Abstract
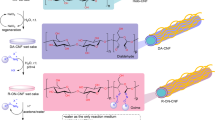
We report a ‘one-pot’ synthetic approach for TEMPO (2,2,6,6-tetramethyl-piperidin-1-oxyl) oxidized cellulose nanofibrils (TOCN) surface amidation by coupling up to 70% of superficial carboxylic units in TOCN with long alkyl chain primary amines (dodecylamine and octadecylamine) using TBTU [O-(1H-benzotriazol-1-yl)-N,N,N’,N’-tetramethyluronium tetrafluoroborate] uronium salt as coupling agent. Raw TOCNs were produced from commercial microcrystalline cellulose and have an elongated highly tangled morphology with nanometric diameters and lengths up to several micrometers with a crystallinity of 62%. TOCN´s degree of oxidation was 1.25 mmol –COOH groups/g cellulose. The TOCN-amidated products named TOCN-AMDC12 and TOCN-AMDC18 depending upon to the primary amine used for the synthesis (dodecylamine –C12– for the former and octadecylamine –C18– for the latter), were characterized using field emission scanning electron microscopy (FESEM), Fourier transform infrared spectroscopy with attenuated total reflectance and X-ray diffraction. After amidation the hydrated gel-like TOCNs become a cream-colored gel insoluble in water and soluble in toluene. IR data indicates formation of an amide bond after the reaction, and FESEM images suggest a slight increase in fibril width as a result of the long-chain amide derivatives grafted onto the TOCN’s surface. Thermal stability and hydrophobicity of the products were determined using thermogravimetrical analysis and contact angle (CA) measurements. The TOCN-amidated products showed improved thermal properties with maximum decomposition temperatures of 347 and 358 °C, for TOCN-AMDC12 and TOCN-AMDC18 respectively, when compared to the raw TOCN (Td 340 °C); and high hydrophobicity with CA of 61° and 67°, respectively. Finally, the proposed method for TOCN amidation has advantages over current amidation approaches in that it is performed in aqueous media, does not require heating and occurs rapidly (2 h).










Similar content being viewed by others
References
Abdul Khalil HPS, Davoudpour Y, Islam MN et al (2014) Production and modification of nanofibrillated cellulose using various mechanical processes: a review. Carbohydr Polym 99:649–665. doi:10.1016/j.carbpol.2013.08.069
Araki J, Wada M, Kuga S (2001) Steric stabilization of a cellulose microcrystal suspension by poly(ethylene glycol) grafting. Langmuir 17:21–27. doi:10.1021/la001070m
Basiuk (Golovataya-Dzhymbeeva) EV, Ochoa-Olmos O, Contreras-Torres FF et al (2011) “Green” functionalization of pristine multi-walled carbon nanotubes with long-chain aliphatic amines. J Nanosci Nanotechnol 11:5546–5554. doi:10.1166/jnn.2011.3431
Benkaddour A, Journoux-Lapp C, Jradi K et al (2014) Study of the hydrophobization of TEMPO-oxidized cellulose gel through two routes: amidation and esterification process. J Mater Sci 49:2832–2843. doi:10.1007/s10853-013-7989-y
Bulpitt P, Aeschlimann D (1999) New strategy for chemical modification of hyaluronic acid: preparation of functionalized derivatives and their use in the formation of novel biocompatible hydrogels. J Biomed Mater Res 47:152–169
Chinchilla R, Dodsworth DJ, Nájera C, Soriano JM (2000) Polymer-bound TBTU as a new solid-supported reagent for peptide synthesis. Tetrahedron Lett 41:2463–2466
Coseri S, Biliuta G, Simionescu BC et al (2013) Oxidized cellulose—survey of the most recent achievements. Carbohydr Polym 93:207–215. doi:10.1016/j.carbpol.2012.03.086
Davidson TC, Newman RH, Ryan MJ (2004) Variations in the fibre repeat between samples of cellulose I from different sources. Carbohydr Res 339:2889–2893. doi:10.1016/j.carres.2004.10.005
Dufresne A (2012) Nanocellulose: from nature to high performance tailored materials. Walter De Gruyter Incorporated, Hawthorne
Follain N, Montanari S, Jeacomine I et al (2008) Coupling of amines with polyglucuronic acid: evidence for amide bond formation. Carbohydr Polym 74:333–343. doi:10.1016/j.carbpol.2008.02.016
Follain N, Marais M-F, Montanari S, Vignon MR (2010) Coupling onto surface carboxylated cellulose nanocrystals. Polymer (Guildf) 51:5332–5344. doi:10.1016/j.polymer.2010.09.001
Fraczyk J, Kolesinska B, Relich I, Kaminski ZJ (2013) Cellulose functionalysed with grafted oligopeptides. In: Van De Ven TGM (ed) Cellulose—Medical, pharmaceutical and electronic applications. InTech. doi:10.5772/52302
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21:885–896. doi:10.1007/s10570-013-0030-4
Fujisawa S, Okita Y, Fukuzumi H et al (2011) Preparation and characterization of TEMPO-oxidized cellulose nanofibril films with free carboxyl groups. Carbohydr Polym 84:579–583. doi:10.1016/j.carbpol.2010.12.029
Fujisawa S, Saito T, Isogai A (2012) Nano-dispersion of TEMPO-oxidized cellulose/aliphatic amine salts in isopropyl alcohol. Cellulose 19:459–466. doi:10.1007/s10570-011-9648-2
Fukuzumi H, Saito T, Okita Y, Isogai A (2010) Thermal stabilization of TEMPO-oxidized cellulose. Polym Degrad Stab 95:1502–1508. doi:10.1016/j.polymdegradstab.2010.06.015
Gandini A, Belgacem MN (2015) The surface and in-depth modification of cellulose fibers. In: Rojas O (ed) Cellulose chemistry and properties: fibers, nanocelluloses and advanced materials, vol 271. Advances in Polymer Science. Springer International Publishing, pp 169–206. doi:10.1007/12_2015_305
Gao N, Yan Y (2012) Nanoscale characterisation of surface wettability based on nanoparticles. Nanoscale 4:2202–2218. doi:10.1039/c2nr11736c
Habibi Y, Chanzy H, Vignon MR (2006) TEMPO-mediated surface oxidation of cellulose whiskers. Cellulose 13:679–687. doi:10.1007/s10570-006-9075-y
Habibi Y, Lucia LA, Rojas OJ (2010) Cellulose nanocrystals: chemistry, self-assembly, and applications. Chem Rev 110:3479–3500
Hubbe MA, Rojas OJ, Lucia LA et al (2008) Cellulosic nanocomposites: a review. BioResources 3:929–980
Isogai A, Kato Y (1998) Preparation of polyuronic acid from cellulose by TEMPO-mediated oxidation. Cellulose 5:153–164. doi:10.1023/A:1009208603673
Isogai A, Saito T, Fukuzumi H (2011) TEMPO-oxidized cellulose nanofibers. Nanoscale. Nanoscale 3:71–85. doi:10.1039/c0nr00583e
Johnson RK, Zink-Sharp A, Glasser WG (2011) Preparation and characterization of hydrophobic derivatives of TEMPO-oxidized nanocelluloses. Cellulose 18:1599–1609. doi:10.1007/s10570-011-9579-y
Klemm D, Kramer F, Moritz S et al (2011) Nanocelluloses: a new family of nature-based materials. Angew Chem Int Ed Engl 50:5438–5466. doi:10.1002/anie.201001273
Lasseuguette E (2008) Grafting onto microfibrils of native cellulose. Cellulose 15:571–580. doi:10.1007/s10570-008-9200-1
LeVan SL (1989) Thermal degradation. In: Schniewind AP (ed) Concise encyclopedia of wood and wood-based materials. MIT Press, Boston, pp 271–273
Lif A, Stenstad P, Syverud K et al (2010) Fischer–Tropsch diesel emulsions stabilised by microfibrillated cellulose and nonionic surfactants. J Colloid Interface Sci 352:585–592. doi:10.1016/j.jcis.2010.08.052
Mandal A, Chakrabarty D (2011) Isolation of nanocellulose from waste sugarcane bagasse (SCB) and its characterization. Carbohydr Polym 86:1291–1299. doi:10.1016/j.carbpol.2011.06.030
Missoum K, Belgacem MN, Bras J (2013) Nanofibrillated cellulose surface modification: a review. Materials (Basel) 6:1745–1766. doi:10.3390/ma6051745
Montalbetti CAGN, Falque V (2005) Amide bond formation and peptide coupling. Tetrahedron 61:10827–10852. doi:10.1016/j.tet.2005.08.031
Moon RJ, Martini A, Nairn J et al (2011) Cellulose nanomaterials review: structure, properties and nanocomposites. Chem Soc Rev 40:3941–3994. doi:10.1039/C0CS00108B
Morais JPS, Rosa MDF, de Souza Filho MDSM et al (2013) Extraction and characterization of nanocellulose structures from raw cotton linter. Carbohydr Polym 91:229–235. doi:10.1016/j.carbpol.2012.08.010
Morán JI, Alvarez VA, Cyras VP, Vazquez A (2008) Extraction of cellulose and preparation of nanocellulose from sisal fibers. Cellulose 15:149–159. doi:10.1007/s10570-007-9145-9
Okita Y, Saito T, Isogai A (2010) Entire surface oxidation of various cellulose microfibrils by TEMPO-mediated oxidation. Biomacromolecules 11:1696–1700. doi:10.1021/bm100214b
Okita Y, Fujisawa S, Saito T, Isogai A (2011) TEMPO-oxidized cellulose nanofibrils dispersed in organic solvents. Biomacromolecules 12:518–522. doi:10.1021/bm101255x
Pretsch E, Bühlmann P, Affolter C (2013) Structure determination of organic compounds: tables of spectral data, third edit. Springer, New York
Rehman N, de Miranda MIG, Rosa SML et al (2014) Cellulose and nanocellulose from maize straw: an insight on the crystal properties. J Polym Environ 22:252–259. doi:10.1007/s10924-013-0624-9
Saito T, Isogai A (2004) TEMPO-mediated oxidation of native cellulose. The effect of oxidation conditions on chemical and crystal structures of the water-insoluble fractions. Biomacromolecules 5:1983–1989. doi:10.1021/bm0497769
Saito T, Okita Y, Nge TT et al (2006) TEMPO-mediated oxidation of native cellulose: microscopic analysis of fibrous fractions in the oxidized products. Carbohydr Polym 65:435–440. doi:10.1016/j.carbpol.2006.01.034
Sèbe G, Ham-Pichavant F, Ibarboure E et al (2012) Supramolecular structure characterization of cellulose II nanowhiskers produced by acid hydrolysis of cellulose i substrates. Biomacromolecules 13:570–578. doi:10.1021/bm201777j
Segal L, Creely JJ, Martin AE, Conrad CM (1959) An empirical method for estimating the degree of crystallinity of native cellulose using the x-ray diffractometer. Text Res J 29:786–794. doi:10.1177/004051755902901003
Sehgal D, Vijay IK (1994) A method for the high efficiency of water-soluble carbodiimide-mediated amidation. Anal Biochem 218:87–91
Shen DK, Gu S (2009) The mechanism for thermal decomposition of cellulose and its main products. Bioresour Technol 100:6496–6504. doi:10.1016/j.biortech.2009.06.095
Taubner T, Čopíková J, Havelka P, Synytsya A (2013) Preparation of amidated derivatives of monocarboxy cellulose. Cellulose 20:2045–2055. doi:10.1007/s10570-013-9938-y
Tesoro G (1986) Cellulose chemistry and its applications.In: Nevell TP, Zeronian SH (eds) Journal of Polymer Science Part C: Polymer Letters, Halsted (Wiley), New York, 1985, 552 pp. pp 294–295
Toffey A, Glasser WG (2001) Chitin derivatives III Formation of amidized homologs of chitosan. Cellulose 8:35–47. doi:10.1023/A:1016695522968
Tsuboi M (1957) Infrared spectrum and crystal structure of cellulose. J Polym Sci 25:159–171. doi:10.1002/pol.1957.1202510904
Valeur E, Bradley M (2009) Amide bond formation: beyond the myth of coupling reagents. Chem Soc Rev 38:606–631. doi:10.1039/b701677h
Wada M, Kondo T, Takeshi O, Wada M (2003) Thermally induced crystal transformation from cellulose Iα to Iβ.pdf. Polym J 35:155–159. doi:10.1295/polymj.35.155
Wertz J-L, Bédué O, Mercier JP (2010) Cellulose science and technology. EPFL Press, Switzerland
Yurkshtovich TL, Alinovskaya Va (2008) Sorption of aliphatic amines by monocarboxycellulose from aqueous and water-ethanol solutions. Colloid J 70:377–383. doi:10.1134/S1061933X08030174
Zabivalova NM, Bochek AM, Kalyuzhnaya LM et al (2003) Carboxymethyl cellulose amides and their properties. Russ J Appl Chem 76:1998–2002. doi:10.1023/B:RJAC.0000022456.23088.b6
Zhang X, Shi F, Niu J et al (2008) Superhydrophobic surfaces: from structural control to functional application. J Mater Chem 18:621. doi:10.1039/b711226b
Zhu L, Kumar V, Banker GS (2001) Examination of oxidized cellulose as a macromolecular prodrug carrier: preparation and characterization of an oxidized cellulose-phenylpropanolamine conjugate. Int J Pharm 223:35–47
Acknowledgments
We acknowledge financial support from ECOPETROL trough the Cooperation Agreement No. 5211794 - 2016. In addition, infrastructural support from the Central Research Laboratory Facility at Guatiguará Campus and the Vicerrectoría de Investigación at Universidad Industrial de Santander is greatly appreciated.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gómez, F.N., Combariza, M.Y. & Blanco-Tirado, C. Facile cellulose nanofibrils amidation using a ‘one-pot’ approach. Cellulose 24, 717–730 (2017). https://doi.org/10.1007/s10570-016-1174-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-016-1174-9