Abstract
Purpose & Methods
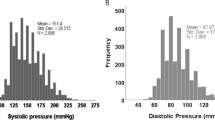
Not all hypertensive patients respond well to ACE inhibition. Here we determined whether renin-angiotensin system (RAS) phenotyping, i.e., the measurement of renin or ACE, can predict the individual response to RAS blockade, either chronically (enalapril vs. enalapril + candesartan) or acutely (enalapril ± hydrochlorothiazide, HCT).
Results
Chronic enalapril + candesartan induced larger renin rises, but did not lower blood pressure (BP) more than enalapril. Similar observations were made for enalapril + HCT vs. enalapril when given acutely. Baseline renin predicted the peak changes in BP chronically, but not acutely. Baseline ACE levels had no predictive value. Yet, after acute drug intake, the degree of ACE inhibition, like Δrenin, did correlate with ΔBP. Only the relationship with Δrenin remained significant after chronic RAS blockade. Thus, a high degree of ACE inhibition and a steep renin rise associate with larger acute responses to enalapril. However, variation was large, ranging >50 mm Hg for a given degree of ACE inhibition or Δrenin. The same was true for the relationships between Δrenin and ΔBP, and between baseline renin and the maximum reduction in BP in the chronic study.
Conclusions
Our data do not support that RAS phenotyping will help to predict the individual BP response to RAS blockade. Notably, these conclusions were reached in a carefully characterized, homogenous population, and when taking into account the known fluctuations in renin that relate to gender, age, ethnicity, salt intake and diuretic treatment, it seems unlikely that a cut-off renin level can be defined that has predictive value.




Similar content being viewed by others
References
Brugts JJ, Isaacs A, Boersma E, van Duijn CM, Uitterlinden AG, Remme W, et al. Genetic determinants of treatment benefit of the angiotensin-converting enzyme-inhibitor perindopril in patients with stable coronary artery disease. Eur Heart J. 2010;31:1854–64.
Brugts JJ, Isaacs A, de Maat MP, Boersma E, van Duijn CM, Akkerhuis KM, et al. A pharmacogenetic analysis of determinants of hypertension and blood pressure response to angiotensin-converting enzyme inhibitor therapy in patients with vascular disease and healthy individuals. J Hypertens. 2011;29:509–19.
Costa-Scharplatz M, van Asselt AD, Bachmann LM, Kessels AG, Severens JL. Cost-effectiveness of pharmacogenetic testing to predict treatment response to angiotensin-converting enzyme inhibitor. Pharmacogenet Genomics. 2007;17:359–68.
Danser AHJ, Batenburg WW, van den Meiracker AH, Danilov SM. ACE phenotyping as a first step toward personalized medicine for ACE inhibitors. Why does ACE genotyping not predict the therapeutic efficacy of ACE inhibition? Pharmacol Ther. 2007;113:607–18.
Mattace-Raso FU, Sie MP, van der Cammen TJ, Safar ME, Hofman A, van Duijn CM, et al. Insertion/deletion gene polymorphism of the angiotensin-converting enzyme and blood pressure changes in older adults. The Rotterdam study. J Hum Hypertens. 2007;21:736–40.
Harrap SB, Tzourio C, Cambien F, Poirier O, Raoux S, Chalmers J, et al. The ACE gene I/D polymorphism is not associated with the blood pressure and cardiovascular benefits of ACE inhibition. Hypertension. 2003;42:297–303.
Bhatnagar V, O’Connor DT, Schork NJ, Salem RM, Nievergelt CM, Rana BK, et al. Angiotensin-converting enzyme gene polymorphism predicts the time-course of blood pressure response to angiotensin converting enzyme inhibition in the AASK trial. J Hypertens. 2007;25:2082–92.
Lely AT, Heerspink HJ, Zuurman M, Visser FW, Kocks MJ, Boomsma F, et al. Response to angiotensin-converting enzyme inhibition is selectively blunted by high sodium in angiotensin-converting enzyme DD genotype: evidence for gene-environment interaction in healthy volunteers. J Hypertens. 2010;28:2414–21.
Schwartz GL, Turner ST, Chapman AB, Boerwinkle E. Interacting effects of gender and genotype on blood pressure response to hydrochlorothiazide. Kidney Int. 2002;62:1718–23.
Tu W, Eckert GJ, Pratt JH, Danser AHJ. Plasma levels of prorenin and renin in blacks and whites: their relative abundance and associations with plasma aldosterone concentration. Am J Hypertens. 2012;25:1030–4.
Rice GI, Jones AL, Grant PJ, Carter AM, Turner AJ, Hooper NM. Circulating activities of angiotensin-converting enzyme, its homologue, angiotensin-converting enzyme 2, and neprilysin in a family study. Hypertension. 2006;48:914–20.
Alhenc-Gelas F, Richard J, Courbon D, Warnet JM, Corvol P. Distribution of plasma angiotensin I-converting enzyme levels in healthy men: relationship to environmental and hormonal parameters. J Lab Clin Med. 1991;117:33–9.
Danser AHJ, Schalekamp MADH, Bax WA, van den Brink AM, Saxena PR, Riegger GAJ, et al. Angiotensin-converting enzyme in the human heart. Effect of the deletion/insertion polymorphism. Circulation. 1995;92:1387–8.
Persu A, Lambert M, Deinum J, Cossu M, de Visscher N, Irenge L, et al. A novel splice-site mutation in angiotensin I-converting enzyme (ACE) gene, c.3691 + 1G > A (IVS25 + 1G > A), causes a dramatic increase in circulating ACE through deletion of the transmembrane anchor. PLoS One. 2013;8:e59537.
Tom B, Garrelds IM, Scalbert E, Stegmann APA, Boomsma F, Saxena PR, et al. ACE- versus chymase-dependent angiotensin II generation in human coronary arteries: a matter of efficiency? Arterioscler Thromb Vasc Biol. 2003;23:251–6.
Alexiou T, Boon WM, Denton DA, Nicolantonio RD, Walker LL, McKinley MJ, et al. Angiotensinogen and angiotensin-converting enzyme gene copy number and angiotensin and bradykinin peptide levels in mice. J Hypertens. 2005;23:945–54.
Danser AHJ. Renin and prorenin as biomarkers in hypertension. Curr Opin Nephrol Hypertens. 2012;21:508–14.
Laragh JH, Sealey JE. The plasma renin test reveals the contribution of body sodium-volume content (V) and renin-angiotensin (R) vasoconstriction to long-term blood pressure. Am J Hypertens. 2011;24:1164–80.
Nussberger J, Gradman AH, Schmieder RE, Lins RL, Chiang Y, Prescott MF. Plasma renin and the antihypertensive effect of the orally active renin inhibitor aliskiren in clinical hypertension. Int J Clin Pract. 2007;61:1461–8.
Stanton AV, Dicker P, O’Brien ET. Aliskiren monotherapy results in the greatest and the least blood pressure lowering in patients with high- and low-baseline PRA levels, respectively. Am J Hypertens. 2009;22:954–7.
Azizi M, Bissery A, Lamarre-Cliche M, Ménard J. Integrating drug pharmacokinetics for phenotyping individual renin response to angiotensin II blockade in humans. Hypertension. 2004;43:785–90.
Jorde UP, Ennezat PV, Lisker J, Suryadevara V, Infeld J, Cukon S, et al. Maximally recommended doses of angiotensin-converting enzyme (ACE) inhibitors do not completely prevent ACE-mediated formation of angiotensin II in chronic heart failure. Circulation. 2000;101:844–6.
Krop M, Lu X, Verdonk K, Schalekamp MADH, van Gool JM, McKeever BM, et al. New renin inhibitor VTP-27999 alters renin immunoreactivity and does not unfold prorenin. Hypertension. 2013;61:1075–82.
Danilov SM, Balyasnikova IV, Albrecht 2nd RF, Kost OA. Simultaneous determination of ACE activity with 2 substrates provides information on the status of somatic ACE and allows detection of inhibitors in human blood. J Cardiovasc Pharmacol. 2008;52:90–103.
Danilov SM, Balyasnikova IV, Danilova AS, Naperova IA, Arablinskaya NE, Borisov SE, et al. Conformational fingerprinting of the angiotensin I-converting enzyme (ACE). 1. Application in sarcoidosis. J Proteome Res. 2010;9:5782–93.
Gordon K, Balyasnikova IV, Nesterovitch AB, Schwartz DE, Sturrock ED, Danilov SM. Fine epitope mapping of monoclonal antibodies 9B9 and 3G8 to the N domain of angiotensin-converting enzyme (CD143) defines a region involved in regulating angiotensin-converting enzyme dimerization and shedding. Tissue Antigens. 2010;75:136–50.
Klotz S, Burkhoff D, Garrelds IM, Boomsma F, Danser AHJ. The impact of left ventricular assist device-induced left ventricular unloading on the myocardial renin-angiotensin-aldosterone system: therapeutic consequences? Eur Heart J. 2009;30:805–12.
Fraune C, Lange S, Krebs C, Holzel A, Baucke J, Divac N, et al. AT1 antagonism and renin inhibition in mice: pivotal role of targeting angiotensin II in chronic kidney disease. Am J Physiol Renal Physiol. 2012;303:F1037–1048.
van Kats JP, Schalekamp MADH, Verdouw PD, Duncker DJ, Danser AHJ. Intrarenal angiotensin II: interstitial and cellular levels and site of production. Kidney Int. 2001;60:2311–7.
Metzger R, Bohle RM, Pauls K, Eichner G, Alhenc-Gelas F, Danilov SM, et al. Angiotensin-converting enzyme in non-neoplastic kidney diseases. Kidney Int. 1999;56:1442–54.
Hollenberg NK, Fisher ND, Nussberger J, Moukarbel GV, Barkoudah E, Danser AHJ. Renal responses to three types of renin-angiotensin system blockers in patients with diabetes mellitus on a high-salt diet: a need for higher doses in diabetic patients? J Hypertens. 2011;29:2454–61.
Price DA, Porter LE, Gordon M, Fisher NDL, De’Oliveira JM, Laffel LM, et al. The paradox of the low-renin state in diabetic nephropathy. J Am Soc Nephrol. 1999;10:2382–91.
Schrier RW. Aldosterone ‘escape’ vs ‘breakthrough’. Nat Rev Nephrol. 2010;6:61.
Sevá Pessôa B, van der Lubbe N, Verdonk K, Roks AJM, Hoorn EJ, Danser AHJ. Key developments in renin-angiotensin-aldosterone system inhibition. Nat Rev Nephrol. 2013;9:26–36.
de Boer RA, Azizi M, Danser AHJ, Nguyen G, Nussberger J, Ruilope LM, et al. Dual RAAS suppression: recent developments and implications in light of the ALTITUDE study. J Renin-Angiotensin-Aldosterone Syst. 2012;13:409–12.
Parving HH, Brenner BM, McMurray JJ, de Zeeuw D, Haffner SM, Solomon SD, et al. Cardiorenal end points in a trial of aliskiren for type 2 diabetes. N Engl J Med. 2012;367:2204–13.
Telmisartan Randomised AssessmeNt Study in ACE-iNtolerant subjects with cardiovascular Disease (TRANSCEND) Investigators, Yusuf S, Teo K, Anderson C, Pogue J, Dyal L, et al. Effects of the angiotensin-receptor blocker telmisartan on cardiovascular events in high-risk patients intolerant to angiotensin-converting enzyme inhibitors: a randomised controlled trial. Lancet. 2008;372:1174–83.
Danser AHJ, Derkx FHM, Schalekamp MADH, Hense HW, Riegger GAJ, Schunkert H. Determinants of interindividual variation of renin and prorenin concentrations: evidence for a sexual dimorphism of (pro)renin levels in humans. J Hypertens. 1998;16:853–62.
Pizzolo F, Raffaelli R, Memmo A, Chiecchi L, Pavan C, Guarini P, et al. Effects of female sex hormones and contraceptive pill on the diagnostic work-up for primary aldosteronism. J Hypertens. 2010;28:135–42.
Newton-Cheh C, Guo CY, Gona P, Larson MG, Benjamin EJ, Wang TJ, et al. Clinical and genetic correlates of aldosterone-to-renin ratio and relations to blood pressure in a community sample. Hypertension. 2007;49:846–56.
Boomsma F, de Bruyn JHB, Derkx FHM, Schalekamp MADH. Opposite effects of captopril on angiotensin I-converting enzyme ‘activity’ and ‘concentration’; relation between enzyme inhibition and long-term blood pressure response. Clin Sci (Lond). 1981;60:491–8.
Kost OA, Balyasnikova IV, Chemodanova EE, Nikolskaya II, Albrecht 2nd RF, Danilov SM. Epitope-dependent blocking of the angiotensin-converting enzyme dimerization by monoclonal antibodies to the N-terminal domain of ACE: possible link of ACE dimerization and shedding from the cell surface. Biochemistry. 2003;42:6965–76.
Schunkert H, Ingelfinger JR, Hirsch AT, Pinto Y, Remme WJ, Jacob H, et al. Feedback regulation of angiotensin converting enzyme activity and mRNA levels by angiotensin II. Circ Res. 1993;72:312–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schilders, J.E., Wu, H., Boomsma, F. et al. Renin-Angiotensin System Phenotyping as a Guidance Toward Personalized Medicine for ACE Inhibitors: can the Response to ACE Inhibition be Predicted on the Basis of Plasma Renin or ACE?. Cardiovasc Drugs Ther 28, 335–345 (2014). https://doi.org/10.1007/s10557-014-6537-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10557-014-6537-6