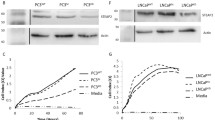
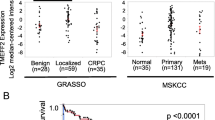
Abstract
Prostate adenocarcinoma is the second most frequent cancer worldwide and is one of the leading causes of male cancer-related deaths. However, it varies greatly in its behaviour, from indolent non-progressive disease to metastatic cancers with high associated mortality. The aim of this study was to identify predictive biomarkers for patients with localised prostate tumours most likely to progress to aggressive disease, to facilitate future tailored clinical treatment and identify novel therapeutic targets. The expression of 602 genes was profiled using oligoarrays, across three prostate cancer cell lines: CA-HPV-10, LNCaP and PC3, qualitatively identifying several potential prognostic biomarkers. Of particular interest was six transmembrane epithelial antigen of the prostate (STEAP) 1 and STEAP 2 which was subsequently analysed further in prostate cancer tissue samples following optimisation of an RNA extraction method from laser captured cells isolated from formalin-fixed paraffin-embedded biopsy samples. Quantitative analysis of STEAP1 and 2 gene expression were statistically significantly associated with the metastatic cell lines DU145 and PC3 as compared to the normal prostate epithelial cell line, PNT2. This expression pattern was also mirrored at the protein level in the cells. Furthermore, STEAP2 up-regulation was observed within a small patient cohort and was associated with those that had locally advanced disease. Subsequent mechanistic studies in the PNT2 cell line demonstrated that an over-expression of STEAP2 resulted in these normal prostate cells gaining an ability to migrate and invade, suggesting that STEAP2 expression may be a crucial molecule in driving the invasive ability of prostate cancer cells.




Similar content being viewed by others
Abbreviations
- BMP:
-
Bone morphogenic protein
- DISC:
-
Death inducing signalling complex
- ECM:
-
Extracellular matrix
- ERK:
-
Extracellular signal-regulated kinases
- FFPE:
-
Formalin fixed paraffin embedded
- H&E:
-
Haematoxylin and eosin
- HRL:
-
High risk localised
- LCM:
-
Laser capture microdissection
- LRL:
-
Low risk localised
- BPH:
-
Matrix metalloproteinase
- MMP:
-
Benign prostate hyperplasia
- MAPK:
-
Mitogen activated protein kinase
- PCa:
-
Prostate cancer
- PSA:
-
Prostate specific antigen
- TIMP:
-
Tissue inhibitor metalloproteinase
- TGF:
-
Transforming growth factor
- TRUS:
-
Transrectal ultrasound
- STEAP:
-
Six transmembrane epithelial antigen of the prostate
References
Cancer Statistics http://www.cancerresearchuk.org/cancerinfo/cancerstats/types/prostate/incidence/#age. Accessed 15 Mar 2013
Ihlaseh‐Catalano SM, Drigo SA, Jesus C, Domingues MAC, Trindade Filho JCS, Camargo JLV et al (2013) STEAP1 protein overexpression is an independent marker for biochemical recurrence in prostate carcinoma. Histopathology 63(5):678–685
Wright JL, Lange PH (2007) Newer potential biomarkers in prostate cancer. Rev Urol 9(4):207–213
D’Amico AV, Whittington R, Malkowicz SB, Schultz D, Blank K, Broderick GA et al (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy, or interstitial radiation therapy for clinically localized prostate cancer. JAMA 280(11):969–974
Partin AW, Mangold LA, Lamm DM, Walsh PC, Epstein JI, Pearson JD (2001) Contemporary update of prostate cancer staging nomograms (Partin Tables) for the new millennium. Urology. 58(6):843–848
Duffy M, McGowan P, Gallagher W (2008) Cancer invasion and metastasis: changing views. J Pathol 214(3):283–293
Frantz C, Stewart KM, Weaver VM (2010) The extracellular matrix at a glance. J Cell Sci 123(24):4195–4200
Gialeli C, Theocharis AD, Karamanos NK (2011) Roles of matrix metalloproteinases in cancer progression and their pharmacological targeting. FEBS J 278(1):16–27
Murphy G (2008) The ADAMs: signalling scissors in the tumour microenvironment. Nat Rev Cancer 8(12):932–941
Brehmer B, Biesterfeld S, Jakse G (2003) Expression of matrix metalloproteinases (MMP-2 and-9) and their inhibitors (TIMP-1 and-2) in prostate cancer tissue. Prostate Cancer Prostatic Dis 6(3):217–222
Pang ST, Flores-Morales A, Skoog L, Chuan YC, Nordstedt G, Pousette A (2004) Regulation of matrix metalloproteinase 13 expression by androgen in prostate cancer. Oncol Rep 11(6):1187–1192
Cao J, Chiarelli C, Richman O, Zarrabi K, Kozarekar P, Zucker S (2008) Membrane type 1 matrix metalloproteinase induces epithelial-to-mesenchymal transition in prostate cancer. J Biol Chem 283(10):6232–6240
Millimaggi D, Mari M, D’Ascenzo S, Carosa E, Jannini EA, Zucker S et al (2007) Tumor vesicle-associated CD147 modulates the angiogenic capability of endothelial cells. Neoplasia (New York, NY) 9(4):349
Wang L, Jin Y, Arnoldussen YJ, Jonson I, Qu S, Mælandsmo GM et al (2010) STAMP1 is both a proliferative and an antiapoptotic factor in prostate cancer. Cancer Res 70(14):5818
Korkmaz KS, Elbi C, Korkmaz CG, Loda M, Hager GL, Saatcioglu F (2002) Molecular cloning and characterization of STAMP1, a highly prostate-specific six transmembrane protein that is overexpressed in prostate cancer. J Biol Chem 277(39):36689–36696
Ohgami RS, Campagna DR, McDonald A, Fleming MD (2006) The Steap proteins are metalloreductases. Blood 108(4):1388
Yamamoto T, Tamura Y, Kobayashi J-i, Kamiguchi K, Hirohashi Y, Miyazaki A et al (2013) Six-transmembrane epithelial antigen of the prostate-1 plays a role for in vivo tumor growth via intercellular communication. Exp Cell Res 319(17):2617–2626
Moreaux J, Kassambara A, Hose D, Klein B (2012) STEAP1 is overexpressed in cancers: a promising therapeutic target. Biochem Biophys Res Commun 429(3):148–155
Gomes IM, Maia CJ, Santos CR (2012) STEAP proteins: from structure to applications in cancer therapy. Mol Cancer Res 10(5):573–587
Challita-Eid PM, Morrison K, Etessami S, An Z, Morrison KJ, Perez-Villar JJ et al (2007) Monoclonal antibodies to six-transmembrane epithelial antigen of the prostate-1 inhibit intercellular communication in vitro and growth of human tumor xenografts in vivo. Cancer Res 67(12):5798
Lalani EN, Laniado ME, Abel PD (1997) Molecular and cellular biology of prostate cancer. Cancer Metastasis Rev 16(1–2):29–66
Smith P, Rhodes NP, Shortland AP, Fraser SP, Djamgoz M, Ke Y et al (1998) Sodium channel protein expression enhances the invasiveness of rat and human prostate cancer cells. FEBS Lett 423(1):19–24
Bennett ES, Smith BA, Harper JM (2004) Voltage-gated Na + channels confer invasive properties on human prostate cancer cells. Pflügers Archiv 447(6):908–914
Laniado ME, Lalani EN, Fraser SP, Grimes JA, Bhangal G, Djamgoz M et al (1997) Expression and functional analysis of voltage-activated Na+ channels in human prostate cancer cell lines and their contribution to invasion in vitro. Am J Pathol 150(4):1213
Laniado ME, Fraser SP, Djamgoz M (2001) Voltage-gated K+ channel activity in human prostate cancer cell lines of markedly different metastatic potential: distinguishing characteristics of PC-3 and LNCaP cells. Prostate 46(4):262–274
Prevarskaya N, Skryma R, Bidaux G, Flourakis M, Shuba Y (2007) Ion channels in death and differentiation of prostate cancer cells. Cell Death Differ 14(7):1295–1304
De Mattia F, Gubser C, van Dommelen MMT, Visch HJ, Distelmaier F, Postigo A et al (2009) Human Golgi antiapoptotic protein modulates intracellular calcium fluxes. Mol Biol Cell 20(16):3638–3645
Kaarbo M, Kokk TI, Saatcioglu F (2007) Androgen signaling and its interactions with other signaling pathways in prostate cancer. BioEssays 29(12):1227–1238
Arnoldussen YJ, Saatcioglu F (2009) Dual specificity phosphatases in prostate cancer. Mol Cell Endocrinol 309(1):1–7
Krueger JS, Keshamouni VG, Atanaskova N, Reddy KB (2001) Temporal and quantitative regulation of mitogen-activated protein kinase (MAPK) modulates cell motility and invasion. Oncogene 20(31):4209
Reddy KB, Nabha SM, Atanaskova N (2003) Role of MAP kinase in tumor progression and invasion. Cancer Metastasis Rev 22(4):395–403
Royuela M, Arenas MI, Bethencourt FR, Fraile B, Paniagua R (2002) Regulation of proliferation/apoptosis equilibrium by mitogen-activated protein kinases in normal, hyperplastic, and carcinomatous human prostate. Hum Pathol 33(3):299–306
Gioeli D, Mandell JW, Petroni GR, Frierson HF, Weber MJ (1999) Activation of mitogen-activated protein kinase associated with prostate cancer progression. Cancer Res 59(2):279
Uzgare AR, Kaplan PJ, Greenberg NM (2003) Differential expression and/or activation of P38MAPK, erk1/2, and jnk during the initiation and progression of prostate cancer. Prostate 55(2):128–139
Merlo LM, Pepper JW, Reid BJ, Maley CC (2006) Cancer as an evolutionary and ecological process. Nat Rev Cancer 6(12):924–935
Grunewald T, Herbst SM, Heinze J, Burdach S (2011) Understanding tumor heterogeneity as functional compartments-superorganisms revisited. J Transl Med 9(1):79
Acknowledgments
Funding for this work was kindly provided by grants to SHD from the National Institute for Social Care and Health Research (NISCHR) and the Swansea Prostate Cancer Research Fund.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10585_2014_9679_MOESM1_ESM.doc
Supplemental Fig. 1: List of all genes screened in the oligoarray analysis. A comprehensive list of genes analysed in the oligoarray analysis displayed based on the signalling pathway array type they were positioned upon. Supplementary material 1 (DOC 91 kb)
10585_2014_9679_MOESM2_ESM.doc
Supplemental Fig. 2: Oligoarray gene spot intensities for all four cell lines. Using oligoarrays tailored to four various pathways: WNT signalling pathway, TGFβ/BMP signalling pathway, ECM and adhesion molecules, and PCa biomarkers; over 600 genes were analysed for qualitative identification of potential biomarkers of aggressive PCa. Supplementary material 2 (DOC 72 kb)
Rights and permissions
About this article
Cite this article
Whiteland, H., Spencer-Harty, S., Morgan, C. et al. A role for STEAP2 in prostate cancer progression. Clin Exp Metastasis 31, 909–920 (2014). https://doi.org/10.1007/s10585-014-9679-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-014-9679-9