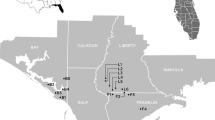
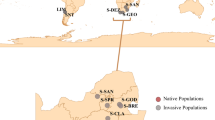
Abstract
Tallgrass prairie habitats within North America have suffered severe fragmentation and habitat loss as land has been converted for agricultural purposes. Habitat loss and fragmentation can affect gene flow and the genetic structure of insect populations. Neoconocephalus bivocatus is a prairie obligate katydid found only in isolated prairie patches. We compared genetic diversity and population differentiation using AFLP markers in N. bivocatus and N. robustus, a grassland generalist that is not isolated to prairie fragments and occupies a more contiguous range. Similar levels of genetic diversity were present within populations of both species. While population genetic structure was found in both species, there was no relationship between assigned genotypes and sampling localities. This genetic structure may instead be evidence of a past barrier to gene flow that has since been removed. Genetic differentiation within both species was low, with no evidence of a correlation with geographic distance, indicating neither species is dispersal limited at these distances. We see no significant reduction in genetic diversity or genetic differentiation within N. bivocatus when compared to N. robustus. We therefore conclude that while N. bivocatus utilizes a fragmented landscape, long-distance dispersal likely maintains gene flow between isolated prairie patches.





Similar content being viewed by others
References
Audacity Team (2008): Audacity (Version 1.3.13) [Computer program] http://audacityteam.org
Austin JD, Lougheed SC, Neidrauer L, Chek AA, Boag PT (2002) Cryptic lineages in a small frog: the post-glacial history of the spring peeper, Pseudacris crucifer (Anura: Hylidae). Mol Phylogenet Evol 25(2):316–329
Avise JC (2000) Phylogeography: the history and formation of species. Harvard University Press, Cambridge
Aymes ND, Mavoungou JF, De Stordeur E, Duvallet G (2009) Landscape, population structure and genetic diversity of Stomoxys calcitrans. Parasite 16(1):37–41
Bailey WJ, McCrae AWR (1978) The general biology and phenology of swarming in the East African Tettigoniid Ruspolia differens (Serville) (Orthoptera). J Nat Hist 12:259–288
Bandelt H, Forster P, Röhl A (1999) Median-joining networks for inferring intraspecific phylogenies. Mol Biol Evol 16(1):37–48
Berwaerts K, Van Dyck H, Van Dongen S, Matthysen E (1998) Morphological and genetic variation in the speckled wood butterfly (Pararge aegeria L) among differently fragmented landscapes. Neth. J Zool 48(3):241–253
Blair C, Weigel DE, Balazik M, Keeley AT, Walker FM, Landguth E, Cushman SA, Murphy M, Waits L, Balkenhol N. (2012). A simulation-based evaluation of methods for inferring linear barriers to gene flow. Mol Ecol Resour 12(5):822–833
Bonin A, Ehrich D, Manel S (2007) Statistical analysis of amplified fragment length polymorphism data: a toolbox for molecular ecologists and evolutionists. Mol Ecol 16:3737–3758
Brown JH, Kodric-Brown A (1977) Turnover rates in insular biogeography: effect of immigration on extinction. Ecology 58:445–449
Burger JC, Redak RA, Allen EB, Rotenberry JT, Allen MF (2003) Restoring arthropod communities in coastal sage scrub. Conserv Biol 17(2):460–467
Bush SL, Schul J (2010) Evolution of novel signal traits in the absence of female preferences in Neoconocephalus katydids (Orthoptera, Tettigoniidae). PloS one 5(8):e12457
Chaput-Bardy A, Lemaire C, Picard D, Secondi J (2008) In-stream and overland dispersal across a river network influences gene flow in a freshwater insect, Calopteryx splendens. Mol Ecol 17(15):3496–3505
Collinge SK, Palmer TM (2002) The influences of patch shape and boundary contrast on insect response to fragmentation in California grasslands. Landsc Ecol 17(7):647–656
Deily JA, Schul J (2004) Recognition of calls with exceptionally fast pulse rates: female phonotaxis in the genus Neoconocephalus (Orthoptera: Tettigoniidae). J Exp Biol 207(20):3523–3529
Deily JA, Schul J (2006) Spectral selectivity during phonotaxis: a comparative study in Neoconocephalus (Orthoptera: Tettigoniidae). J Exp Biol 209(9):1757–1764
Dhuyvetter H, Gaublomme E, Verdyck P, Desender K (2005) Genetic differentiation among populations of the salt marsh beetle Pogonus littoralis (Coleoptera: Carabidae): a comparison between Atlantic and Mediterranean populations. J Hered 96(4):381–387
Dobson AP, Bradshaw AD, Baker AÁ (1997) Hopes for the future: restoration ecology and conservation biology. Science 277(5325):515–522
Earl DA, von Holdt BM (2012) Structure Harvester: a website and program for visualizing STRUCTURE output and implementing the Evanno method. Conserv Genet Resour 4(2):359–361
Evanno G, Regnaut S, Goudet J (2005) Detecting the number of clusters of individuals using the software STRUCTURE: a simulation study. Mol Ecol 14(8):2611–2620
Frederick KH (2006) Phylogenetic structure of katydid (Orthoptera: Tettigoniidae) communities in Northeast Missouri grasslands. MS thesis, Truman State University, Kirksville
Frederick, KH (2013) Investigating an adaptive radiation in temperate Neoconocephalus (Orthoptera: Tettigoniidae). Dissertation, University of Missouri, Columbia
Garnier S, Alibert P, Audiot P, Prieur B, Rasplus JY (2004) Isolation by distance and sharp discontinuities in gene frequencies: implications for the phylogeography of an alpine insect species, Carabus solieri. Mol Ecol 13(7):1883–1897
Gauffre B, Mallez S, Chapuis MP, Leblois R, Litrico, I, Delaunay S, Badenhausser I (2015) Spatial heterogeneity in landscape structure influences dispersal and genetic structure: empirical evidence from a grasshopper in an agricultural landscape. Mol Ecol 24(8):1713–1728
Gilman RT, Behm JE (2011) Hybridization, species collapse, and species reemergence after disturbance to premating mechanisms of reproductive isolation. Evol Int J org Evol 65(9):2592–2605
Golden DM, Crist TO (1999) Experimental effects of habitat fragmentation on old-field canopy insects: community, guild and species responses. Oecologia 118(3):371–380
Greenfield MD (1990) Evolution of acoustic communication in the genus Neoconocephalus: discontinuous songs, synchrony, and interspecific interactions. In: Bailey WJ, Rentz DCF (eds) The tettigoniidae: biology, systematics and evolution. Springer, Berlin, pp 71–97
Hoerling M, Eischeid J, Kumar A, Leung, R, Mariotti, A, Mo, K, and Seager, R (2014) Causes and predictability of the 2012 Great Plains drought. B Am Meteorol Soc 95(2):269–282
Holland BR, Clarke AC, Meudt HM (2008) Optimizing automated AFLP scoring parameters to improve phylogenetic resolution. Syst Biol 57(3):347–366
Huson DH, Bryant D (2006) Application of phylogenetic networks in evolutionary studies. Mol Biol Evol 23(2):254–267
Jakobsson M, Rosenberg NA (2007) CLUMPP: a cluster matching and permutation program for dealing with label switching and multimodality in analysis of population structure. Bioinformatics 23(14):1801–1806
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C, Thierer T (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28(12):1647–1649
Keyghobadi N, Roland J, Strobeck C (2005) Genetic differentiation and gene flow among populations of the alpine butterfly, Parnassius smintheus, vary with landscape connectivity. Mol Ecol 14(7):1897–1909
Knutsen H, Rukke BA, Jorde PE, Ims RA (2000) Genetic differentiation among populations of the beetle Bolitophagus reticulatus (Coleoptera: Tenebrionidae) in a fragmented and a continuous landscape. Heredity 84(6):667–676
Korman AK, Mallet J, Goodenough JL, Graves JB, Hayes JL, Hendricks DE, Luttrell R, Pair SD, Wall M (1993) Population structure in Heliothis virescens (Lepidoptera: Noctuidae): an estimate of gene flow. Ann Entomol Soc Am 86(2):182–188
Krauss J, Schmitt T, Seitz A, Steffan-Dewenter I, Tscharntke T (2004) Effects of habitat fragmentation on the genetic structure of the monophagous butterfly Polyommatus coridon along its northern range margin. Mol Ecol 13(2):311–320
Kruess A, Tscharntke T (2000) Species richness and parasitism in a fragmented landscape: experiments and field studies with insects on Vicia sepium. Oecologia 122(1):129–137
Lamont BB, He T, Enright NJ, Krauss SL, Miller BP (2003) Anthropogenic disturbance promotes hybridization between Banksia species by altering their biology. J Evol Biol 16(4):551–557
Lande R (1993) Risks of population extinction from demographic and environmental stochasticity and random catastrophes. Am Nat 142:911–927
Lynch M, Milligan BG (1994) Analysis of population genetic structure with RAPD markers. Mol Ecol 3(2):91–99
Ming QL, Wang CZ (2006) Genetic differentiation of Helicoverpa armigera (Hübner) and H assulta (Guenée)(Lepidoptera: Noctuidae) based on AFLP markers. Insect Sci 13(6):437–444
Monaghan MT, Spaak P, Robinson CT, Ward JV (2001) Genetic differentiation of Baetis alpinus Pictet (Ephemeroptera: Baetidae) in fragmented alpine streams. Heredity 86(4):395–403
Ney G, Schul J (In prep) Population structure within the one-dimensional range of a coastal plain katydid
Ortego J, Aguirre MP, Cordero PJ (2010) Population genetics of Mioscirtus wagneri, a grasshopper showing a highly fragmented distribution. Mol Ecol 19(3):472–483
Paris M, Despres L (2012) Identifying insecticide resistance genes in mosquito by combining AFLP genome scans and 454 pyrosequencing. Mol Ecol 21(7):1672–1686
Paris M, Boyer S, Bonin A, Collado A, David JP, Despres L (2010) Genome scan in the mosquito Aedes rusticus: population structure and detection of positive selection after insecticide treatment. Mol Ecol 19(2):325–337
Peakall ROD, Smouse PE (2006) GENALEX 6: genetic analysis in Excel. Population genetic software for teaching and research. Mol Ecol Notes 6(1):288–295
Phillipsen IC, Lytle DA (2013) Aquatic insects in a sea of desert: population genetic structure is shaped by limited dispersal in a naturally fragmented landscape. Ecography 36(6):731–743
Phillipsen IC, Kirk EH, Bogan MT, Mims MC, Olden JD, Lytle DA (2015) Dispersal ability and habitat requirements determine landscape-level genetic patterns in desert aquatic insects. Mol Ecol 24(1):54–69
Pritchard JK, Wen X, Falush D (2010) Documentation for structure software: Version 23. University of Chicago, Chicago
Rickletts, TH, Dinerstein E, Olsen DM, Loucks CJ, Eichbaum W, Dellasala D, Kavanagh K, Hedao P, Hurley PT, Carney KM, Abell R, Walters S (1999) Terrestrial ecoregions of North America. Island Press, Washington DC
Roach JL, Stapp P, Van Horne B, Antolin MF (2001) Genetic structure of a metapopulation of black-tailed prairie dogs. J Mammal 82:946–959
Saarinen EV, Reilly PF, Austin JD (2016) Conservation genetics of an endangered grassland butterfly (Oarisma poweshiek) reveals historically high gene flow despite recent and rapid range loss. Insect Conserv Divers 9(6):517–528
Saccheri I, Kuussaari M, Kankare M, Vikman P, Fortelius W, Hanski I (1998) Inbreeding and extinction in a butterfly metapopulation. Nature 392(6675):491–494
Samson FB, Knopf FL, Ostlie WR (2004) Great plains ecosystems: past, present, and future. Wildlife Soc B 32(1):6–15
Schmitt T, Seitz A (2002) Postglacial distribution area expansion of Polyommatus coridon (Lepidoptera: Lycaenidae) from its Ponto-Mediterranean glacial refugium. Heredity 89(1):20–26
Schneider JC (1999) Dispersal of a highly vagile insect in a heterogeneous environment. Ecology 80(8):2740–2749
Schul J, Patterson AC (2003) What determines the tuning of hearing organs and the frequency of calls? A comparative study in the katydid genus Neoconocephalus (Orthoptera, Tettigoniidae). J Exp Biol 206(1):141–152
Seehausen O (2006) Conservation: losing biodiversity by reverse speciation. Curr Biol 16(9):334–337
Seehausen OLE, Takimoto G, Roy D, Jokela J (2008) Speciation reversal and biodiversity dynamics with hybridization in changing environments. Mol Ecol 17(1):30–44
Snyder RL, Frederick-Hudson KH, Schul J (2009) Molecular phylogenetics of the genus Neoconocephalus (Orthoptera, Tettigoniidae) and the evolution of temperate life histories. PloS one 4(9):e7203
Stevens VM, Verkenne C, Vandewoestijne S, Wesselingh RA, Baguette M (2006) Gene flow and functional connectivity in the natterjack toad. Mol Ecol 15(9):2333–2344
Stireman JO III, Nason JD, Heard SB (2005) Host-associated genetic differentiation in phytophagous insects: general phenomenon or isolated exceptions? Evidence from a goldenrod-insect community. Evol Int J org Evol 59(12):2573–2587
Stoner KJ, Joern A (2004) Landscape vs local habitat scale influences to insect communities from tallgrass prairie remnants. Ecol Appl 14(5):1306–1320
Taylor SJ, Downie DA, Paterson ID (2011) Genetic diversity of introduced populations of the water hyacinth biological control agent Eccritotarsus catarinensis (Hemiptera: Miridae). Biol Control 58(3):330–336
Tscharntke T, Greiler HJ (1995) Insect communities, grasses, and grasslands. Annu Rev Entomol 40(1):535–558
Tscharntke T, Steffan-Dewenter I, Kruess A, Thies C (2002) Contribution of small habitat fragments to conservation of insect communities of grassland-cropland landscapes. Ecol Appl 12(2):354–363
Vandergast AG, Bohonak AJ, Weissman DB, Fisher RN (2007) Understanding the genetic effects of recent habitat fragmentation in the context of evolutionary history: phylogeography and landscape genetics of a southern California endemic Jerusalem cricket (Orthoptera: Stenopelmatidae: Stenopelmatus). Mol Ecol 16(5):977–992
Vekemans X, Beauwens T, Lemaire M, Roldán-Ruiz I (2002) Data from amplified fragment length polymorphism (AFLP) markers show indication of size homoplasy and of a relationship between degree of homoplasy and fragment size. Mol Ecol 11(1):139–151
Walker TJ (1975) Stridulatory movements in eight species of Neoconocephalus (Tettigoniidae). J Insect Physiol 21(3):595–603
Walker TJ (2000) Neoconocephalus Singing Insects of North America. Available from http://entnemdept.ufl.edu/walker/buzz/g185a.htm. Accessed November, 2015<bib id="bib70">Watts K, Vanhala T, Connolly T, Cottrell J (2016) Striking the right balance between site and landscape-scale conservation actions for a woodland insect within a highly fragmented landscape: a landscape genetics perspective. Biol Conserv 195:146–155
Walker TJ, Whitesell JJ, Alexander RD (1973) The robust conehead: two widespread sibling species (Orthoptera: Tettigoniidae: Neoconocephalus “Robustus”). Ohio J Sci 73(6):321–330
Whitesell JJ (1969) Biology of United States coneheaded katydids of the genus Neoconocephalus (Orthoptera: Tettigoniidae). MS thesis, University of Florida, Gainesville
Wiesner KR, Habel JC, Gossner MM, Loxdale HD, Köhler G, Schneider AR, Tiedemann R, Weisser WW (2014). Effects of habitat structure and land-use intensity on the genetic structure of the grasshopper species Chorthippus parallelus. R Soc Open Sci 1(2):140133
Williams BL, Brawn JD, Paige KN (2003) Landscape scale genetic effects of habitat fragmentation on a high gene flow species: Speyeria idalia (Nymphalidae). Mol Ecol 12(1):11–20
Wright S (1943) Isolation by distance. Genetics 28(2):114
Young A, Boyle T, Brown T (1996) The population genetic consequences of habitat fragmentation for plants. Trends Ecol Evol 11(10):413–418
Zhang H, Hare MP (2012) Identifying and reducing AFLP genotyping error: an example of tradeoffs when comparing population structure in broadcast spawning versus brooding oysters. Heredity 108(6):616–625
Zhivotovsky LA (1999) Estimating population structure in diploids with multilocus dominant DNA markers. Mol Ecol 8(6):907–913
Acknowledgements
This work was supported by a grant of the National Science Foundation (IOS 1146878) and through a grant from the Missouri Department of Conservation. We thank Katy Frederick and Nathan Harness for assistance with specimen collection and recording males. We thank Lori Eggert, the editor, and two anonymous reviewers for valuable feedback on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10592_2017_987_MOESM1_ESM.eps
Supplementary Fig. 1 AFLP neighbor-joining network analyses produced from data sets each consisting of all 2015 individuals and one of the replicate sets of eight individuals form 2013. Letters ‘a’ and ‘c’ denote which plate samples were run on. Replicate samples always fell with their matching species cluster. Networks showed no reduction in the between species bootstrap results (>0.99). Bootstrap values within species clusters were low (<0.04), resulting in replicate samples falling in different places within the two analyses (EPS 5787 KB)
10592_2017_987_MOESM2_ESM.eps
Supplementary Fig. 2 (A) Neighbor-joining AFLP network and (B) median-joining haplotype map of 2013 samples. Colors indicate species-specific call assignments, N. robustus (red), N. bivocatus (blue), intermediate phenotype (green), and unknown call type (black). Numbered individuals are those that possess a mismatched AFLP cluster assignment and call phenotype. Within the median-joining CO1 haplotype network three primary haplotype clusters were observed with little genetic differentiation within clusters. Two were made up of primarily N. bivocatus individuals and one was made up of primarily N. robustus individuals. All individuals found in one of the two CO1 N. bivocatus clusters fell within the N. bivocatus AFLP cluster and all individuals in the N. robustus CO1 cluster were found in the N. robustus AFLP cluster. This concordance of nuclear and mitochondrial relationships held true even among individuals possessing the heterospecific call type within genetic clusters (EPS 6678 KB)
10592_2017_987_MOESM3_ESM.docx
Supplementary Table 1 N. robustus and N. bivocatus collection site abbreviations and names, coordinates, dates of collection, and numbers of each species collected (DOCX 89 KB)
10592_2017_987_MOESM4_ESM.docx
Supplementary Table 2 Estimated membership coefficient (Q) assignments for population 1 as measured from two independent Structure runs. Data sets consisted of all 2015 individuals and one of the replicate sets of eight individuals form 2013 (DOCX 38 KB)
Rights and permissions
About this article
Cite this article
Ney, G., Schul, J. Low genetic differentiation between populations of an endemic prairie katydid despite habitat loss and fragmentation. Conserv Genet 18, 1389–1401 (2017). https://doi.org/10.1007/s10592-017-0987-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10592-017-0987-x