Abstract
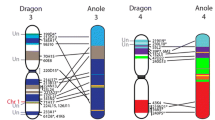
Reptiles, as the sister group to birds and mammals, are particularly valuable for comparative genomic studies among amniotes. The Australian central bearded dragon (Pogona vitticeps) is being developed as a reptilian model for such comparisons, with whole-genome sequencing near completion. The karyotype consists of 6 pairs of macrochromosomes and 10 pairs microchromosomes (2n = 32), including a female heterogametic ZW sex microchromosome pair. Here, we present a molecular cytogenetic map for P. vitticeps comprising 87 anchor bacterial artificial chromosome clones that together span each macro- and microchromosome. It is the first comprehensive cytogenetic map for any non-avian reptile. We identified an active nucleolus organizer region (NOR) on the sub-telomeric region of 2q by mapping 18S rDNA and Ag-NOR staining. We identified interstitial telomeric sequences in two microchromosome pairs and the W chromosome, indicating that microchromosome fusion has been a mechanism of karyotypic evolution in Australian agamids within the last 21 to 19 million years. Orthology searches against the chicken genome revealed an intrachromosomal rearrangement of P. vitticeps 1q, identified regions orthologous to chicken Z on P. vitticeps 2q, snake Z on P. vitticeps 6q and the autosomal microchromosome pair in P. vitticeps orthologous to turtle Pelodiscus sinensis ZW and lizard Anolis carolinensis XY. This cytogenetic map will be a valuable reference tool for future gene mapping studies and will provide the framework for the work currently underway to physically anchor genome sequences to chromosomes for this model Australian squamate.






Similar content being viewed by others
Abbreviations
- APTX :
-
Aprataxin
- ATP5A1 :
-
ATP synthase, H+ transporting, mitochondrial F1 complex, alpha subunit 1, cardiac muscle
- BAC:
-
Bacterial artificial chromosome
- BCL6 :
-
B cell CLL/lymphoma 6
- BLAST:
-
Basic Local Alignment Search Tool
- BLAT:
-
BLAST-like alignment tool
- CA10 :
-
Carbonic anhydrase X
- CHD1 :
-
Chromodomain helicase DNA-binding protein 1
- CTBP2 :
-
C-terminal binding protein 2
- CTNNB1 :
-
Catenin (cadherin-associated protein), beta 1, 88 kDa
- DAPI:
-
4′,6-Diamidino-2-phenylindole
- DDX58 :
-
DEAD (Asp-Glu-Ala-Asp) box polypeptide 58
- DMRT1 :
-
Doublesex and mab-3-related transcription factor 1
- dUTP:
-
2′-Deoxyuridine 5′-triphosphate
- EIF3H :
-
Eukaryotic translation initiation factor 3, subunit H
- FAM83B :
-
Family with sequence similarity 83, member B
- FBRSL1 :
-
Fibrosin-like 1
- FISH:
-
Fluorescence in situ hybridization
- GHR :
-
Growth hormone receptor
- GMPPA :
-
GDP-mannose pyrophosphorylase A
- HCRTR2 :
-
Hypocretin (orexin) receptor 2
- HMGCLL1 :
-
3-Hydroxymethyl-3-methylglutaryl-CoA lyase-like 1
- IBSP :
-
Integrin-binding sialoprotein
- IPO7 :
-
Importin 7
- IQSEC3 :
-
IQ motif and Sec7 domain 3
- KAT2B :
-
K(lysine) acetyltransferase 2B
- KAT7 :
-
K(lysine) acetyltransferase 7
- KLF6 :
-
Kruppel-like factor 6
- NAV2 :
-
Neuron navigator 2
- NOR:
-
Nucleolus organizer region
- NPRL3 :
-
Nitrogen permease regulator-like 3
- PSMA2 :
-
Proteasome (prosome, macropain) subunit, alpha type, 2
- RAB5A :
-
RAB5A, member RAS oncogene family
- rDNA:
-
Ribosomal DNA
- RRM1 :
-
Ribonucleotide reductase M1
- SRY :
-
Sex-determining region Y
- TAX1BP1 :
-
Tax1 (human T-cell leukemia virus type I) binding protein 1
- TMEM41B :
-
Transmembrane protein 41B
- TNFRSF11B :
-
Tumor necrosis factor receptor superfamily, member 11b
- TTN :
-
Titin
- WAC :
-
WW domain-containing adaptor with coiled coil
- ZNF143 :
-
Zinc finger protein 143
References
Ahl E (1926) Neue Eidechsen und Amphibien. Zoologischer Anzeiger 67:186–192
Alföldi J, Di Palma F, Grabherr M, Williams C, Kong L, Mauceli E, Russell P, Lowe CB, Glor RE, Jaffe JD (2011) The genome of the green anole lizard and a comparative analysis with birds and mammals. Nature 477:587–591
Chinwalla AT, Cook LL, Delehaunty KD, Fewell GA, Fulton LA, Fulton RS, Graves TA, Hillier LDW, Mardis ER, McPherson JD (2002) Initial sequencing and comparative analysis of the mouse genome. Nature 420:520–562
Crawford NG, Faircloth BC, McCormack JE, Brumfield RT, Winker K, Glenn TC (2012) More than 1000 ultraconserved elements provide evidence that turtles are the sister group of archosaurs. Biol Lett 8:783–786
Dalloul RA, Long JA, Zimin AV, Aslam L, Beal K, Bouffard P, Burt DW, Crasta O, Crooijmans RPMA, Cooper K (2010) Multi-platform next-generation sequencing of the domestic turkey (Meleagris gallopavo): genome assembly and analysis. PLoS Biology 8:e1000475
Dolezel J, Bartos J, Voglmayr H, Greilhuber J (2003) Nuclear DNA content and genome size of trout and human. Cytometry Part A: The Journal of the International Society for Analytical Cytology 51:127
Ezaz T, Moritz B, Waters P, Marshall Graves JA, Georges A, Sarre SD (2009a) The ZW sex microchromosomes of an Australian dragon lizard share no homology with those of other reptiles or birds. Chromosome Res 17:965–973
Ezaz T, Quinn AE, Miura I, Sarre SD, Georges A, Graves JAM (2005) The dragon lizard Pogona vitticeps has ZZ/ZW micro-sex chromosomes. Chromosome Res 13:763–776
Ezaz T, Quinn AE, Sarre SD, O’Meally D, Georges A, Marshall Graves JA (2009b) Molecular marker suggests rapid changes of sex-determining mechanisms in Australian dragon lizards. Chromosome Res 17:91–98
Fujita MK, Edwards SV, Ponting CP (2011) The Anolis lizard genome: an amniote genome without isochores. Genome Biol Evol 3:974
Hedges SB, Dudley J, Kumar S (2006) TimeTree: a public knowledge-base of divergence times among organisms. Bioinformatics 22:2971–2972
Hillier LDW, Miller W, Birney E, Warren W, Hardison RC, Ponting CP, Bork P, Burt DW, Groenen MAM, Delany ME (2004) Sequence and comparative analysis of the chicken genome provide unique perspectives on vertebrate evolution. Nature 432:695–716
Howell WM, Black DA (1980) Controlled silver-staining of nucleolus organizer regions with a protective colloidal developer: a 1-step method. Experientia 36:1014–1015
Hugall AF, Foster R, Hutchinson M, Lee MSY (2008) Phylogeny of Australasian agamid lizards based on nuclear and mitochondrial genes: implications for morphological evolution and biogeography. Biol J Linn Soc 93:343–358
Kasai F, O’Brien PCM, Ferguson-Smith MA (2012) Reassessment of genome size in turtle and crocodile based on chromosome measurement by flow karyotyping: close similarity to chicken. Biol Lett 8:631–635
Kawagoshi T, Uno Y, Matsubara K, Matsuda Y, Nishida C (2009) The ZW micro-sex chromosomes of the Chinese soft-shelled turtle (Pelodiscus sinensis, Trionychidae, Testudines) have the same origin as chicken chromosome 15. Cytogenet Genome Res 125:125–131
Kawai A, Nishida-Umehara C, Ishijima J, Tsuda Y, Ota H, Matsuda Y (2007) Different origins of bird and reptile sex chromosomes inferred from comparative mapping of chicken Z-linked genes. Cytogenet Genome Res 117:92–102
Kohn M, Högel J, Vogel W, Minich P, Kehrer-Sawatzki H, Graves JAM, Hameister H (2006) Reconstruction of a 450-My-old ancestral vertebrate protokaryotype. Trends Genet 22:203–210
Kuraku S, Ishijima J, Nishida-Umehara C, Agata K, Kuratani S, Matsuda Y (2006) cDNA-based gene mapping and GC 3 profiling in the soft-shelled turtle suggest a chromosomal size-dependent GC bias shared by sauropsids. Chromosome Res 14:187–202
MacCulloch RD, Upton DE, Murphy RW (1996) Trends in nuclear DNA content among amphibians and reptiles. Comp Biochem Physiol B Biochem Mol Biol 113:601–605
Martinez PA, Ezaz T, Valenzuela N, Georges A, Marshall Graves JA (2008) An XX/XY heteromorphic sex chromosome system in the Australian chelid turtle Emydura macquarii: a new piece in the puzzle of sex chromosome evolution in turtles. Chromosome Res 16:815–825
Matsubara K, Kuraku S, Tarui H, Nishimura O, Nishida C, Agata K, Kumazawa Y, Matsuda Y (2012) Intra-genomic GC heterogeneity in sauropsids: evolutionary insights from cDNA mapping and GC3 profiling in snake. BMC Genomics 13:604
Matsuda Y, Nishida-Umehara C, Tarui H, Kuroiwa A, Yamada K, Isobe T, Ando J, Fujiwara A, Hirao Y, Nishimura O (2005) Highly conserved linkage homology between birds and turtles: bird and turtle chromosomes are precise counterparts of each other. Chromosome Res 13:601–615
Meyne J, Baker RJ, Hobart HH, Hsu T, Ryder OA, Ward OG, Wiley JE, Wurster-Hill DH, Yates TL, Moyzis RK (1990) Distribution of non-telomeric sites of the (TTAGGG)n telomeric sequence in vertebrate chromosomes. Chromosoma 99:3–10
Mikkelsen TS, Wakefield MJ, Aken B, Amemiya CT, Chang JL, Duke S, Garber M, Gentles AJ, Goodstadt L, Heger A (2007) Genome of the marsupial Monodelphis domestica reveals innovation in non-coding sequences. Nature 447:167–177
Nakatani Y, Takeda H, Kohara Y, Morishita S (2007) Reconstruction of the vertebrate ancestral genome reveals dynamic genome reorganization in early vertebrates. Genome Res 17:1254–1265
O’Meally D, Miller H, Patel H, Marshall Graves JA, Ezaz T (2009) The first cytogenetic map of the tuatara, Sphenodon punctatus. Cytogenet genome res 127:213–223
O’Meally D, Ezaz T, Georges A, Sarre SD, Graves JA (2012) Are some chromosomes particularly good at sex? Insights from amniotes. Chromosome Res 20:7–19
Olmo E, Signorino G (2005) Chromorep: a reptile chromosomes database. http://chromorep.univpm.it. Accessed 7 May 2013
Organ CL, Janes DE (2008) Evolution of sex chromosomes in Sauropsida. Integr Comp Biol 48:512–519
Patel VS, Ezaz T, Deakin JE, Marshall Graves JA (2010) Globin gene structure in a reptile supports the transpositional model for amniote α- and β-globin gene evolution. Chromosome Res 18:897–907
Paull D, Williams EE, Hall WP (1976) Lizard karyotypes from the Galapagos Islands: chromosomes in phylogeny and evolution. Breviora 441:1–31
Pokorná M, Giovannotti M, Kratochvíl L, Caputo V, Olmo E, Ferguson-Smith MA, Rens W (2012) Conservation of chromosomes syntenic with avian autosomes in squamate reptiles revealed by comparative chromosome painting. Chromosoma 121:409–418
Pokorná M, Giovannotti M, Kratochvíl L, Kasai F, Trifonov VA, O’Brien PCM, Caputo V, Olmo E, Ferguson-Smith MA, Rens W (2011) Strong conservation of the bird Z chromosome in reptilian genomes is revealed by comparative painting despite 275 million years divergence. Chromosoma 120:455–468
Porter CA, Hamilton MJ, Sites Jr JW, Baker RJ (1991) Location of ribosomal DNA in chromosomes of squamate reptiles: systematic and evolutionary implications. Herpetologica 47:271–280
Quinn AE, Georges A, Sarre SD, Guarino F, Ezaz T, Graves JAM (2007) Temperature sex reversal implies sex gene dosage in a reptile. Science 316:411–411
Ruiz-Herrera A, Nergadze S, Santagostino M, Giulotto E (2008) Telomeric repeats far from the ends: mechanisms of origin and role in evolution. Cytogenet Genome Res 122:219–228
Sarre SD, Ezaz T, Georges A (2011) Transitions between sex-determining systems in reptiles and amphibians. Annu Rev Genom Hum Genet 12:391–406
Sarre SD, Georges A, Quinn A (2004) The ends of a continuum: genetic and temperature-dependent sex determination in reptiles. BioEssays 26:639–645
Shedlock AM, Edwards SV (2009) Amniotes (amniota). In: Hedges SB, Kumar S (eds) The timetree of life. Oxford University Press, New York, pp 375–379
Srikulnath K, Nishida C, Matsubara K, Uno Y, Thongpan A, Suputtitada S, Apisitwanich S, Matsuda Y (2009) Karyotypic evolution in squamate reptiles: comparative gene mapping revealed highly conserved linkage homology between the butterfly lizard (Leiolepis reevesii rubritaeniata, Agamidae, Lacertilia) and the Japanese four-striped rat snake (Elaphe quadrivirgata, Colubridae, Serpentes). Chromosome Res 17:975–986
Uno Y, Nishida C, Tarui H, Ishishita S, Takagi C, Nishimura O, Ishijima J, Ota H, Kosaka A, Matsubara K (2012) Inference of the protokaryotypes of amniotes and tetrapods and the evolutionary processes of microchromosomes from comparative gene mapping. PLoS One 7:e53027
Wakefield MJ, Graves JAM (2003) The kangaroo genome. EMBO Rep 4:143–147
Warren WC, Clayton DF, Ellegren H, Arnold AP, Hillier LDW, Künstner A, Searle S, White S, Vilella AJ, Fairley S (2010) The genome of a songbird. Nature 464:757–762
Warren WC, Hillier LDW, Graves JAM, Birney E, Ponting CP, Grützner F, Belov K, Miller W, Clarke L, Chinwalla AT (2008) Genome analysis of the platypus reveals unique signatures of evolution. Nature 453:175–183
Witten G (1983) Some karyotypes of Australian agamids (Reptilia: Lacertilia). Aust J Zool 31:533–540
Acknowledgments
This work was funded by an ARC DP awarded to SD, AG and Scott Edwards, as was the purchase of the P. vitticeps BAC Library. This work was undertaken by MY as a Bachelor of Applied Science Honours with the Institute of Applied Ecology at the University of Canberra. We would like to thank Jacqui Richardson and Alistair Zealey for their care of captive animals and Juliet Ward for laboratory assistance.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Walther Traut.
Electronic supplementary materials
Below is the link to the electronic supplementary material.
ESM 1
(DOC 117 kb)
Rights and permissions
About this article
Cite this article
Young, M.J., O’Meally, D., Sarre, S.D. et al. Molecular cytogenetic map of the central bearded dragon, Pogona vitticeps (Squamata: Agamidae). Chromosome Res 21, 361–374 (2013). https://doi.org/10.1007/s10577-013-9362-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10577-013-9362-z