Abstract
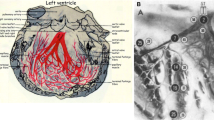
In cardiac resynchronization therapy (CRT), specific changes in motion/deformation happen with left-bundle-branch-block (LBBB) and following treatment. However, they remain sub-optimally studied. We propose a two-fold improvement of their characterization. This includes controlling them through an experimental model and using more suitable quantification techniques. We used a swine model of acute LBBB and CRT with/without chronic infarct (pure-LBBB: N = 11; LBBB + left-anterior-descending infarct: N = 11). Myocardial displacement, velocity and strain were extracted from short-axis echocardiographic sequences using 2D speckle-tracking. The data was transformed to a single spatiotemporal system of coordinates to perform subject comparisons and quantify pattern changes at similar locations and instants. Pure-LBBB animals showed a specific intra-ventricular dyssynchrony pattern with LBBB (11/11 animals), and the recovery towards a normal pattern with CRT (10/11 animals). Pattern variability was low within the pure-LBBB population, as quantified by our method. This was not correctly assessed by more conventional measurements. Infarct presence affected the pattern distribution and CRT efficiency (improvements in 6/11 animals). Pattern changes correlated with global cardiac function (global circumferential strain) changes in all the animals (corrected: pLBBBvsBaseline < 0.001, pCRTvsBaseline = NS; non-corrected: pLBBBvsBaseline = NS, pCRTvsBaseline = 0.028). Our LBBB/CRT experimental model allowed controlling specific factors responsible for changes in mechanical dyssynchrony and therapy. We illustrated the importance of our quantification method to study these changes and their variability. Our findings confirm the importance of myocardial viability and of specific LBBB-related mechanical dyssynchrony patterns.






Similar content being viewed by others
Notes
Strain is a spatially differential measure and is not affected by this correction.
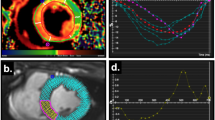
The temporal alignment required the identification of 6 events per cycle. The onset of QRS (beginning and ending of the cycle) was identified on the ECG. Valve events (aortic/mitral valve opening/closure [AVO/AVC/MVO/MVC]) were identified using continuous-wave Doppler imaging on the corresponding valve, when possible, or visually on short-axis 2D views.
NS: Non-significant statistical difference (p value > 0.05).
References
Bijnens BH, Cikes M, Butakoff C, Sitges M, Crispi F (2012) Myocardial motion and deformation: what does it tell us and how does it relate to function? Fetal Diagn Ther 32:5–16. doi:10.1159/000335649
Fornwalt BK, Delfino JG, Sprague WW, Oshinski JN (2009) It’s time for a paradigm shift in the quantitative evaluation of left ventricular dyssynchrony. J Am Soc Echocardiogr 22:672–676. doi:10.1016/j.echo.2009.04.012
Fornwalt BK (2011) The dyssynchrony in predicting response to cardiac resynchronization therapy: a call for change. J Am Soc Echocardiogr 24:180–184. doi:10.1016/j.echo.2010.12.015
Fornwalt BK, Sprague WW, BeDell P, Suever JD, Gerritse B, Merlino JD, Fyfe DA, León AR, Oshinski JN (2010) Agreement is poor among current criteria used to define response to cardiac resynchronization therapy. Circulation 121:1985–1991. doi:10.1161/CIRCULATIONAHA.109.910778
Cleland JG, Ghio S (2012) The determinants of clinical outcome and clinical response to CRT are not the same. Heart Fail Rev 17:755–766. doi:10.1007/s10741-011-9268-9
Parsai C, Bijnens B, Sutherland GR, Baltabaeva A, Claus P, Marciniak M, Paul V, Scheffer M, Donal E, Derumeaux G, Anderson L (2009) Toward understanding response to cardiac resynchronization therapy: left ventricular dyssynchrony is only one of multiple mechanisms. Eur Heart J 30:940–949. doi:10.1093/eurheartj/ehn481
Leenders GE, Lumens J, Cramer MJ, De Boeck BW, Doevendans PA, Delhaas T, Prinzen FW (2012) Septal deformation patterns delineate mechanical dyssynchrony and regional differences in contractility: analysis of patient data using a computer model. Circ Heart Fail 5:87–96. doi:10.1161/CIRCHEARTFAILURE.111.962704
De Boeck BW, Teske AJ, Meine M, Leenders GE, Cramer MJ, Prinzen FW, Doevendans PA (2009) Septal rebound stretch reflects the functional substrate to cardiac resynchronization therapy and predicts volumetric and neurohormonal response. Eur J Heart Fail 11:863–871. doi:10.1093/eurjhf/hfp107
Szulik M, Tillekaerts M, Vangeel V, Ganame J, Willems R, Lenarczyk R, Rademakers F, Kalarus Z, Kukulski T, Voigt JU (2010) Assessment of apical rocking: a new, integrative approach for selection of candidates for cardiac resynchronization therapy. Eur J Echocardiogr 11:863–869. doi:10.1093/ejechocard/jeq081
Risum N, Jons C, Olsen NT, Fritz-Hansen T, Bruun NE, Hojgaard MV, Valeur N, Kronborg MB, Kisslo J, Sogaard P (2012) Simple regional strain pattern analysis to predict response to cardiac resynchronization therapy: rationale, initial results, and advantages. Am Heart J 163:697–704. doi:10.1016/j.ahj.2012.01.025
Prinzen FW, Peschar M (2002) Relation between the pacing induced sequence of activation and left ventricular pump function in animals. Pacing Clin Electrophysiol 25:484–498. doi:10.1046/j.1460-9592.2002.00484.x
Strik M, van Middendorp LB, Vernooy K (2012) Animal models of dyssynchrony. J Cardiovasc Transl Res 5:135–145. doi:10.1007/s12265-011-9336-5
Rademakers LM, van Kerckhoven R, van Deursen CJ, Strik M, van Hunnik A, Kuiper M, Lampert A, Klersy C, Leyva F, Auricchio A, Maessen JG, Prinzen FW (2010) Myocardial infarction does not preclude electrical and hemodynamic benefits of cardiac resynchronization therapy in dyssynchronous canine hearts. Circ Arrhythm Electrophysiol 3:361–368. doi:10.1161/CIRCEP.109.931865
Duchateau N, De Craene M, Piella G, Silva E, Doltra A, Sitges M, Bijnens BH, Frangi AF (2011) A spatiotemporal statistical atlas of motion for the quantification of abnormal myocardial tissue velocities. Med Image Anal 15:316–328. doi:10.1016/j.media.2010.12.006
Rigol M, Solanes N, Farré J, Roura S, Roqué M, Berruezo A, Bellera N, Novensà L, Tamborero D, Prat-Vidal C, Huzman MA, Batlle M, Hoefsloot M, Sitges M, Ramírez J, Dantas AP, Merino A, Sanz G, Brugada J, Bayés-Genís A, Heras M (2010) Effects of adipose tissue-derived stem cell therapy after myocardial infarction: impact of the route of administration. J Card Fail 16:357–366. doi:10.1016/j.cardfail.2009
Rigol M, Solanes N, Fernandez-Armenta J, Silva E, Doltra A, Duchateau N, Barcelo A, Gabrielli L, Bijnens B, Berruezo A, Brugada J, Sitges M (2013) Development of a swine model of left bundle branch block for experimental studies of cardiac resynchronization therapy. J Cardiovasc Transl Res 6:616–622. doi:10.1007/s12265-013-9464-1
National Research Council (2011) Guide for the care and use of laboratory animals, 8th edn. The National Academies Press, Washington, DC
Duchateau N, De Craene M, Pennec X, Merino B, Sitges M, Bijnens BH (2012) Which reorientation framework for the atlas-based comparison of motion from cardiac image sequences? In: Proceedings of spatio-temporal image analysis for longitudinal and time-series image data, MICCAI’12 workshop. Springer LNCS vol. 7570, pp 25–37. doi:10.1007/978-3-642-33555-6_3
De Craene M, Duchateau N, Tobon-Gomez C, Ghafaryasl B, Piella G, Rhode KS, Frangi AF (2012) SPM to the heart: mapping of 4D continuous velocities for motion abnormality quantification. In: Proceedings of IEEE international symposium on biomedical imaging, pp 454-457. doi:10.1109/ISBI.2012.6235582
Duchateau N, Doltra A, Silva E, De Craene M, Piella G, Castel MA, Mont L, Brugada J, Frangi AF, Sitges M (2012) Atlas-based quantification of myocardial motion abnormalities: added-value for the understanding the effect of cardiac resynchronization therapy. Ultrasound Med Biol 38:2186–2197. doi:10.1016/j.ultrasmedbio.2012.08.009
Delgado V, Ypenburg C, van Bommel RJ, Tops LF, Mollema SA, Marsan NA, Bleeker GB, Schalij MJ, Bax JJ (2008) Assessment of left ventricular dyssynchrony by speckle tracking strain imaging comparison between longitudinal, circumferential, and radial strain in cardiac resynchronization therapy. J Am Coll Cardiol 51:1944–1952. doi:10.1016/j.jacc.2008.02.040
Kydd AC, Khan FZ, O’Halloran D, Pugh PJ, Virdee MS, Dutka DP (2013) Radial strain delay based on segmental timing and strain amplitude predicts left ventricular reverse remodeling and survival after cardiac resynchronization therapy. Circ Cardiovasc Imaging 6:177–184. doi:10.1161/CIRCIMAGING.112.000191
Anderson LJ, Miyazaki C, Sutherland GR, Oh JK (2008) Patient selection and echocardiographic assessment of dyssynchrony in cardiac resynchronization therapy. Circulation 117:1023–2009. doi:10.1161/CIRCULATIONAHA.107.721332
Delgado V, Bax JJ (2011) Assessment of systolic dyssynchrony for cardiac resynchronization therapy is clinically useful. Circulation 123:640–655. doi:10.1161/CIRCULATIONAHA.110.954404
Sung RK, Foster E (2011) Assessment of systolic dyssynchrony for cardiac resynchronization therapy is not clinically useful. Circulation 123:656–662. doi:10.1161/CIRCULATIONAHA.110.954420
Silva E, Sitges M, Doltra A, Mont L, Vidal B, Castel MA, Tolosana JM, Berruezo A, González Juanatey JR, Brugada J (2010) Analysis of temporal delay in myocardial deformation throughout the cardiac cycle: utility for selecting candidates for cardiac resynchronization therapy. Heart Rhythm 7:1580–1586. doi:10.1016/j.hrthm.2010.06.034
Bertola B, Rondano E, Sulis M, Sarasso G, Piccinino C, Marti G, Devecchi P, Magnani A, Francalacci G, Marino PN (2009) Cardiac dyssynchrony quantitated by time-to-peak or temporal uniformity of strain at longitudinal, circumferential, and radial level: implications for resynchronization therapy. J Am Soc Echocardiogr 22:665–671. doi:10.1016/j.echo.2009.03.010
Chan YH, Kuo CT, Yeh YH, Wu LS, Wang CL, Ho WJ, Hsu LA (2013) Incremental value of radial discoordination index for the prediction of response to cardiac resynchronization therapy. Eur Heart J 14:213–222. doi:10.1093/ehjci/jes112
Tobon-Gomez C, Duchateau N, Sebastian R, Marchesseau S, Camara O, Donal E, De Craene M, Pashaei A, Relan J, Steghofer M, Lamata P, Delingette H, Duckett S, Garreau M, Hernandez A, Rhode KS, Sermesant M, Ayache N, Leclercq C, Razavi R, Smith NP, Frangi AF (2013) Understanding the mechanisms amenable to CRT response: from pre-operative multimodal image data to patient-specific computational models. Med Biol Eng Comput 51:1235–1250. doi:10.1007/s11517-013-1044-7
Ring M, Persson H, Mejhert M, Edner M (2007) Post-systolic motion in patients with heart failure—a marker of left ventricular dyssynchrony? Eur J Echocardiogr 8:352–359. doi:10.1016/j.euje.2006.07.006
McDonald IG (1973) Echocardiographic demonstration of abnormal motion of the interventricular septum in left bundle branch block. Circulation 48:272–280. doi:10.1161/01.CIR.48.2.272
Dillon JC, Chang S, Feigenbaum H (1974) Echocardiographic manifestations of left-bundle branch-block. Circulation 49:876–880. doi:10.1161/01.CIR.49.5.876
Little WC, Reeves RC, Arciniegas J, Katholi RE, Rogers EW (1982) Mechanism of abnormal inter-ventricular septal motion during delayed left-ventricular activation. Circulation 65:1486–1491. doi:10.1161/01.CIR.65.7.1486
Grines CL, Bashore TM, Boudoulas H, Olson S, Shafer P, Wooley CF (1989) Functional abnormalities in isolated left bundle branch block. The effect of interventricular asynchrony. Circulation 79:845–853. doi:10.1161/01.CIR.79.4.845
Kerckhoffs RC, Omens JH, McCulloch AD (2012) Mechanical discoordination increases continuously after the onset of left bundle branch block despite constant electrical dyssynchrony in a computational model of cardiac electromechanics and growth. Europace 14:v65–v72. doi:10.1093/europace/eus274
Gjesdal O, Remme EW, Opdahl A, Skulstad H, Russell K, Kongsgaard E, Edvardsen T, Smiseth OA (2011) Mechanisms of abnormal systolic motion of the interventricular septum during left bundle-branch block. Circ Cardiovasc Imaging 4:264–273. doi:10.1161/CIRCIMAGING.110.961417
Duckett SG, Camara O, Ginks MR, Bostock J, Chinchapatnam P, Sermesant M, Pashaei A, Lambiase PD, Gill JS, Carr-White GS, Frangi AF, Razavi R, Bijnens BH, Rinaldi CA (2012) Relationship between endocardial activation sequences defined by high-density mapping to early septal contraction (septal flash) in patients with left bundle branch block undergoing cardiac resynchronization therapy. Europace 14:99–106. doi:10.1093/europace/eur235
Risum N, Strauss D, Sogaard P, Loring Z, Fritz-Hansen T, Bruun NE, Wagner G, Kisslo J (2013) Left bundle-branch block: the relationship between electrocardiogram electrical activation and echocardiography mechanical contraction. Am Heart J 166:340–348. doi:10.1016/j.ahj.2013.04.005
Parsai C, Baltabaeva A, Anderson L, Chaparro M, Bijnens BH, Sutherland GR (2009) Low-dose dobutamine stress echo to quantify the degree of remodelling after cardiac resynchronization therapy. Eur Heart J 30:950–958. doi:10.1093/eurheartj/ehp050
Duchateau N, Bijnens BH, D’hooge J, Sitges M (2013) Three-dimensional assessment of cardiac motion and deformation. In: Shiota T (ed) 3D echocardiography, 2nd edn. CRC Press, Boca Raton, pp 201–213
Acknowledgments
This work was partially supported by the Centre for the Development of Industrial Technology, Madrid, Spain (cvREMOD CEN-20091044) and the European Union Seventh Framework Programme (VP2HF FP7-2013-611823). AD had a post-residency grant from Fundació Clínic, Barcelona, Spain. The authors acknowledge David Andreu and Santiago Sotes (Hospital Clínic, Barcelona, Spain), Daniel Traver (Biosense Webster, Barcelona, Spain), Laia Bayarri and Filippo Ziglio (Sorin Group, Barcelona/Milan, Spain/Italy) for their support on the experimental devices.
Conflict of interest
None declared.
Author information
Authors and Affiliations
Corresponding author
Additional information
Josep Brugada and Bart Bijnens share senior authorship.
Rights and permissions
About this article
Cite this article
Duchateau, N., Sitges, M., Doltra, A. et al. Myocardial motion and deformation patterns in an experimental swine model of acute LBBB/CRT and chronic infarct. Int J Cardiovasc Imaging 30, 875–887 (2014). https://doi.org/10.1007/s10554-014-0403-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10554-014-0403-2