Abstract
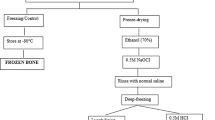
Processed cancellous bone has been regarded as one alternative for the treatment of bone defects. In order to avoid immunogenic effects and preserve the natural properties of the bone, the optimal processing method should be determined. To observe the influence of hydrogen peroxide on the mineral status and mechanical properties of cancellous bone for various time periods and find the optimal processing time. Cancellous bone granules from bovine femur condyles were treated with 30% hydrogen dioxide for 0, 12, 24, 36, 48, 60 and 72 h separately. The microstructure and mineral content of the granules were evaluated by ash analysis, Micro-CT, scanning electron micrograph and energy dispersive X-ray. The biomechanical properties were analyzed by applying cranial-caudal compression in a materials testing machine. With increasing exposure to hydrogen peroxide, the BMD and BMC of granules gradually decreased, and the Ca/P molar ratios clearly increased (P < 0.05). Meanwhile, the mineral content of the granules increased from 48.5 ± 1.3 to 79.5 ± 2.1%. Substantial decreases in the strength of the granules were observed, and after 48 h severe decreases were noted. The decrease in strength was also evident after normalizing the parameters to the cross-sectional area. Granules of bovine cancellous bone matrix should be processed by hydrogen peroxide for 12 to 36 h to fulfill the basic requirements of a bone tissue engineering scaffold. These granules could potentially be useful during orthopedic operations.





Similar content being viewed by others
Abbreviations
- SEM:
-
Scanning electron micrograph
- EDS:
-
Energy dispersive X-ray
- BMD:
-
Bone mineral density
- BMC:
-
Bone mineral content
- BV/TV:
-
Bone volume fraction
- Tb. Th:
-
Trabecular thickness
- Tb. SP:
-
Trabecular separation
- DA:
-
Direction anisotropy
- WW:
-
Wet weight
- DW:
-
Dry weight
- AW:
-
Ash weight
References
Accorsi-Mendonça T, Conz MB, Barros TC, Sena LA, Soares Gde A, Granjeiro JM (2008) Physicochemical characterization of two deproteinized bovine xenografts. Braz Oral Res 22:5–10
Breitbart AS, Grande DA, Kessler R, Ryaby JT, Fitzsimmons RJ, Grant RT (1998) Tissue engineered bone repair of calvarial defects using cultured periosteal cells. Plast Reconstr Surg 101:567–574
Dehghani SN, Bigham AS, Torabi Nezhad S, Shafiei Z (2008) Effect of bovine fetal growth plate as a new xenograft in experimental bone defect healing: radiological, histopathological and biomechanical evaluation. Cell Tissue Bank 9:91–99
Ejersted C, Andreassen T, Hauge EM, Melsen F, Oxlund HT (1995) Parathyroid hormone (1–34) increases vertebral bone mass, compressive strength, and quality in old rats. Bone 17:507–511
Ejersted C, Oxlund H, Eriksen EF, Andreassen TT (1998) Withdrawal of parathyroid hormone treatment causes rapid resorption of newly formed. Bone 23(1):43–52
Guizzardi S, Raspanti M, Martini D, Scandroglio R, Govoni P, Ruggeri A (1995) Low-temperature heat-deproteinated compact bone to heal large bone defects. Biomaterials 16:931–936
Hak DJ (2007) The use of osteoconductive bone graft substitutes in orthopaedic trauma. J Am Acad Orthop Surg 15:525–536
Hofmann C, Schädel-Höpfner M, Berns T, Sitter H, Gotzen L (2003) Influence of processing and sterilization on the mechanical properties of pins made from bovine cortical bone. Unfallchirurg 106:478–482
Hosokawa A (2006) Ultrasonic pulse waves in cancellous bone analyzed by finite-difference time-domain methods. Ultrasonics Suppl 1:e227–e231
Ignatius AA, Betz O, Augat P, Claes LE (2001) In vivo investigations on composites made of resorbable ceramics and poly (lactide) used as bone graft substitutes. J Biomed Mater Res 58:701–709
Kadiyala S, Young RG, Thiede MA, Bruder SP (1997) Culture expanded canine mesenchymal stem cells possess osteochondrogenic potential in vivo and in vitro. Cell Transplant 6:125–134
Khan Y, Yaszemski MJ, Mikos AG, Laurencin CT (2008) Tissue engineering of bone: material and matrix considerations. J Bone Joint Surg Am 90(Suppl 1):36–42
Kneser U, Stangenberg L, Ohnolz J, Buettner O, Stern-Straeter J, Möbest D et al (2006) Evaluation of processed bovine cancellous bone matrix seeded with syngenic osteoblasts in a critical size calvarial defect rat model. J Cell Mol Med 10:695–707
Kretlow JD, Mikos AG (2007) Review: mineralization of synthetic polymer scaffolds for bone tissue engineering. Tissue Eng 13:927–938
Meyer S, Floerkemeier T, Windhagen H (2008) Histological osseointegration of Tutobone(R): first results in human. Arch Orthop Trauma Surg 128:539–544
Monem AS, ElBatal HA, Khalil EM, Azooz MA, Hamdy YM (2008) In vivo behavior of bioactive phosphate glass–ceramics from the system P2O5-Na2O-CaO containing TiO2. J Mater Sci Mater Med 19:1097–1108
Nyman JS, Reyes M, Wang X (2005) Effect of ultrastructural changes on the toughness of bone. Micron 36(7–8):566–582
Ogose A, Kondo N, Umezu H, Hotta T, Kawashima H, Tokunaga K et al (2006) Histological assessment in grafts of highly purified beta-tricalcium phosphate (OSferion) in human bones. Biomaterials 27:1542–1549
Ohgushi H, Miyake J, Tateishi T (2003) Mesenchymal stem cells and bioceramics: strategies to regenerate the skeleton. Novartis Found Symp 249:118–127
Schantz JT, Hutmacher DW, Lam CX, Brinkmann M, Wong KM, Lim TC et al (2003) Repair of calvarial defects with customised tissue-engineered bone grafts II. Evaluation of cellular efficiency and efficacy in vivo. Tissue Eng 9(Suppl 1):127–139
Stützle H, Hallfeldt K, Mandelkow H, Kessler S, Schweiberer L (1998) Bone regeneration stimulated by bone substitute materials. Orthopade 27:118–125
Trentz OA, Hoerstrup SP, Sun LK, Bestmann L, Platz A, Trentz OL (2003) Osteoblasts response to allogenic and xenogenic solvent dehydrated cancellous bone in vitro. Biomaterials 24:3417–3426
Urabe K, Itoman M, Toyama Y, Yanase Y, Iwamoto Y, Ohgushi H et al (2007) Current trends in bone grafting and the issue of banked bone allografts based on the fourth nationwide survey of bone grafting status from 2000 to 2004. J Orthop Sci 12:520–525
Van Heest A, Swiontkowski M (1999) Bone-graft substitutes. Lancet 353(Suppl 1):28–29
Wenz B, Oesch B, Horst M (2001) Analysis of the risk of transmitting bovine spongiform encephalopathy through bone grafts derived from bovine bone. Biomaterials 22:1599–1606
Acknowledgements
This work was supported by a research grant from the National High Technology Research and Development Program of P. R. China (Grant No. 01Z079).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, D., Bi, L., Meng, G. et al. Mineral status and mechanical properties of cancellous bone exposed to hydrogen peroxide for various time periods. Cell Tissue Bank 12, 51–58 (2011). https://doi.org/10.1007/s10561-009-9161-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-009-9161-0