang, Org. Lett., 13, 1362 (2011).
J. Hobley, V. Malatesta, R. Millini, L. Montanari, and W. O. N. Parker, Phys. Chem. Chem. Phys., 1, 3259 (1999).
J. Hobley, V. Malatesta, W. Giroldini, and W. Stringo, Phys. Chem. Chem. Phys., 2, 53 (2000).
J. Hobley and V. Malatesta, Phys. Chem. Chem. Phys., 2, 57 (2000).
C. D. Gabbut, J. D. Hepworth, and B. M. Heron, Dyes Pigm., 42, 35 (1999).
A. V. Chernyshev, N. A. Voloshin, A. V. Metelitsa, V. V. Tkachev, S. M. Aldoshin, E. V. Solov’eva, I. A. Rostovtseva, and V. I. Minkin, J. Photochem. Photobiol., A, 265, 1 (2013).
E. Pottier, M. Sergent, R. Phan Tan Luu, and R. Guglielmetti, Bull. Soc. Chim. Belg., 101, 719 (1992).
C. Niu, Y. Song, and L. Yang, Chin. J. Chem., 27, 2001 (2009).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiya Geterotsiklicheskikh Soedinenii, No. 12, pp. 1959-1964, 2013.
*For Communication 39, see [1].
Rights and permissions
About this article
Cite this article
Voloshin, N.A., Chernyshev, A.V., Solov’eva, E.V. et al. Photo- and Thermochromic Spirans 40*. Spiropyrans based on 5-Benzoxazolyl-4-Hydroxyisophthalic Aldehyde. Chem Heterocycl Comp 49, 1815–1820 (2014). https://doi.org/10.1007/s10593-014-1434-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10593-014-1434-2