Abstract
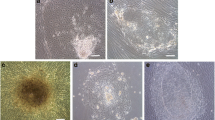
A novel approach for stem cell generation is the attempt to induce conversion of the adult somatic cells into pluripotent stem cells so called induced pluripotent stem cells (iPSCs) by introducing specific transcription factors. iPSCs have two essential cell characteristics, they are pluripotent and posses long term cell-renewal capacity. Additionally, iPSCs can be derived from patient-specific somatic cells, thus bypassing ethical and immunological issues. The aim of our study was to reprogram long-term cryopreserved human neonatal fibroblasts by new method using lipid nano-particle technology (Lipofectamine 3000 reagent transfection system) in combination with Epi 5 reprogramming vectors. Obtained iPSCs were characterized by several sophisticated methods of molecular biology and microscopy. Distinct colonies of iPSCs started to appear by day 20 after reprogramming. The presence of iPSCs colonies was proved by alkaline phosphatase (AP) live staining. After manual picking the colonies and their subsequent passaging, they did not lose ability to form embryoid bodies, they were positive for AP, Tra-1-60, and SSEA-5. Moreover, obtained iPSCs expressed pluripotency markers Oct4, Sox2 and Nanog, and the expression levels of chondrogenic, osteogenic and adipogenic markers were significantly higher in comparison to control (p < 0.05). In summary, we have demonstrated that long-term cryopreserved human neonatal fibroblasts can be reprogrammed into iPSCs and after further analysis concerns on their biological safety they may be used as patient-specific cells in regenerative medicine.










Similar content being viewed by others
References
Chen G, Gulbranson DR, Hou Z et al (2011) Chemically defined conditions for human iPSC derivation and culture. Nat Methods 8:424–429. doi:10.1038/nmeth.1593
Gai H, Leung EL, Costantino PD et al (2009) Generation and characterization of functional cardiomyocytes using induced pluripotent stem cells derived from human fibroblasts. Cell Biol Int 33:1184–1193. doi:10.1016/j.cellbi.2009.08.008
Hou P, Li Y, Zhang X et al (2013) Pluripotent stem cells induced from mouse somatic cells by small-molecule compounds. Science 341:651–654. doi:10.1126/science.1239278
Jia F, Wilson KD, Sun N et al (2010) A nonviral minicircle vector for deriving human iPS cells. Nat Methods 7:197–199. doi:10.1038/nmeth.1426
Kahler DJ, Ahmad FS, Ritz A et al (2013) Improved methods for reprogramming human dermal fibroblasts using fluorescence activated cell sorting. PLoS ONE 8:e59867. doi:10.1371/journal.pone.0059867
Kim D, Kim C-H, Moon J-I et al (2009) Generation of human induced pluripotent stem cells by direct delivery of reprogramming proteins. Cell Stem Cell 4:472–476. doi:10.1016/j.stem.2009.05.005
Loh Y-H, Agarwal S, Park I-H et al (2009) Generation of induced pluripotent stem cells from human blood. Blood 113:5476–5479. doi:10.1182/blood-2009-02-204800
Lu X, Zhao T (2013) Clinical therapy using iPSCs: hopes and challenges. Genomics Proteomics Bioinform 11:294–298. doi:10.1016/j.gpb.2013.09.002
Manzini S, Viiri LE, Marttila S, Aalto-Setälä K (2015) A comparative view on easy to deploy non-integrating methods for patient-specific iPSC production. Stem Cell Rev 11:900–908. doi:10.1007/s12015-015-9619-3
Masip M, Veiga A, Belmonte JCI, Simon C (2010) Reprogramming with defined factors: from induced pluripotency to induced transdifferentiation. Mol Hum Reprod 16:856–868. doi:10.1093/molehr/gaq059
Miyazaki S, Yamamoto H, Miyoshi N et al (2012) Emerging methods for preparing iPS cells. Jpn J Clin Oncol 42:773–779. doi:10.1093/jjco/hys108
Nie B, Wang H, Laurent T, Ding S (2012) Cellular reprogramming: a small molecule perspective. Curr Opin Cell Biol 24:784–792. doi:10.1016/j.ceb.2012.08.010
Okita K, Yamanaka S (2011) Induced pluripotent stem cells: opportunities and challenges. Philos Trans R Soc B Biol Sci 366:2198–2207. doi:10.1098/rstb.2011.0016
Okita K, Nakagawa M, Hyenjong H et al (2008) Generation of mouse induced pluripotent stem cells without viral vectors. Science 322:949–953. doi:10.1126/science.1164270
Okita K, Matsumura Y, Sato Y et al (2011) A more efficient method to generate integration-free human iPS cells. Nat Methods 8:409–412. doi:10.1038/nmeth.1591
Okita K, Yamakawa T, Matsumura Y et al (2013) An efficient nonviral method to generate integration-free human-induced pluripotent stem cells from cord blood and peripheral blood cells. Stem Cells 31:458–466. doi:10.1002/stem.1293
Repiská V, Varga I, Lehocký I et al (2010) Biological and morphological characterization of human neonatal fibroblast cell culture B-HNF-1. Biologia. doi:10.2478/s11756-010-0095-6
Sayed N, Liu C, Wu JC (2016) Translation of human-induced pluripotent stem cells. J Am Coll Cardiol 67:2161–2176. doi:10.1016/j.jacc.2016.01.083
Soldner F, Hockemeyer D, Beard C et al (2009) Parkinson’s disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell 136:964–977. doi:10.1016/j.cell.2009.02.013
Takahashi K, Yamanaka S (2006) Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126:663–676. doi:10.1016/j.cell.2006.07.024
Warren L, Manos PD, Ahfeldt T et al (2010) Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7:618–630. doi:10.1016/j.stem.2010.08.012
Woltjen K, Michael IP, Mohseni P et al (2009) piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature 458:766–770. doi:10.1038/nature07863
Yu J, Vodyanik MA, Smuga-Otto K et al (2007) Induced pluripotent stem cell lines derived from human somatic cells. Science 318:1917–1920. doi:10.1126/science.1151526
Yu J, Hu K, Smuga-Otto K et al (2009) Human induced pluripotent stem cells free of vector and transgene sequences. Science 324:797–801. doi:10.1126/science.1172482
Funding
The study was supported by the grant of the Slovak Research and Development Agency (No. APVV-14-0032).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Csobonyeiova, M., Krajciova, L., Nicodemou, A. et al. Induction of pluripotency in long-term cryopreserved human neonatal fibroblasts in feeder-free condition. Cell Tissue Bank 18, 45–52 (2017). https://doi.org/10.1007/s10561-016-9602-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-016-9602-5