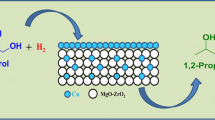
Hydrogenolysis of biomass-derived glycerol is an alternative route to sustainable production of propylene glycol. Cu–ZnO catalysts were prepared by coprecipitation with a range of Cu/Zn atomic ratio (0.6–2.0) and examined in glycerol hydrogenolysis to propylene glycol at 453–513 K and 4.2 MPa H2. These catalysts possess acid and hydrogenation sites required for bifunctional glycerol reaction pathways, most likely involving glycerol dehydration to acetol and glycidol intermediates on acidic ZnO surfaces, and their subsequent hydrogenation on Cu surfaces. Glycerol hydrogenolysis conversions and selectivities depend on Cu and ZnO particle sizes. Smaller ZnO and Cu domains led to higher conversions and propylene glycol selectivities, respectively. A high propylene glycol selectivity (83.6%), with a 94.3% combined selectivity to propylene glycol and ethylene glycol (also a valuable product) was achieved at 22.5% glycerol conversion at 473 K on Cu–ZnO (Cu/Zn = 1.0) with relatively small Cu particles. Reaction temperature effects showed that optimal temperatures (e.g. 493 K) are required for high propylene glycol selectivities, probably as a result of optimized adsorption and transformation of the reaction intermediates on the catalyst surfaces. These preliminary results provide guidance for the synthesis of more efficient Cu–ZnO catalysts and for the optimization of reaction parameters for selective glycerol hydrogenolysis to produce propylene glycol.




Similar content being viewed by others
References
R.D. Cortright, M. Sanchez-Castillo and J.A. Dumesic, Appl. Catal. B 39 (2002) 353.
J. Chaminand, L. Djakovitch, P. Gallezot, P. Marion, C. Pinel and C. Rosier, Green Chem. 6 (2004) 359.
M.A. Dasari, P.-P. Kiatsimkul, W.R. Sutterlin and G.J. Suppes, Appl. Catal. A 281 (2005) 225.
A. Perosa and P. Tundo, Ind. Eng. Chem. Res. 44 (2005) 8535.
C.-W. Chiu, M.A. Dasari, W.R. Sutterlin and G.J. Suppes, Ind. Eng. Chem. Res. 45 (2006) 791.
Y. Kusunoki, T. Miyazawa, K. Kunimori, K. Tomishige, Catal. Commun. 6 (2005) 645.
B. Casale and A.M. Gomze U.S. Patent 5 (1993) 214.
T. Miyazawa, Y. Kusunoki, K. Kunimori and K.J. Tomishige Catalysis 240 (2006) 213.
C. Montassier, J.M. Dumas, P. Granger and J. Barbier, Appl. Catal. A 121 (1995) 231.
G.J. de A. A. Soler-Illia, R.J. Candal, A.E. Regazzoni and M.A. Blesa, Chem. Mater. 9 (1997) 184.
T. Shishido, M. Yamamoto, D. Li, Y. Tian, H. Morioka, M. Honda, T. Sano and K. Takehira, Appl. Catal. A 303 (2006) 62.
A. Guinier, Theorie et Technique de la Radiocristallographie, 3rd edn., Paris, 1964, p. 482.
M.R. Nimlos, S.J. Blanksby, X. Qian, M.E. Himmel and D.K. Johnson, J. Phys. Chem. A 110 (2006) 6145.
Q. Li, L. Gao, W. Luan and D. Yan, J. Inorg. Mater (China). 14 (1999) 813.
P.C. Hiemenz, Principles of Colloid and Surface Chemistry. New York: Marcel Dekker Inc., 1986.
Acknowledgments
This work was supported by the National Basic Research Project of China (No.2006CB806100) and National Natural Science Foundation of China (Grant No. 20673005, 20573004). This work was also supported in part by Program for New Century Excellent Talents in University (NECT-05–0010), State Education Ministry. The authors thank Professor E. Min of SINOPEC for his insightful discussions.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wang, S., Liu, H. Selective hydrogenolysis of glycerol to propylene glycol on Cu–ZnO catalysts. Catal Lett 117, 62–67 (2007). https://doi.org/10.1007/s10562-007-9106-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-007-9106-9