Abstract
Purpose
Studies of cancer patient survival typically report relative survival or cause-specific survival using data from patients diagnosed many years in the past. From a risk-communication perspective, such measures are suboptimal for several reasons; their interpretation is not transparent for non-specialists, competing causes of death are ignored and the estimates are unsuitable to predict the outcome of newly diagnosed patients. In this paper, we discuss the relative merits of recently developed alternatives to traditionally reported measures of cancer patient survival.
Methods
In a relative survival framework, using a period approach, we estimated probabilities of death in the presence of competing risks. To illustrate the methods, we present estimates of survival among 23,353 initially untreated, or hormonally treated men with intermediate- or high-risk localized prostate cancer using Swedish population-based data.
Results
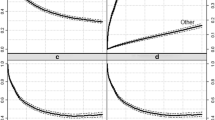
Among all groups of newly diagnosed patients, the probability of dying from prostate cancer, accounting for competing risks, was lower compared to the corresponding estimates where competing risks were ignored. Accounting for competing deaths was particularly important for patients aged more than 70 years at diagnosis in order to avoid overestimating the risk of dying from prostate cancer.
Conclusions
We argue that period estimates of survival, accounting for competing risks, provide the tools to communicate the actual risk that cancer patients, diagnosed today, face to die from their disease. Such measures should offer a more useful basis for risk communication between patients and clinicians and we advocate their use as means to answer prognostic questions.




Similar content being viewed by others
References
Dickman PW, Adami HO (2006) Interpreting trends in cancer patient survival. J Intern Med 260:103–117
Perme MP, Stare J, Esteve J (2012) On estimation in relative survival. Biometrics 68:113–120
Dignam JJ, Kocherginsky MN (2008) Choice and interpretation of statistical tests used when competing risks are present. J Clin Oncol 26:4027–4034
Putter H, Fiocco M, Geskus RB (2007) Tutorial in biostatistics: competing risks and multi-state models. Stat Med 26:2389–2430
Gail MH (2008) Estimation and interpretation of models of absolute risk from epidemiologic data, including family-based studies. Lifetime Data Anal 14:18–36
Adolfsson J, Garmo H, Varenhorst E, Ahlgren G, Ahlstrand C, Andren O, Bill-Axelsson BO, Damber JE, Hellström K, Hellström M, Holmberg E et al (2007) Clinical characteristics and primary treatment of prostate cancer in Sweden between 1996 and 2005. Scand J Urol Nephrol 2007(41):456–477
National Comprehensive Cancer Network (2010) Clinical practice guidelines in oncology, Prostate cancer, Version 1.2010
D’Amico AV, Denham JW, Crook J, Chen MH, Goldhaber SZ, Lamb DS, Joseph D, Tai KH, Malone S, Ludgate C, Steigler A, Kantoff PW (2007) Influence of androgen suppression therapy for prostate cancer on the frequency and timing of fatal myocardial infarctions. J Clin Oncol 25:2420–2425
Tsai HK, D’Amico AV, Sadetsky N, Chen MH, Carroll PR (2007) Androgen deprivation therapy for localized prostate cancer and the risk of cardiovascular mortality. J Natl Cancer Inst 99:1516–1524
Van Hemelrijck M, Garmo H, Holmberg L, Ingelsson E, Bratt O, Bill-Axelson A, Lambe M, Stattin P, Adolfsson J (2010) Absolute and relative risk of cardiovascular disease in men with prostate cancer: results from the Population-Based PCBaSe Sweden. J Clin Oncol 28:3448–3456
Fang F, Fall K, Mittleman MA, Sparen P, Ye W, Adami HO, Valdimarsdóttir U (2012) Suicide and cardiovascular death after a cancer diagnosis. N Engl J Med 366:1310–1318
Brenner H, Gefeller O, Hakulinen T (2004) Period analysis for ‘up-to-date’ cancer survival data: theory, empirical evaluation, computational realisation and applications. Eur J Cancer 40:326–335
Brenner H, Hakulinen T (2002) Advanced detection of time trends in long-term cancer patient survival: experience from 50 years of cancer registration in Finland. Am J Epidemiol 156:566–577
Dickman PW, Sloggett A, Hills M, Hakulinen T (2004) Regression models for relative survival. Stat Med 23:51–64
Perme MP, Henderson R, Stare J (2009) An approach to estimation in relative survival regression. Biostatistics 10:136–146
Nelson CP, Lambert PC, Squire IB, Jones DR (2007) Flexible parametric models for relative survival, with application in coronary heart disease. Stat Med 26:5486–5498
Lambert PC (2009) Further development of flexible parametric models for survival analysis. Stata J 9:265–290
Durrleman S, Simon R (1989) Flexible regression models with cubic splines. Stat Med 8:551–561
Lambert PC, Dickman PW, Nelson CP, Royston P (2010) Estimating the crude probability of death due to cancer and other causes using relative survival models. Stat Med 29:885–895
Brenner H, Arndt V (2005) Long-term survival rates of patients with prostate cancer in the prostate-specific antigen screening era: population-based estimates for the year 2000 by period analysis. J Clin Oncol 23:441–447
Holmberg L, Robinson D, Sandin F, Bray F, Linklater KM, Klint A, Lambert PC, Adolfsson J, Hamdy FC, Catto J, Møller H (2012) A comparison of prostate cancer survival in England, Norway and Sweden: a population-based study. Cancer Epidemiol 36:e7–e12
Stattin P, Holmberg E, Johansson JE, Holmberg L, Adolfsson J, Hugosson J (2010) Outcomes in localized prostate cancer: National Prostate Cancer Register of Sweden follow-up study. J Natl Cancer Inst 102:950–958
Hinchliffe SR, Abrams KR, Lambert PC (2012) The impact of under and over-recording of cancer on death certificates in a competing risks analysis: a simulation study. Cancer Epidemiol (Epub ahead of print). doi:10.1016/j.canep.2012.08.012
Howlader N, Ries LA, Mariotto AB, Reichman ME, Ruhl J, Cronin KA (2010) Improved estimates of cancer-specific survival rates from population-based data. J Natl Cancer Inst 102:1584–1598
Finnish Cancer Registry-Institute for Statistical and Epidemiological Cancer Research (2009) Cancer in Finland 2006 and 2007. Helsinki 2009. Publication No. 76
Koller MT, Raatz H, Steyerberg EW, Wolbers M (2011) Competing risks and the clinical community: irrelevance or ignorance? Stat Med 31:1089–1097
Feuer EJ, Lee M, Mariotto AB, Cronin KA, Scoppa S, Penson DF et al (2012) The Cancer Survival Query System: making survival estimates from the Surveillance, Epidemiology, and End Results program more timely and relevant for recently diagnosed patients. Cancer 118:5652–5662
Brenner H, Hakulinen T (2003) On crude and age-adjusted relative survival rates. J Clin Epidemiol 56:1185–1191
Pokhrel A, Hakulinen T (2008) How to interpret the relative survival ratios of cancer patients. Eur J Cancer 44:2661–2667
Fagerlin A, Zikmund-Fisher BJ, Ubel PA (2011) Helping patients decide: ten steps to better risk communication. J Natl Cancer Inst 103:1436–1443
Lipkus IM (2007) Numeric, verbal, and visual formats of conveying health risks: suggested best practices and future recommendations. Med Decis Mak 27:696–713
Lambert PC, Royston P (2009) Further development of flexible parametric models for survival analysis. Stata J 9:265–290
Acknowledgments
This project was made possible by the continuous work of the National Prostate Cancer Register of Sweden steering group: Pär Stattin chair, Anders Widmark, Stefan Carlsson, Magnus Törnblom, Jan Adolfsson, Anna Bill-Axelson, Ove Andrén, David Robinson, Bill Pettersson, Jonas Hugosson, Jan-Erik Damber, Ola Bratt, Göran Ahlgren, Lars Egevad, and Mats Lambe. This work was supported by the Swedish Cancer Society (grant number CAN 2010/676).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Eloranta, S., Adolfsson, J., Lambert, P.C. et al. How can we make cancer survival statistics more useful for patients and clinicians: An illustration using localized prostate cancer in Sweden. Cancer Causes Control 24, 505–515 (2013). https://doi.org/10.1007/s10552-012-0141-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10552-012-0141-5